One-lung ventilation (OLV) is frequently used to facilitate surgical access in thoracic procedures. As many other non-physiological interventions in anesthesia, OLV is not a benign ventilation technique and therefore carries an increased risk of lung injury due to various mechanisms including a higher risk of alveolar collapse, barotrauma secondary to extrinsic forces (i.e., mediastinal shift and limited chest wall excursion) and ischemia-reperfusion phenomena during the transition of OLV to two-lung ventilation (TLV).1 Based on experimental models, the incidence of subclinical lung injury has been reported in up to 70-80% of cases, which may become clinically significant in about 10-15% of the cases if other risk factors are present (e.g., obstructive pulmonary disease, sleep apnea, smoking history, large lung resections, liberal fluid therapy). New anesthetic interventions have been postulated to minimize lung injury during thoracic surgery (e.g., individualized positive end-pressure ventilation, pharmacological preconditioning, permissive hypercapnia), yet their effectiveness is still controversial and need further evidence to support its clinical practice.2 This editorial article aims to discuss current gaps in the literature about OLV-induced lung injury and ongoing randomized trials that might elucidate new paradigms in thoracic anesthesia.
Literature gaps and controversies in one-lung ventilation
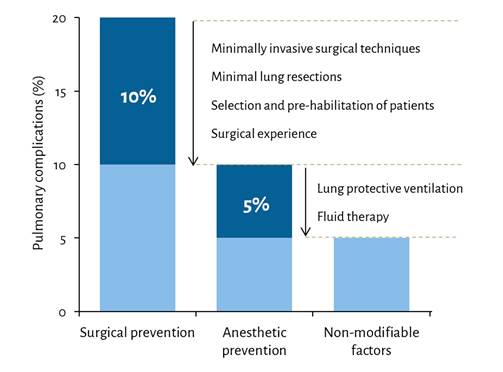
Pulmonary protection can be achieved through surgical practices or anesthetic interventions. It is thought that special surgical techniques, such as thoracoscopy assisted lung procedures or parenchymal-sparing resections, are responsible for a halfreduction in the rates of pulmonary complications over the past decade (Figure 1). Specific anesthetic interventions can further improve pulmonary outcomes if used appropriately (Table 1) - however, the current evidence on OLV has yielded several controversies as noted in the following list of interventions:
Source: Author.
Figure 1 Contribution of surgical and anesthetic interventions to reduce pulmonary complications after one-lung ventilation.
Table 1 Summary of evidence-based clinical practices for one-lung ventilation.
| Clinical practice | Recommendation | Advantages | Disadvantages |
|---|---|---|---|
| Low tidal volume | ≤ 6mL/kg | Lower risk of overinflation (↓strain) | Risk of alveolar collapse and hypoxemia if not used with recruitment maneuvers |
| Positive end-expiratory pressure | 5-10 (individualized based upon driving pressure) | Improved oxygenation and compliance | Hemodynamic compromise and venous congestion |
| Alveolar recruitment | Transition to OLV | Lower risk of alveolar collapse | Transient hypoxemia due to increased shunting |
| Inspiratory oxygen fraction | 60-80% | Less oxidative stress and radical oxygen species | Risk of hypoxemia |
| Permissive hypercapnia | EtCO2 45-50 mmHg | Decreased inflammation and improved respiratory compliance | Caution in pulmonary hypertension (right heart failure or obstructive pulmonary disease) |
| Type of anesthesia | Inhaled anesthetics | Diminished ischemia-reperfusion injury | Disturbance of hypoxic pulmonary vasoconstriction |
| Dexmedetomidine | 0.5 mcg/kg/hr | Enhancement of hypoxic pulmonary vasoconstriction | Postoperative sedation levels |
| Goal-directed fluid therapy | SVV > 13% | Less alveolar edema | Lack of clinical application for open chest procedures |
Source: Author.
Tidal volume. This is perhaps the least controversial strategy among other anesthetic interventions. A recent meta-analysis of 18 studies concluded that a low tidal volume strategy (≤ 6mL/kg) in thoracic surgery is associated with lower rates of pulmonary complications.3 Importantly, oxygenation does not seem to be affected negatively compared to higher tidal volumes.3 It is also worth noting, however, that the use of low tidal volumes in the absence of sufficient positive end-expiratory pressure (PEEP) may lead to worse pulmonary outcomes due to an increased risk of atelectrauma.4
PEEP. There is universal consensus in favor of using PEEP greater than zero, but little consensus exists regarding the optimal use of PEEP in thoracic surgery. A recent multicentric trial from Korea demonstrated that PEEP guided by driving pressure did not reduce any pulmonary complication compared to standard PEEP of 5 cmH2O in thoracic surgery.5 Additionally, there is no evidence comparing the various techniques of PEEP optimization (e.g., guided by driving pressure, inflection of the compliance curve, impedance tomography).
Ventilation mode. It has been thought that pressure-regulated modes (i.e., pressure-regulated volume guaranteed or pressure-controlled) are preferred over volume-controlled modes as the former can attenuate high airway pressures -often seen during OLV-, thereby reducing the risk of barotrauma. However, a recent multicentric trial including 1224 patients from China did not demonstrate any pulmonary benefit between ventilation modes in thoracic surgery.6
Permissive hypercapnia. A single-center trial from Korea showed superior oxygenation and better respiratory mechanics among patients exposed to permissive levels of hypercapnia (45-50 mmHg) by adjusting respiratory rates. 7 Even higher levels of hypercapnia (6070 mmHg) can be safe and effectively reduce inflammatory response from OLV. 8 Although preliminary results seem promising, one should carefully consider the exclusion of heart failure patients or severe obstructive pulmonary disease from this intervention. It is still unknown whether this intervention improves lung recovery and postoperative pulmonary outcomes.
Type of anesthesia. Although inhaled anesthetics had been theoretically avoided during OLV due to its negative effect on hypoxic pulmonary vasoconstriction, recent evidence suggests that sevoflurane reduces inflammatory mediators and is clinically non-inferior compared to total intravenous anesthesia.9,10
Dexmedetomidine. There is growing evidence on dexmedetomidine and its clinical benefits to attenuate lung inflammation and improve respiratory mechanics during OLV.11 However, the evidence is still inconclusive as to the potential utility of dexmedetomidine for preventing clinical pulmonary complications.
Evolving evidence
Several trials are being conducted to determine the impact of some of the interventions mentioned above. For instance, some of the controversies around the optimal use of PEEP in OLV are being addressed by large randomized trials, such as the PROTHOR trial (PEEP of 10 cmH2O vs 5 cmH2O)12 and iPROVE-OLV trial (Individualized PEEP guided by driving pressure vs 5 cmH2O).13 Another trial is also underway to evaluate the effect of hypercapnia (vs normocapnia) on pulmonary complications.14 Further trials are necessary to clarify the optimal tidal volume (4-6 mL/kg versus 6-8 mL/ kg) and the utility of dexmedetomidine during OLV. Perhaps in the future, more trials will be designed to assess the impact of personalized ventilatory approaches (guided by mechanical power, driving pressure, or respiratory compliance) as well as perioperative bundles including pre-habilitation, incentive spirometry, and continuous positive airway pressure.











 texto em
texto em 



