Introduction
Histoplasmosis is an infectious disease caused by inhaling microconidia or small hyphae of the Histoplasma capsulatum fungus, a dimorphic microorganism found in bird and bat guano 1. It should be noted that it is thought to be the most common systemic and endemic mycosis in the world, with a practically global presence and an approximate incidence of 500,000 cases per year, worldwide. Its mortality ranges from approximately 12 to 39% in the United States and 13 to 43% in Latin America 2. In Colombia, the 19922008 national survey reported that 70.5% of the patients were adults with an HIV/AIDS diagnosis, and another 7% with another type of immunosuppression 3. In another observational study of a series of cases in a hospital in Bogotá (Colombia) from January 2012 to December 2016, a total of 34 patients were evaluated, 73.5% of whom had a history of HIV infection 4. The clinical involvement in more than 90% of cases is pulmonary, which is usually asymptomatic or has nonspecific symptoms like a dry cough, fever, headache, and chest pain, with this clinical presentation very much influenced by the patients' immune status.
In one out of 2,000 cases of Histoplasma capsulatum, the disease may disseminate to different anatomical sites, like the gastrointestinal system in 50-70% of cases 5. Therefore, it is not uncommon to find infection at this site when the patient has a pulmonary primary focus, and especially in immunocompromised patients, particularly those with human immunodeficiency virus (HIV) infection, solid organ transplants, hematological cancers, on immunosuppressant treatment like corticosteroids or tumor necrosis factor antagonists, or with congenital T-cell immunodeficiency. However, it is rare to find isolated gastrointestinal histoplasmosis and, to date, cases have been described related to liver transplant and in one HIV positive patient with early colon cancer.
Below, we present the case of a 24-year-old patient with a history of systemic lupus erythematosus (SLE) being treated with high-dose prednisolone, who developed ulcerative colitis due to Histoplasma capsulatum, with no evidence of involvement of another anatomical site.
Clinical case
This was a 24-year-old female Afro-descendant patient from Buenaventura, Valle del Cauca. She had a history of SLE with recognized cutaneous involvement with discoid lupus, joint involvement with inflammatory polyarthralgia and Raynaud's phenomenon, and had received 50 mg of prednisone per day for several months due to persistent symptoms of active disease and low tolerance to steroid-sparing drugs. She also had a history of sickle cell trait.
She complained of 15 days with a fever of 38-39°C, predominantly in the evening, partially improved by taking acetaminophen, associated with moderate colicky abdominal pain in the flank and right iliac fossa, multiple episodes of rectal bleeding and hematochezia, emesis, inability to eat, arthralgias, general malaise and a 14 kg weight loss. Her physical exam on admission showed a blood pressure of 124/76 mmHg, heart rate of 100 beats per minute, 99% SO2 on room air, weight of 64 kg, intact oral mucosa, and colicky abdominal pain on palpation with no signs of peritoneal irritation.
The laboratory studies on admission showed a complete blood count with mild normocytic anemia, lymphopenia without leukopenia, a normal platelet count, and elevated serum creatinine; however, a prior level was not available to indicate how long it had been elevated. She had positive ANAs 1:2,560 with a speckled pattern and positive ENAs (Table 1).
Table 1 Diagnostic aids on admission.
