INTRODUCTION
Meiofauna is a small heterotrophic organism (42 µm to 500 µm (Mare, 1942; Fenchel, 1978; Robertson et al., 2000a)) that is one of the crucial ecological groups in streams. Researchers have used more than one definition for meiofauna, for this work we are following the meiofauna definition by Fenchel (1978) and Robertson et al. (2000a) which includes protists (amoebas [naked and testate] and ciliates) and metazoans. They contribute to stream biodiversity, secondary productivity, and food web structure (Robertson et al., 2000b; Schmid-Araya et al., 2002; Stead et al., 2005; Schratzberger and Ingels, 2018; Wang et al., 2020). Meiofaunal species are easily dispersed and often diverse in stream ecosystems (Rundle et al., 2002). At least 50 % to 80 % of the stream invertebrate diversity is attributed to meiofauna (Robertson et al., 2000b). There are 13 major meiofaunal taxonomic groups that can be found within freshwater ecosystems (Robertson et al., 2000b). Nematodes, rotifers, and phagotrophic protists are among the dominant groups of meiofauna in freshwater ecosystems (Majdi et al., 2020). These organisms have fast turnover rates (Rundle et al., 2002; Majdi et al., 2020), representing a significant component of stream secondary production. Approximately 50 % of the secondary production of an acidic stream in England was due to meiofauna (Stead et al., 2005). Therefore, it is not surprising that meiofauna increases food web complexity in streams (Schmid-Araya et al., 2002). Furthermore, even though meiofauna is small body-size organisms, they can occupy various trophic levels within the food web, as demonstrated by isotope analyses (Majdi and Traunspurger, 2017).
Being small organisms and often associated with fine particles, environmental variables often play an important role in structuring their assemblages. Abiotic factors are known to be strongly related to lotic meiofaunal abundance and composition (Whitman and Clark, 1984; Swan and Palmer, 2000; Radwell and Brown, 2008; Gaudes et al., 2010; Araújo et al., 2017; Majdi and Traunspurger, 2017). For example, fluctuations in discharge have been suggested as the main abiotic factor structuring lotic meiofaunal communities (Swan and Palmer, 2000; Winemiller et al., 2014). High water velocities can dislodge organisms, thus redistributing them along the stream (Richardson, 1992; Swan and Palmer, 2000; Silver et al., 2002; Gaudes et al., 2010; Araújo et al., 2017). However, intermediate velocities promote the deposition of organisms and organic matter and prevent the accumulation of small-sized particles, such as silt and clay (Whitman and Clark, 1984), enhancing biodiversity and preventing sediment compaction. In Mill Creek in Texas, the highest abundance of meiofauna was found in places with velocities at the sediment surface of around 30 cm/s (Whitman and Clark, 1984). However, discharge can interact with other environmental variables in streams, resulting in complex interactions with meiofauna. Sediment composition affects meiofaunal distribution (Silver et al., 2002; Radwell and Brown, 2008). The type of substratum (based on particle size) is a predictor of the meiofaunal community and abundance in temperate streams (Radwell and Brown, 2008). Streambeds dominated by small particles limit meiofauna's ability to survive (Hakenkamp and Palmer, 2000). Species adapted to low oxygen conditions thrive in streambeds dominated by small particles (Hakenkamp and Palmer 2000). Thus, streambed particles could play a role in regulating meiofaunal community composition in streams.
