Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
International Journal of Psychological Research
Print version ISSN 2011-2084
int.j.psychol.res. vol.9 no.2 Medellín Jul./Dec. 2016
https://doi.org/10.21500/20112084.2425
DOI: http://dx.doi.org/10.21500/20112084.2425
Research
Consummatory Successive Negative Contrast in Mongolian Gerbil (Meriones unguiculatus)
Contraste Sucesivo Negativo Consumatorio en Jerbos de Mongolia (Meriones unguiculatus)
Andrés M. Pérez-Acostaa*, Gladys S. Martínezb, Karen Eliana Corredor Páezb, Cristina Vargas Irwinb and Luisa Fernanda Méndez Pardoc
a Universidad del Rosario, Bogotá, Colombia.
b Centro de Investigaciones en Biomodelos, Colombia
c Universidad INCCA de Colombia, Colombia
* Corresponding author: Andrés M. Pérez-Acosta, Programa de Psicología, Escuela de Medicina y Ciencias de la Salud, Universidad del Rosario, Sede Quinta de Mutis, carrera 24 # 63C-69, código postal: 111221, Bogotá, Colombia. E-mail address: andres.perez@urosario.edu.co.
Article history:
Received: 24-05-2016. Revised: 13-06-2016. Accepted: 21-06-2016
ABSTRACT
Successive Negative Contrast (SNC) is a phenomenon in which there is a sharp decrease in the rate of reinforced behavior immediately after a sudden reduction in quality or quantity of a habitual reinforcer. This phenomenon has become a solid experimental paradigm for the study of frustration in mammals but it has not been reported to date in the Mongolian gerbil (Meriones unguiculatus). This species is monogamous and biparental, and these differential characteristics make it interesting for comparative analysis. This work includes modifications to the SNC paradigm for this species, assessing its response to sudden changes in positive reinforcement and its differences by sex. In the first phase of the experiment, a multiple choice test was used to identify the preferred reinforcer for the species (sesame paste). After the selection of the primary incentive, the contrast procedure was carried out by presenting sesame paste in the pre-shift phase and LabDiet, Laboratory Rodent Diet, food during post-shift one. A repeated measures ANOVA of showed a significant reduction of consummatory behavior in the experimental group when compared with the control group. No differences were observed by sex. These data expand the possibility of using gerbils in the comparative study of the relationship between learning and emotion in mammals, both in behavioral and in neurobiological levels.
Keywords: successive negative contrast; positive reinforcement; food preferences; frustration; Mongolian gerbil.
RESUMEN
El Contraste Sucesivo Negativo (CSN) es un fenómeno en el cual hay una brusca disminución conductual asociada a una sorpresiva reducción sustancial en la calidad o cantidad de reforzador que se recibía habitualmente. Este fenómeno se ha constituido en un sólido paradigma experimental para el estudio de la frustración en especies mamíferas pero no ha sido aún evidenciado en la especie jerbo de Mongolia (Meriones unguiculatus). Este jerbo es monógamo y biparental, características diferenciales que lo hacen interesante para el análisis comparado. El presente trabajo incluye modificaciones al paradigma de CSN para esta especie, evaluando su respuesta a cambios sorpresivos en el reforzamiento positivo y sus diferencias por sexo. En la primera fase, se usó una prueba de elección múltiple para determinar el reforzador preferido (pasta de ajonjolí). Posterior a la selección del incentivo primario, se llevó a cabo el procedimiento de contraste presentando pasta de ajonjolí durante la fase de pre-cambio y alimento LabDiet, Laboratory Rodent Diet, durante el post-cambio. Un análisis de varianza para medidas repetidas mostró una reducción significativa en la conducta consumatoria en el grupo experimental comparado con el grupo control. No se observaron diferencias por sexo. Estos datos situarían al jerbo como apto para ampliar el estudio comparado de las relaciones entre aprendizaje y emoción en los mamíferos, tanto a nivel conductual como neurobiológico.
Palabras clave: contraste sucesivo negativo; reforzamiento positivo; preferencia de comida; frustración; jerbos de Mongolia.