| Diagnostic test | Admission | Performed during hospitalization | Prior to discharge |
|---|---|---|---|
| Kidney function | |||
| Creatinine (mg/dl) | 3.22 | 2.29 | 2.0 |
| Blood urea nitrogen (mg/dL) | 26 | 24 | 22 |
| Spot urine creatinine | 149 | 86 | 57 |
| Alb/creat ratio (mg/g) | 320.40 | ||
| Spot microalbuminuria (mg/L) | 466 | ||
| Complete blood count | |||
| Leukocytes | 6,760 | 9,300 | 9,400 |
| Neutrophils | 6,090 | 6,440 | 5,600 |
| Lymphocytes | 530 | 750 | 1,090 |
| Hemoglobin (mg/dl) | 10.6 | 6.25 | 8.5 |
| MCV (fl) | 91.4 | 87 | |
| MCH | 29.1 | ||
| MCHC | 31.8 | ||
| RDW (%) | 15.1 | 17 | 17.1 |
| Platelets | 272,000 | 103,000 | 236,000 |
| Lipid profile | |||
| Total cholesterol (mg/dL) | 145 | ||
| Triglycerides (mg/dL) | 264 | ||
| HDL (mg/dL) | 23 | ||
| LDL (mg/dL) | 72 | ||
| VLDL (mg/dL) | 53 | ||
In light of the described symptoms, a colonoscopy was ordered which reported deep, irregular, friable ulcerated lesions in segments of the ascending and descending colon and an ulcer on the anal border. Biopsies were taken showing ulcerative colitis associated with Histoplasma capsulatum (Figure 1). An upper GI endoscopy with biopsy showed Kodsi grade III esophageal candidiasis and superficial gastric antral disease with no other pathological findings.
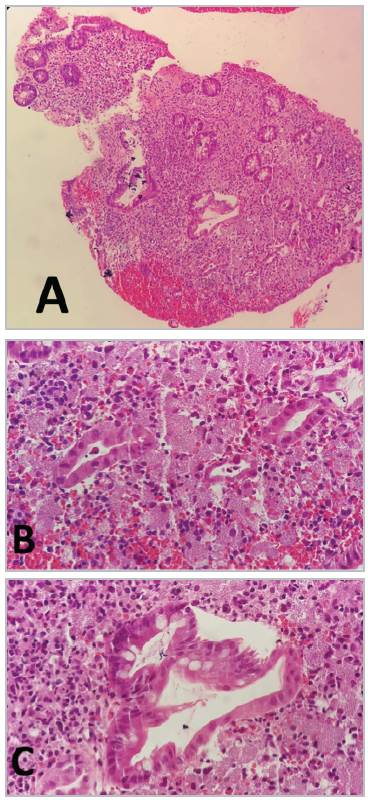
Figure 1 Histopathology A. 10x B. 40X C. 100X. Colon mucosa with widespread ulceration, covered by fibrinous material and neutrophils arranged in microabscesses; the crypt epithelium has reactive changes, the lamina propria is enlarged with mixed inflammatory infiltrate and numerous histiocytes with abundant cytoplasm and eosinophils with uncountable yeast-like structures characteristic of ulcerative colitis associated with histoplasmosis.
The patient was considered to have ulcerative colitis due to Histoplasma capsulatum and treatment was begun with liposomal amphotericin B. During her hospital stay she showed signs of a systemic inflammatory response, with an ESBL-positive germ isolated on urine culture. Due to the high risk of multiple organ failure and her history, she required comprehensive management by rheumatology and infectious disease, and therefore was referred to a higher-complexity institution where treatment was continued with liposomal amphotericin B.
During her hospital stay, she continued to have episodes of hematochezia and her laboratory tests showed multifactorial moderate normocytic anemia related to gastrointestinal hemorrhage, chronic autoimmune disease and the use of myelotoxic medications (amphotericin); she therefore required several blood product transfusions. Her immune profile was extended, showing complement consumption, negative anti-DNA antibodies, elevated nitrogen compounds and proteinuria, and therefore a kidney biopsy was ordered showing evidence of type 2 AI 1, CI lupus nephritis. With this, the patient was diagnosed with lupus reactivation and immunosuppression secondary to chronic corticosteroid use (Table 2).