Biotic controls are also important in determining meiofaunal composition (Schmid-Araya and Schmid, 2000; Swan and Palmer, 2000; Silver et al., 2002; Weber and Traunspurger, 2016). However, these interactions might be weaker than the effects generated by abiotic controls (Silver et al., 2002). Meiofauna is prey for many species (macroinvertebrates, fishes) (Schmid-Araya and Schmid, 2000; Weber and Traunspurger, 2016). Intestinal dissections of the red cherry shrimp (Neocaridina davidi Bouvier, 1904) in laboratory cultures showed that meiofauna is commonly ingested (Weber and Traunspurger, 2016). Top predators can affect meiofauna indirectly by controlling intermediate predators that consume meiofauna. Fishes in the Eel River in northern California (Oncorhynchus mykiss Walbaum, 1792, Hesperoleucus symmetricus S. F. Baird and Girard, 1854, and Gasterosteus aculeatus Linnaeus, 1758) feed on predatory aquatic insects, thus reducing their population. As a consequence, small macroinvertebrates (e.g., chironomids) are more abundant when fishes are present (Power, 1990). Macroinvertebrates interact indirectly with meiofauna by fragmenting organic matter (Anderson and Sedell, 1979; Wallace and Webster, 1996), thus making small particles available to meiofauna. Some meiofaunal groups feed on fine organic matter and dissolved organic matter (Schmid-Araya and Schmid, 2000). Macroinvertebrates could influence meiofaunal abundance and distribution by facilitating the production of fine organic matter. As headwater streams often lack predatory fishes (Richardson, 2019), but have abundant macroinvertebrates (Ramírez and Hernández-Cruz, 2004), the relationships between macroinvertebrates and meiofauna should be explored.
Most of our understanding of freshwater meiofauna comes from research in temperate streams (Majdi et al., 2020). Most tropical freshwater meiofauna knowledge is based on species inventories and taxonomy as few publications had dealt with the ecological aspects of this community in a tropical setting (Araújo et al., 2017; Garraffoni et al., 2017). Here, we provide new information on tropical stream meiofauna by addressing three objectives: (1) to describe the meiofaunal community of a tropical headwater stream, (2) to determine which environmental variables are related to meiofauna, and (3) to assess whether these community patterns are similar to those described for temperate zone ecosystems. We hypothesize that nematodes, testate amoebae, and ciliates (protists) will dominate the community. In addition, we expect that fluctuations in discharge will be strongly related to meiofauna richness, abundance, and composition; and that temporal variation of meiofauna richness, abundance, and composition will reflect the lack of seasonality in the tropics, for a main difference with temperate zones.
STUDY SITE
The study stream was Quebrada Prieta (18°19' N, 65°48' W), part of the Espíritu Santo watershed in the Luquillo Experimental Forest, part of El Yunque National Forest, Puerto Rico. The study reaches approximately 400 m above sea level. Quebrada Prieta has two main tributaries: Quebrada Prieta A and Quebrada Prieta B hereafter). We selected a 100 m section at each tributary for the study and six pools within each reach for sampling. The geomorphology is typical of other streams in the zone. Quebrada Prieta has low sinuosity and a steep gradient, with the channel boulder dominated and a series of pools and steps or jumps connecting them (Ahmad et al., 1993). The climate is characterized by a relatively stable mean monthly air temperature, ranging from 21 to 26°C with an annual mean of 23°C, and a mean monthly rainfall between 200 to 400 mm (Gutiérrez-Fonseca et al., 2020). This forest is aseasonal, though the months between September and December could be wetter (Gutiérrez-Fonseca et al., 2020).
MEIOFAUNAL COMMUNITY
Our twelve study pools were sampled monthly for eight months (January to August) in 2017 when we stopped sampling due to two intense hurricanes (Hurricane Irma and Maria) impacting the study site in September 2017. Our study pools were small, with an area usually smaller than 2 m2 and sand accumulations of 1 to 2 cm in depth. Since standard meiofaunal sampling techniques are designed for habitats with deeper sediment layers (Hauer and Lamberti, 2006), we designed a sampling methodology adapted to our study site. Our sampler consisted of a small polyvinyl chloride (PVC) ring (0.0062 m2) and a pipet (with an aperture of 5 mm) to remove sediments within the ring. The sampler was randomly inserted (1.5 cm depth) in each study pool, selecting areas with sediments. A sample consisted of 290 mL of sediment and water collected from the sampler plus 10 mL of 50 % cold glutaraldehyde, resulting in 300 mL of sample fixed in 2 % glutaraldehyde. Samples were stored cold until processed (Xu et al., 2010).