1. INTRODUCTION
Successive Negative Contrast (SNC), broadly reviewed by Flaherty (1996), Papini (2008), and Justel, Ruetti & Mustaca (2010), is one of the paradoxical effects of positive reinforcement, i.e. a phenomenon that don't fit Thorndike's classic law of effect (Herrnstein, 1970; Thorndike, 1911), which predicts an association between behavior and its consequences. This rupture can be clearly perceived when individuals are exposed to a surprising devaluations in the reward quality or quantity received in response to instrumental (Crespi, 1942) or consummatory behavior (Vogel, Mikulka & Spear, 1968). In a typical SNC experiment, a group of rats receives a 32% sucrose solution in a 5-min-long sessions for 10 days (preshift) and then are presented with a 4% sucrose solution in 5-min-long sessions for 5 days (postshift). Consumption during postshift in the 32-4% sucrose solution group is significantly lower than that of the 4-4% control group (Ruetti & Justel, 2010).
SNC is relevant not only because it is an example of paradoxical reward phenomena, but also because it has become a strong experimental paradigm for comparative research on emotion, particularly on frustration (Amsel, 2006). Evidence from the initial experimental findings (Elliott, 1928; Tinklepaugh, 1928) and from more recent studies (Cuenya, Kamenetzky, Fosacheca & Mustaca, 2013; Manzo, Donaire, Sabariego, Papini & Torres, 2015; Ortega et al., 2013), seems to support the assumption that the sudden behavioral reduction is mediated by negative affect rather than by perceptual factors, such as the reward taste during postshift (reduction in sucrose palatability; Olszewski & Levine, 2007).
Some authors claim that the SNC and other phenomenon associated to a surprising omission of reward are unique to mammals and reveal an egocentric mechanism that allows the animal to process the internal changes occurring in response to the reward; this mechanism differs from mechanisms in some other vertebrates in which the almost complete absence of paradoxical phenomenon and the generalized adjustment to the law of effect suggest an allocentric mechanism by which the individual simply processes changes about the environment (Papini, 2003). SNC studies on a wide variety of species during the last eight decades confirm the presence of this mechanism in mammals, including rats (Elliott, 1928), mice (Mustaca, Bentosela, & Papini, 2000), dogs (Bentosela, Jakovcevic, Elgier, Mustaca & Papini, 2009), pigs (Dantzer, Arnone & Mormede, 1980), monkeys (Tinklepaugh, 1928), and humans (Cuenya et al., 2013). As a non-mammal exception, there is a study with a bird, the European starling (Freidin, Cuello & Kacelnik, 2009).
If the SNC and the egocentric processing of reward are unique to mammals, these mechanisms should also be present in mammal species not yet explored. An extensive literature review did not find any evidence of SNC in the Mongolian gerbil (Meriones unguiculatus: Gulotta, 1971)1, a monogamous rodent found in desert regions of China and Mongolia (Razzoli & Valsecchi, 2006), biparental (Wynne-Edwards, 2007), with some nocturnal rhythms (Flórez-Acevedo, Sarmiento & Martínez, 2010). This rodent exhibits some emotional responses, including foot tapping, an alarm signal associated to fear (Woolley, Haman, Higgins, & Ballard, 2006), and epileptic seizures in response to stress or sudden environmental changes (Hwang et al., 2005).
Gerbil's behavior (monogamy, biparental care, and negative emotions) makes these rodents closer to more complex species, including humans, and constitute a fascinating model suitable for comparison to other mammalian species. For example, understanding of underlying mechanisms that explain differences in mother/father abusive behavior can be better achieved by using biparental instead of the traditional monoparental rat model (Martínez, 2008). Particularly, some behaviors resulting from social isolation, such as crying and ultrasonic vocalizations (Mast, Fagen, Rovee-Collier & Sullivan, 1980; Muller, Moore, Myers & Shair, 2009) could be also influenced by incentive reduction.