Table 2 Immune profile.
| Anti-Sm | 166 U |
| Anti-Ro | 108 U |
| Anti-La | 6 U |
| Anti-RNP | 172 U |
| ANA: 1/2,560 | 1/2,560 |
| ENAs | 166 U |
| Anti-native DNA | 1/80 U |
| CD3 | 428 |
| CD4 | 201 |
| CD8 | 199 |
| Complement C4 fraction | 10 |
| Complement C3 fraction | 59 |
| IgG | 10.5 |
| IgM | 0.5 |
| IgA | 0.5 |
Treatment was started with intravenous immunoglobulin for two days, since other immunosuppressant treatment could not be used due to active infections by opportunistic agents. The patient completed antifungal treatment with liposomal amphotericin B and, after 20 days in the hospital, she was discharged with stable hemoglobin levels, no fever, no evidence of lupus activity and no new episodes of gastrointestinal bleeding, with a prescription for prednisolone 10 mg/24 hours, chloroquine 250 mg/24 hours and itraconazole 300 mg/24 hours.
Discussion
As molecular biology techniques have advanced, the phylogenetics of the causal agent of histoplasmosis have been elucidated with increasing precision. Its current taxonomy includes it in the class Euriotiomycetes, order Onygenales, family Onygenacea, genus Ajellomyces - sexual phase - or A. capsulatus - asexual phase - and species Histoplasma1. Regarding the latter, while it was originally grouped in three varieties (capsulatum-duboisii-farciminosum), four cryptic species and eight clades are now included, with Histoplasma capsulatum (ss) and Histoplasma suramericanum being significant in Latin America 2.
There are a variety of clinical presentations ranging from asymptomatic/self-limited infection to more severe forms like progressive disseminated histoplasmosis and acute histoplasmosis, with the latter ones being more frequent in immunocompromised people (mainly those with affected adaptive immunity 7. Regarding systemic lupus erythematosus, surprisingly, the incidence and prevalence of histoplasmosis is very low, represented in limited evidence primarily based on case reports 6-8; furthermore, it tends to present atypically, with a higher incidence of gastrointestinal, cutaneous and central nervous system involvement 4-8,9. Gastrointestinal histoplasmosis deserves special mention, with an approximate incidence of 0.05% in immunocompetent patients, predominantly males, around the fifth decade of life 9-12. Data from the immunocompromised population is poorly defined, especially in the scenario of patients with SLE, in which histoplasmosis is not frequent, much less in its isolated gastrointestinal form.
This case describes the atypical presentation of histoplasmosis restricted to the colon, in the context of a young patient with active SLE, in which the challenge is to balance adequate control of the autoimmune disease while at the same time counteracting the fungal infection. Given its acute kidney component, recognized as an independent predictor of a poor prognosis in histoplasmosis, and the absolute necessity of decreasing the inflammatory activity - with the use of corticosteroids contraindicated - we opted to manage this patient with IV immunoglobulin, whose pleiotropic effects are increasingly recognized in terms of its mechanism of action and safety 13. There were good results in this particular case, at the same time contrasting with the general recommendation of suspending immunosuppressants for at least the first three months of antifungal treatment 2,5, which was not possible in this case due to the degree of inflammatory activity, making therapeutic decision making quite a challenge.
While the literature states that up to 30% of histoplasmosis cases may coexist with a normal chest x-ray, we highlight in this case that Histoplasma spp. was not isolated or documented in other samples or tissues other than the colon. However, considering that gastrointestinal involvement is a normal characteristic of subacute progressive disseminated histoplasmosis, the treatment of this condition is based on amphotericin B (relatively contraindicated in lupus nephritis) or itraconazole (whose initiation was delayed due to the limited availability of this drug in the initial healthcare facility) with a consolidation and maintenance phase which should last at least one year, monitoring therapeutic serum levels and frequently measuring antigenuria, which, together with symptom resolution, are indicators of an adequate clinical and microbiological response 1,2,7,9. As mentioned above, the prognosis is favorable when treatment begins promptly; limitations in health care in general must be acknowledged, especially regarding the availability of first-line medications and diagnostic studies which in this case, fortunately, did not lead to negative consequences for the clinical outcome.











 texto em
texto em 