Species richness and abundance were calculated for each sample. Of the 300 mL fixed sample, one homogenized subsample of 10 mL was separated by density gradient. We extracted organisms using the density gradient method followed by Xu et al. (2010), which consists of using Ludox®. This colloidal silica solution generates a density gradient after centrifugation at 4,300 g. This method is expected to extract up to 99 % of the microorganisms present in sediments (Xu et al. 2010). Since samples were from freshwater, the desalination steps in Xu et al. (2010) were omitted. We separated the organisms with a 25 mm diameter nitrocellulose gridded filter (1.2 µm pore size) to perform a quantitative Protargol staining (QPS) following the modifications made by Skibbe (1994). QPS staining is a standard technique used in meiofaunal research to help identify organisms at a broader range of taxonomic levels (e.g., species, genus, and family) (Reiss and Schmid-Araya, 2008; Xu et al., 2010). Meiofauna was identified to the lowest taxonomic level (species level for testate amoebae and ciliates, morphotype for nematodes, and genus and family level for all other taxonomic groups). Some meiofaunal groups like nematodes, ciliates, and testate amoebas were observed under a total magnification of 1000X. In the case of nematodes, morphospecies were classified by oral structures into major feeding groups based on Pennak (1989). For abundances and richness estimates, filters were thoroughly screened, and all organisms between 42 and 500 µm were counted and measured (AmscopeTM Microscope Camera SKU: MU1000).
ENVIRONMENTAL VARIABLES
Sample volume not used for meiofauna richness and abundance was sieved into the following grain particle classes: coarse sand, medium sand, and fine sand. We choose these grain particle classes because sand has been reported as a present particle in our study site (Ahmad et al., 1993). After sieving, samples were dried at 70° C for two days, then re-weighted and ashed in a muffle furnace at 500°C for 1 hour to calculate the ash-free dry mass (g) (AFDM). Sediment data is presented by size class (coarse, medium, and fine sand according to the Wentworth scale) as g-m-2. We summed the ashed classes to calculate the total percentage of sediment from the sample and named this variable sediment percentage.
The stream water level is measured continuously in each tributary of Quebrada Prieta using the logger HOBO U20L-04. This data logger measures the water level using barometric pressure every 15 minutes. Stream discharge (m3s-1) was estimated using a known relationship between water level and stream discharge for each reach. Daily data were averaged 30 d before the meiofauna sampling date for our statistical analysis.
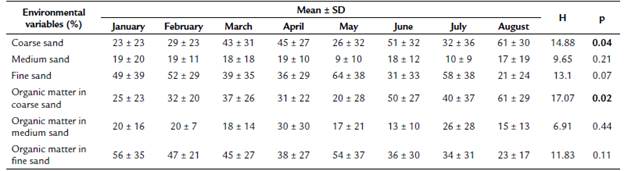
Table 1 Monthly mean ± S.D. of environmental variables measured in Quebrada Prieta, Luquillo Experimental Forest, Puerto Rico. Significant differences, according to the Kruskal Wallis test, are highlighted in bold.