Study of SNC in a new species, however, poses a challenge in the adaptation of the experimental paradigm in all of its parameters, including quality and quantity of reward. In an initial effort to study SNC in Mongolian gerbils Martínez, Vargas-Irwin and Pérez-Acosta (2011) used stimuli and parameters similar to those used in consummatory successive negative contrast experiments with rats: consumption of a 32% sucrose solution by Mongolian gerbils in a skinner box was recorded for 10 session and . Data from this pilot study revealed that gerbils did not consume the sucrose solution (rat's favorite), a response that may be related to the fact that this species comes from desert regions, and therefore has a low overall water consumption. In addition, gerbils in the rat's Skinner box could not maintain the appropriate position needed for consuming the liquid. Instead, animals spent most of the time chewing on the bars of the grid floor.
The study presented here includes some technical changes to the SNC paradigm to adapt it to Mongolian gerbils, in order to generate an experimental setting sensitive to surprising changes in reward. Key changes included the type of reward (sesame paste instead of sucrose solution), and the environment in which the reward omission occurred (housing cage instead of a Skinner box).
2. METHOD
2.1Subjects
Adult male (n=14) and female (n=14) experimentally naïve Mongolian Gerbils bred by the National Institute of Health (Colombia). There was no information about lineage by provider. Animals were singly housed in Tecniplast Polycarbonate Plastic Cages (41.91 x 20.32 x 20.32 cm) in the Universidad INCCA laboratory under a 12:12-h LD cycle (lights on at 06:00 h.). Animals were allowed free access to food (Purina LabDiet Laboratory Rodent Diet® 5001) and water. Temperature was maintained at 23 +/- 2 °C and humidity between 40 y 59 %. To ensure habituation to laboratory conditions, animals were single housed on arrival and left without intervention in the experimental zone for eight days.
During the entire experimental period, Gerbils were kept at an 85% of their ad libitum weight by postrial feeding, about 20 minutes after the daily experimental session. Animals were weighted daily to make sure their weight did not fall below 85%, and their health was supervised by a veterinarian. All testing sessions were performed during the light phase of the daily cycle (between 9:00 and 11:00 h.).
All experiments were performed in accordance with the standards in the NIH Guide for the Care and Use of Laboratory Animals. Measures were taken to minimize pain or discomfort of the animals used in these experiments.
2.2 Apparatus
During experimental sessions, animals were tested in cages similar to those in which they were housed but without bedding (experimental cage). Food was provided in trays placed on the bottom of each cage. Animals in both conditions had free access to water.
3. PROCEDURE
3.1 Food preference test
We used a multiple choice test to determine food preference in Mongolian gerbils. Trays with food (sesame paste, LabDiet: Laboratory Rodent Diet, peanut butter or peanut) were placed on the bottom of each cage (left corner). Each animal (n=30) was then placed on the opposite corner. Animals were left in the cage for 20 minutes and were allowed free access to food and water. Food in each tray was weighted before and after each session, and differences in the amount of food consumed before and after were registered as food consumption.
Gerbils preferred sesame paste over any other food. Based on these observations, sesame paste was used in the present experiment.
3.2 Consummatory Successive Negative Contrast
Pairs matched by sex were randomly assigned to either food pellet (LabDiet: Laboratory Rodent Diet) or sesame paste condition (n=14 per food condition). Cage position on the cage rack was randomly assigned, and researchers were blind to the experimental condition. Sex matching anticipated to the new U. S. National Institutes of Health policy to balance sex in cell and animal studies (see Clayton & Collins, 2014; U. S. National Institutes of Health, 2015).
During the first two days, all subjects received two 20-min sessions of habituation to the experimental cage, one per day. During the next 10 days each animal had access either to LabDiet Laboratory Rodent Diet or sesame paste, depending on the group, for 20 minutes. From days 11 to 15 all animals were given access to LabDiet Laboratory Rodent Diet instead of sesame paste. The amount of food consumed by each animal was recorded at the end of the 20 min period. Animals were weighted at the end of each session. Figure 1 shows the schematic diagram of the experiment.
3.3 Data Analysis
All analyses were carried out with Statistica 13 (Dell Inc., Tulsa, OK, USA) and the significance level was set at P < 0.05. Main effects were assessed with two-way ANOVAS for repeated measures with sex and group as between-subjects factors and day as a within-subjects factor. Bonferroni comparisons were used for post-hoc comparisons.
