Introduction
Lupus anticoagulant-hypoprothrombinemia syndrome (LAHS) is a disorder characterized by an acquired coagu lation factor II, or prothrombin, deficiency, together with the presence of lupus anticoagulant 1. This is a very rare syndrome in the medical literature, with a predominant bleeding tendency ranging from epistaxis and chemosis, which are the most frequent, to cases of hematuria, gastro intestinal bleeding and intracranial bleeding, among others 2, especially if the factor II deficiency is less than 10%.
It has a high prevalence in children and females, com monly associated with viral infections and autoimmune processes 7, especially systemic lupus erythematosus. However, pharmacological and tumoral causes have been described less frequently.
Treatment for LAHS is based on immunosuppression to avoid hemorrhagic events and try to eliminate the factor II inhibitor 3.
Case presentation
This was a 19-year-old male patient who was diagnosed in July 2021 with hypoprothrombinemia due to a factor II deficiency, clinically manifesting with hematuria and nor mally medicated with methylprednisolone 40 mg, as ordered by the attending hematologist.
He was admitted to the emergency room of a private clinic in the city of Buenos Aires (Argentina) with a two-week history of petechiae and purpura, associated with left wrist, elbow and shoulder arthralgia, abdominal pain, nausea, decreased appetite, non-inflammatory diarrhea three or four times a day and a 5 kg weight loss.
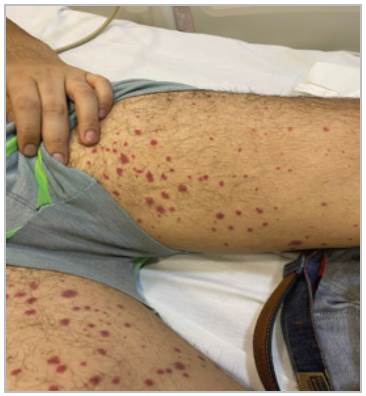
The physical exam was notable for palpable purpura on the internal thighs, calves and feet (Figure 1), a single petechia on the buccal mucosa and splenomegaly. He had no other signs and symptoms correlated with any other hematologic or rheumatic disease. Family hereditary information was unknown due to being adopted.
Admission laboratory tests reported: a complete blood count with Hgb 10.3 g/dL, leukocyte count 1.6 x 109/L with 60% neutrophils and 33% lymphocytes, and platelets 109,000 x mm3; a peripheral blood smear showing a pre served red cell line with no schistocytes, scant white blood cells with preserved shape, aggregated platelets, with no blasts; creatinine 2.42 mg/dL and blood urea nitrogen 56.9 mg/dL; PT 39% INR 2.08, PTT 67 seconds; fibrinogen 486 mg/dL; haptoglobin negative and a urinalysis showing microscopic hematuria.
Subsequent studies showed positive antinuclear antibod ies (ANA) with a homogenous 1/640 pattern, low levels of complement (C3 and C4), anti-DNA 1/320, 19% factor II, positive anti-Sm antibodies, the presence of lupus inhibitor on the KPTT (dVVR) test and a 24-hour urine protein of 1.55 g/dL.
Antiphospholipid syndrome (APS) was ruled out due to negative anticardiolipin antibodies, B2 glycoprotein and not meeting the Sidney criteria for APS; viral etiologies were also rejected. The only finding on radiological studies was homogenous splenomegaly; renal arterial and venous Doppler of the splanchnic-portal axis ruled out thrombi and structural kidney abnormalities, while serositis was ruled out by abdominal ultrasound, chest tomography and an echocardiogram.
Finally, skin and kidney biopsies were done which con firmed the presence of active diffuse lupus nephropathy associated with cutaneous purpuric vasculitis. The patient was diagnosed with SLE with kidney involvement (rapidly progressive glomerulopathy) and, in light of the history of factor II deficiency and the presence of lupus anticoagulant, he was diagnosed with associated LAHS. He was treated with cyclophosphamide and low-dose steroids, which stabi lized his anticoagulation test values (Table 1). After hospital discharge, he continued treatment with oral corticosteroids and, on a subsequent follow up, hydroxychloroquine and mycophenolate mofetil were added, which he is currently tolerating.
Table 1 Laboratory results before and after treatment with corticosteroids and cyclophosphamide.
| Methylprednisolone | Cyclophosphamide | |||||
|---|---|---|---|---|---|---|
| Admission | 1st Day | 2nd Day | 3rd Day | Cycle 1 | Cycle 2 | |
| PT % | 39 | 37 | 39 | 45 | 53 | 98 |
| INR | 2.08 | 2.07 | 1.97 | 1.78 | 1.58 | |
| KPTT (sec) | 67 | 69 | 60 | 54 | 47 | 37 |
| Urea mg/dL | 56.9 | 55.8 | 57.2 | 69 | 50.1 | 48 |
| Creatinine mg/dL | 2.42 | 2.29 | 1.94 | 1.84 | 1.55 | 1.27 |
Discussion
Lupus anticoagulant (LA) is an antiphospholipid anti body which prolongs coagulation times in vitro. This may be associated with a hypercoagulable state resulting in thromboembolic events. Bleeding manifestations are very uncommon in lupus anticoagulant syndrome and, when they occur, an associated thrombocytopenia or factor II deficiency should be suspected 4. Lupus anticoagulant-hypoprothrombinemia syndrome is a rare association that occurs in patients with SLE. In this patient, the main sign of bleeding was the presence of purpura and petechiae, as well as microhematuria, which indicated that his hematological disease was active, with a factor II level of 19%. Mulliez et al. 1 produced case reports on 72 patients with LAHS in which ecchymosis (44%) and epistaxis (35%) were high lighted as the main hemorrhagic manifestations, and, less frequently, hematuria (15%). Hemorrhagic tendencies in this type of patient have been related to the serum level of factor II; in their case report, Sarker T et al. 7 indicated that severe hemorrhagic manifestations occur when the factor II level is less than 10%, and mild when the value is less than 20%.
One of the main complications of SLE is kidney involve ment, as shown in the reported case, in which the patient had diffuse lupus nephropathy (grade IV) and received immunomodulating treatment with corticosteroid and cyclophosphamide bursts, based on what was available at the time, with which he improved. However, this is not the only immunomodulator. Carreño G et al. 5 presented a similar case in a 37-year-old male with LAHS and a history of lupus nephropathy, who debuted with a subdural hematoma as the hemorrhagic manifestation. He was treated with rituximab due to suspected severe refractory SLE, and his anticoagu lation test levels stabilized.
Conclusion
Lupus anticoagulant-hypoprothrombinemia syndrome is a rare disorder that occurs in children, with little evidence in the current literature of cases in adulthood. This type of patient can have mild to severe hemorrhagic manifestations; therefore, it is important for the clinician to recognize this disease, its association with autoimmune diseases and, especially, its signs and symptoms in patients diagnosed with SLE, in whom thromboses or hemorrhages are common. Our case, which manifested with mild hemorrhagic signs but with kidney involvement secondary to SLE, showed that early recognition of systemic diseases and immunosuppressant treatment improve the prognosis for this type of patients, and therefore could be used in similar cases.











 texto em
texto em