The study site is part of a long-term research location (LUQ-LTER); thus, additional data were available to relate to meiofaunal communities. Data sets include water chemistry, shrimp density, macroinvertebrate assemblages, chlorophyll, and coarse organic matter input and flow. Water samples were collected weekly and analyzed. Shrimps, macroinvertebrates, and algal biomass were collected monthly at the same study pools. Shrimps were sampled with traps left in the pool overnight. Macroinvertebrates were collected using core samplers (0.0314 m2). The collected material was fixed with formaldehyde (37 %). Separation of organisms from sediment and organic matter was done in the laboratory. Collected macroinvertebrates were placed in vials with 80 % ethanol and later identified at the family level. Algal biomass was estimated monthly as mean chlorophyll concentrations (as chlorophyll-α [Chl α]) from Loeb samplers. In each pool, seven Loeb samples were collected from rock surfaces. Additional details on the methods can be found in Gutiérrez-Fonseca et al. (2020) and the LUQ-LTER database portal (portal.edirepository. org/nis/advancedSearch.jsp) searching for the following identifiers: water chemistry (knb-lter-luq.20.4923058), shrimp density (knb-lter-luq.222.2), macroinvertebrate assemblages (knb-lter-luq.227.1), chlorophyll (knb-lter-luq.136.313356), organic matter input and flow (knb-lter-luq.223.1). The meiofaunal samples were collected one week after the macroinvertebrate sampling.
STATISTICAL ANALYSES
We contrasted monthly patterns for a better understanding of factors controlling meiofauna in Quebrada Prieta. We estimated the richness of Quebrada Prieta with the estimators chao1 and ACE (Abundance-based coverage estimator). These two estimators are robust for abundance data (Chao et al., 2016; Chao and Chiu, 2016). We compared monthly richness and abundance with a Kruskal Wallis test due to a lack of normality according to a Shapiro-Wilks test (richness: W=0.83 p=0.01; abundance: W=0.76, p=0.01).
Monthly comparisons in sediment percentages were analyzed with a Kruskal-Wallis test due to a lack of normality according to a Shapiro-Wilks test (p= 0.0001 for all sediment percentages). A Principal Component Analysis (PCA) was performed to determine which environmental variables could explain variations in the meiofaunal richness and abundance. The PCA was made with ten environmental variables selected based on literature and monthly differences (Robertson et al., 2000b; Silver et al., 2002). The variables were: Discharge, Nitrate, Phosphate, Silica, Chlorophyll, Coarse Sand, Fine Sand, Organic Matter in Coarse Sand, Organic Matter in Fine Sand, and Percentage of Sediment (the percentage of the sample that was inorganic sediment). Due to differences in the sampling scales, the PCA matrix consisted of monthly averages of the environmental data. These variables were also standardized to make proper comparisons.
Generalized linear models (GLM) were constructed to determine the relationship between environmental variables and the meiofaunal richness and abundance. Additionally, we made models for the abundance of the two dominant taxa groups (Rhizaria and Nematoda) and dominant functional groups (bacterivores and detritivores). Model selection was based on the corrected Akaike's Information Criteria (AICc), which is based on the likelihood of such models corrected for a small sample size. We ended up averaging models due to the possibility of good fits from more than one model (based on delta AICc). Full average models for richness, abundance, the abundance of Nematoda, and the abundance of detritivores were constructed due to individual models' low weight. This way, we selected the most parsimonious model that fits our data (Burnham et al., 2011). All data were revised for the assumptions of the tests.
Multiple comparisons of the community structure after the PERMANOVA test were done in PAST (Hammer et al., 2012). All other tests were analyzed using R (R Core Team, 2018) with the packages SpadeR (Chao et al., 2016) for richness estimates, Vegan for PERMANOVA tests, MASS for quantitative models, and MuMIn and AICcmodavg for model comparisons and model averages, respectively.
MEIOFAUNA CHARACTERIZATION
Most major meiofaunal groups were present in Quebrada Prieta, with 11 out of 13 lotic phyla found during the study (see https://doi.org/10.5281/zenodo.7301955 for the full list of morphospecies). The sampling sites reached asymptotic richness with a total of 62 morphospecies. This richness constituted 80.9 % according to chao1 estimates and 80.8 % according to ACE estimates. The assemblage was composed of testate amoebae (37.45 %, [Rhizaria with 27.9 % and Amoebozoa with 9.55 %]), nematodes (19.1 %), flatworms (15.53 %), ciliates (8.24 %), arthropods (5.81 %), gastrotrichs (5.62 %), oligochaetes (5.43 %), rotifers (2.62 %), and tardigrades (0.19 %). There were no significant monthly differences in richness (H = 10.01, p=0.17) (Fig 1C).