4. RESULTS
ANOVAS for repeated measures showed a significant effect of day by group interaction [F(14, 336) = 34.58; p˃.001], significant effect of group [F(1, 24) = 11.04; p ˃ .001], and day [F(14, 336) = 16.35; p ˃ .001], and no effect of sex [F(1, 24) = .67; p = .41]. While LabDiet Laboratory Rodent Diet consumption was stable over the time, sesame paste consumption decreased from day 11 (Figure 2).
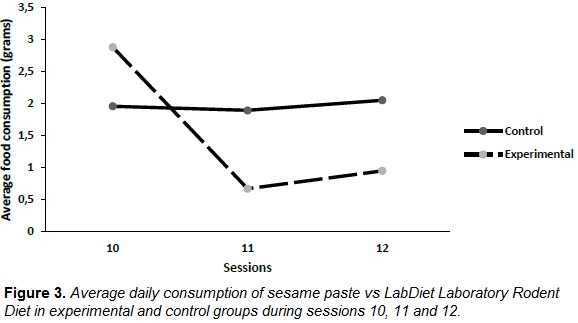
ANOVA (day by group) for repeated measures on LabDiet Laboratory Rodent Diet and sesame paste consumption on three key days, last preshift day (day 10) and two first days postshift (days 11 and 12), showed again a significant effect of day by group interaction, [F(2, 52) = 46.64; p ˃ .001], a significant effect of group [F(1, 26) = 11.13; p ˃ .001], and a significant effect of day [F(2, 52) = 46.78; p ˃ .001]. Figure 3 shows data for consumption during these three days. Total consumption during day 11 was significantly lower with sesame paste compared to LabDiet Laboratory Rodent Diet consumption. Pairwise multiple comparison (Bonferroni test) showed a significant lower consumption in sesame paste group on day 10 compared to days 11 (p ˃ .001) and 12 (p ˃ .001), and compared to LabDiet Laboratory Rodent Diet group (p ˃ .001). There were no significant differences along these three days in LabDiet Laboratory Rodent Diet group.
5. DISCUSSION
First adaptation of successive negative contrast paradigm on Mongolian gerbils failed because it used the same context (Skinner's box) and positive rewards (32% sucrose solution used with rats (Martínez, Vargas-Irwin & Pérez-Acosta, 2011). In the experiment reported here, we show for the first time a SNC effects in gerbils, by adjusting the context (housing cage) and the reward (sesame paste), a reward selected based on data obtained from a food preference test.
Main strength of consummatory successive negative contrast original paradigm in rats is the possibility of consistent replicas of significant reduction in the consumption of a devaluated reinforcer such as the high calorie 32% sucrose solution (Ruetti & Justel, 2010); a reinforcer that is also highly functional in mice (Mustaca et al., 2000). In this study there was also a significant reduction compared to a control group that received as reinforcer the same food given in the house cage. The reward preferred (given to the experimental group) was different in quality from the usual food pellets: sesame paste, also a high calorie food not because of the sugar component but because the high content of fatty acids2.
The prevailing hypothesis on successive negative contrast is a hypothesis emotional in nature: frustration (Amsel, 2006; Manzo et al., 2015; Norris, Pérez-Acosta, Ortega & Papini, 2009; Ruetti & Justel, 2010). The alternative proposal opposing the frustration hypothesis refers instead to the decreased palatability of the sucrose (Olszewski & Levine, 2007). Data presented in this article are in agreement with the frustration hypothesis instead of the hypothesis on decreased palatability: 57% of the calories in the reinforcer of the control group (LabDiet Laboratory Rodent Diet) come from carbohydrate and only 13% come from fat3. In other words, gerbils in the experimental group changed from a reinforcer perceived as “less sweet” (sesame paste) to another perceived as “sweeter” (LabDiet Laboratory Rodent Diet). Despite this, they exhibited the negative contrast phenomenon.
Contrast observed was consistent with data obtained in the food preference test, in which gerbils chose sesame paste instead of LabDiet Laboratory Rodent Diet or the other food with high lipid content (peanut butter and peanut). Sesame paste is therefore an optimal appetitive reinforcer for this species, similar to the 32% sucrose solution used in SNC studies with rats and mice. However, SNC paradigm requires for the control group an appetitive reinforcer that is different in quantity and not in quality with that used with the experimental group (i.e., sesame paste with less fatty acids and more carbohydrate).
Low water consumption and null preference for liquid reinforces high or low sugar in gerbils, (Martínez, Vargas-Irwin & Pérez-Acosta, 2011), together with the high and consistent preference for a pasty reinforcer high in lipids, such as sesame paste, may be related to gerbil's original desert habitat (Gulotta, 1971), which is by definition low in water. The finding about gerbil´s preference for this type of food is by useful for studies with this species in which a positive reinforcer is required.
Gerbil's emotional behavior in this study is, however, comparable to behavior of mammals in previous studies on SNC paradigm (Figures 2 and 3). Although studies that replicate these findings are still needed, results in both groups (during preshift and postshift) are similar to those observed in other species. It is even striking the poor emotional habituation in the experimental group, which compared to data obtained in rats, continue to exhibit significant differences even after five postshift sessions (Figure 2). This apparent “increased emotionality” compared to rats is coherent with gerbil´s special sensitivity to stressful events or sudden changes in the environment, that easily may lead to epileptic seizures (Hwang et al., 2005). This emotionality pattern can be confirmed in studies looking for changes in the social and sexual response, or repetitive behaviors that may be present during preshift/postshift.
Figure 3 supports not only differences in gerbil's emotional response to the surprising omission of reward, but also draws attention on the need for an exploration on its neurobiological basis: in both rats and mice (see Mustaca et al., 2000) there are differences in behavior during session 10 (last preshift session), 11 (first postshift session), and 12 (second postshift session). These differences seem to be related to the differential action of nuclei in the brain opioid subsystem (reviewed in Papini, Wood, Daniel & Norris, 2006). Instead, in gerbils we did not find differences in behavior during session 11 and 12, a results that occurs in rats only when opioid antagonists are applied (see Norris et al., 2009).
Future exploration of behavior and neurobiology of gerbil's consummatory successive negative contrast will contribute to a finer comparison of the interaction between learning and emotion in rodents, mammas, and vertebrates. In this sense, it is necessary to overcome limitations of this study by conducting experiments to further evaluate the de SNC phenomenon in gerbils, increasing the sample and the number of postshift sessions, controlling genetic variables, and using reinforcers that differ in quantity instead of quality. In addition, gerbil's behavioral characteristics such as biparentality and monogamy, may be key for new animal models on negative emotions resulting from loss of conspecifics instead of loss of purely physical reinforcers.
Footnotes
1 Taxonomy for this species is described in the Wilson and Reeder's database of mammalian taxonomy, (third edition), available in: http://www.departments.bucknell.edu/biology/resources/msw3/browse.asp?id=13001155.
2 Nearly half of the nutrient composition in sesame seed is lipids (fat); less than 1% is sugar. As reported by the United States Department of Agriculture (USDA): https://ndb.nal.usda.gov/ndb/foods/show/3620?fgcd=&manu=&lfacet=&format=Full&count=&max=35&offset=&sort=&qlookup=12023.
3 Nutrient composition for LabDiet Laboratory Rodent Diet 5001 can be found on: http://www.labdiet.com/cs/groups/lolweb/@labdiet/documents/web_content/mdrf/mdi4/~edisp/ducm04_028021.pdf.
6. REFERENCES
Amsel, A. (2006). Frustration Theory: An Analysis of Dispositional Learning and Memory. Cambridge, U.K.: Cambridge University Press. [ Links ]
Bentosela, M., Jakovcevic, A., Elgier, A. M., Mustaca, A. E., & Papini, M. R. (2009). Incentive contrast in domestic dogs (Canis familiaris). Journal of Comparative Psychology, 123(2), 125-130. doi: http://dx.doi.org/10.1037/a0013340. [ Links ]
Clayton, J. A. & Collins, F. S. (2014). Policy: NIH to balance sex in cell and animal studies. Nature, 509, 282-283. doi: 10.1038/509282a. [ Links ]
Crespi, L. P. (1942). Quantitative Variation of Incentive and Performance in the White Rat. The American Journal of Psychology, 55(4), 467-517. doi: http://dx.doi.org/10.2307/1417120. [ Links ]
Cuenya, L., Kamenetzky, G. V., Fosacheca, S., & Mustaca, A. E. (2013). Successive negative contrast effect on the emotional evaluation of images. Anales de Psicología, 29(3), 944-952. doi: http://dx.doi.org/10.6018/analesps.29.3.150861. [ Links ]
Dantzer, R., Arnone, M., & Mormede, P. (1980). Effects of frustration on behaviour and plasma corticosteroid levels in pigs. Physiology & Behavior, 24(1), 1-4. doi: http://dx.doi.org/10.1016/0031-9384(80)90005-0. [ Links ]
Elliott, M. H. (1928). The Effect of Change of Reward on the Maze Performance of Rats (Vol. 4). Berkeley: University of California Press. [ Links ]
Flaherty, C. F. (1996). Incentive relativity. New York: Cambridge University Press. [ Links ]
Flórez-Acevedo, S., Sarmiento, N., & Martínez, G. S. (2010). Patrones de alimentación, sueño y actividad reproductiva en jerbos de Mongolia. Suma Psicológica, 17(2), 201-208. [ Links ]
Freidin, E., Cuello, M. I., & Kacelnik, A. (2009). Successive negative contrast in a bird: starlings' behaviour after unpredictable negative changes in food quality. Animal Behaviour, 77(4), 857-865. doi: http://dx.doi.org/10.1016/j.anbehav.2008.12.010. [ Links ]
Gulotta, E. F. (1971). Meriones unguiculatus. Mammalian Species (3), 1-5. doi: http://dx.doi.org/10.2307/3503988. [ Links ]
Herrnstein, R. J. (1970). On the law of effect. Journal of the Experimental Analysis of Behavior, 13(2), 243-266. doi: http://dx.doi.org/10.1901%2Fjeab.1970.13-243. [ Links ]
Hwang, I. K., Lee, Y.-B., Yoo, K.-Y., Kang, T.-C., Kim, D.-W., Moon, W.-K., Won, M. H. (2005). Seizure-induced changes of mineralocorticoid and glucocorticoid receptors in the hippocampus in seizure sensitive gerbils. Neuroscience Research, 53(1), 14-24. doi: http://dx.doi.org/10.1016/j.neures.2005.05.006. [ Links ]
Justel, N. R., Ruetti, E. M., & Mustaca, A. (2010). Aproximaciones teóricas al estudio del contraste sucesivo negativo consumatorio. Suma Psicológica, 17(2), 111-123. [ Links ]
Manzo, L., Donaire, R., Sabariego, M., Papini, M. R., & Torres, C. (2015). Anti-anxiety self-medication in rats: oral consumption of chlordiazepoxide and ethanol after reward devaluation. Behavioural Brain Research, 278, 90-97. doi: http://dx.doi.org/10.1016/j.bbr.2014.09.017. [ Links ]
Martínez, G. S. (2008). El maltrato infantil: mecanismos subyacentes. Avances en Psicología Latinoamericana, 26(2), 171-179. [ Links ]
Martínez, G. S., Vargas-Irwin, C., & Pérez-Acosta, A. M. (2011). Evalución del efecto conductual de la omisión sorpresiva de reforzamiento en jerbos. International Journal of Psychological Research, 4(Número especial: Memorias del XXXIII Congreso Interamericano de Psicología, Medellín), 1621. [ Links ]
Mast, V. K., Fagen, J. W., Rovee-Collier, C. K., & Sullivan, M. W. (1980). Immediate and long-term memory for reinforcement context: the development of learned expectancies in early infancy. Child Development, 51(3), 700-707. doi: http://dx.doi.org/10.2307/1129455. [ Links ]
Muller, J. M., Moore, H., Myers, M. M., & Shair, H. N. (2009). Dopamine's role in social modulation of infant isolation-induced vocalization: II. Maternally modulated infant separation responses are regulated by D1- and D2-family dopamine receptors. Developmental Psychobiology, 51(2), 158-172. doi: http://dx.doi.org/10.1002%2Fdev.20355. [ Links ]
Mustaca, A., Bentosela, M., & Papini, M. R. (2000). Consummatory Successive Negative Contrast in Mice. Learning and Motivation, 31, 272-282. doi: http://dx.doi.org/10.1006/lmot.2000.1055. [ Links ]
Norris, J. N., Pérez-Acosta, A. M., Ortega, L. A., & Papini, M. R. (2009). Naloxone facilitates appetitive extinction and eliminates escape from frustration. Pharmacology, Biochemistry, & Behavior, 94(1), 81-87. doi: http://dx.doi.org/10.1016/j.pbb.2009.07.012. [ Links ]
Olszewski, P. K., & Levine, A. S. (2007). Central opioids and sweet tastants: When reward outweighs homeostasis. Physiology and Behavior, 91(5), 506-512. doi: http://dx.doi.org/10.1016/j.physbeh.2007.01.011. [ Links ]
Ortega, L. A., Prado-Rivera, M. A., Cárdenas-Poveda, D. C., McLinden, K. A., Glueck, A. C., Gutiérrez, G., Papini, M. R. (2013). Tests of the aversive summation hypothesis in rats: Effects of restraint stress on consummatory successive negative contrast and extinction in the Barnes maze. Learning and Motivation, 44(3), 159-173. doi: http://dx.doi.org/10.1016/j.lmot.2013.02.001. [ Links ]
Papini, M. R. (2003). Comparative psychology of surprising nonreward. Brain, Behavior & Evolution, 62(2), 83-95. doi: http://dx.doi.org/10.1159/000072439. [ Links ]
Papini, M. R. (2008). Comparative psychology. Evolution and development of behavior (2nd ed.). New York: Psychology Press. [ Links ]
Papini, M. R., Wood, M., Daniel, A. M., & Norris, J. N. (2006). Reward loss as psychological pain. International Journal of Psychology & Psychological Therapy, 6(2), 189-213. [ Links ]
Razzoli, M., & Valsecchi, P. (2006). Different social bonds produce differential effects on behaviour and physiology in Mongolian gerbils. Ethology Ecology & Evolution, 18(4), 289-306. doi: doi/abs/10.1080/08927014.2006.9522697 [ Links ]
Ruetti, E. M., & Justel, N. R. (2010). Bases neurobiológicas de la frustración. Revista Argentina de Ciencias del Comportamiento, 2(3), 45-60. [ Links ]
Thorndike, E. (1911). Animal intelligence: Experimental studies. New York: The Macmillan Company. [ Links ]
Tinklepaugh, O. L. (1928). An experimental study of representative factors in monkeys. Journal of Comparative Psychology, 8(3), 197-236. [ Links ]
U. S. National Institutes of Health (2015, june 9). Consideration of sex as a biological variable in NIH-funded research. NIH Notice Number NOT-OD-15-102, retrieved 21 june 2016 from: http://grants.nih.gov/grants/guide/notice-files/NOT-OD-15-102.html. [ Links ]
Vogel, J. R., Mikulka, P. J., & Spear, N. E. (1968). Effects of shifts in sucrose and saccharine concentrations on licking behavior in the rat. Journal of Comparative and Physiological Psychology, 66(3), 661-666. doi: http://dx.doi.org/10.1037/h0026556. [ Links ]
Woolley, M. L., Haman, M., Higgins, G. A., & Ballard, T. M. (2006). Investigating the effect of bilateral amygdala lesions on fear conditioning and social interaction in the male Mongolian gerbil. Brain Research, 1078(1), 151-158. doi: http://dx.doi.org/10.1016/j.brainres.2006.01.071. [ Links ]
Wynne-Edwards, K. E. (2007). Paternal care in rodents: Weakening support for hormonal regulation of the transition to behavioral fatherhood in rodent animal models of biparental care. Hormones & Behavior, 52(1), 114-121. doi: http://dx.doi.org/10.1016/j.yhbeh.2007.03.018. [ Links ]