The confidence intervals (95 %) of meiofauna abundance per m2 ranged from 8,913 ind.-m-2 (3,483-14,405) in May to 59,648 ind.-m-2 (31,492-92,228) in February. Monthly differences in abundance were not detected (H=10.06, p=0.18). February had the highest mean abundance with 59,274 ± 58,870 ind.-m-2, although with high variability (Fig 1A and 1B). A PERMANOVA test shows that the community structure did not vary monthly (F816=1.4359, p=0.06).
ENVIRONMENTAL VARIABLES
Percentage particle size significantly varied monthly (Table 1), with June and August having the highest percentages of coarse sand. The month with the lowest amount was January. Medium and fine sand percentages were not significantly different by month. Monthly variations were detected for organic matter in coarse sand, with August and June having the highest percentages. In contrast, the smallest percentages of organic matter in coarse sand were reported for May (Table 1).
Monthly variations in discharge in Quebrada Prieta were large (Fig 1A and Fig 1B). January and February were the two months with the lowest discharge. Increases in discharge occurred from March to August. July had the highest discharge, while February had the lowest.
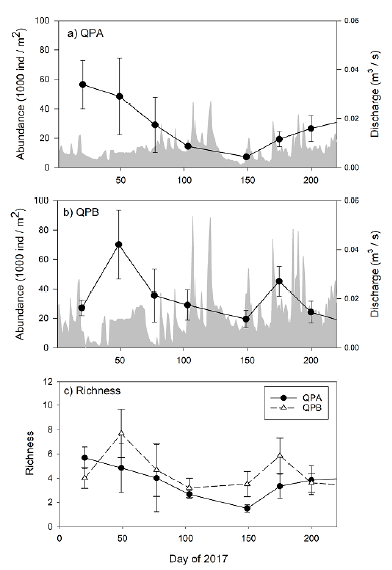
Figure 1 Monthly mean abundance (1000 ind.-m-2) ± SE of meiofauna and mean daily discharge (m3s-1) between January 2017 to August 2017 in Quebrada Prieta, Luquillo Experimental Forest, Puerto Rico. a) Quebrada Prieta A. b) Quebrada Prieta B. c) Monthly mean richness ± SE of meiofauna in Quebrada Prieta, dark dots represent Quebrada Prieta A and empty triangles represent Quebrada Prieta B
PCA shows a gradient for substrate-related variables and stream discharge-related variables (Fig 2). The PCA axis 1 explained 41 % of the variability while axis 2 explained 18 % of the variability. The gradient along axis 1 was caused by fine sand (%), organic matter in fine sand (%), and organic matter in coarse sand (%) and discharge. Two groups were formed along this axis: a group with a high percentage of coarse sand and organic matter in coarse sand and a second group characterized by months of high discharge. Along axis 2, environmental data dispersion was caused by other variables: phosphate, silica, chlorophyll, and sediment percentage. Two groups were also formed along this axis: the first one was characterized by high phosphate and silica, while the other group had high chlorophyll and sediment percentage.
RELATIONSHIP BETWEEN MEIOFAUNA AND ENVIRONMENTAL VARIABLES
The quantitative models with the lowest AIC explaining variations in meiofauna abundance (ind.-m-2) and richness (taxa/10 mL) in Quebrada Prieta were generalized linear models (Table 2). The full average model for the abundance of meiofauna shows that discharge had a negative effect and macroinvertebrates, and the percentage of coarse sand had a positive effect. Similarly, for richness, the average model of the models with delta AICc lower than two (Table 2) included the percentage of coarse sand and macroinvertebrates (positive effect) and discharge (negative effect). The model for the abundance of Rhizaria and Nematoda included only the percentage of coarse sand with a positive effect. For the abundance of bacterivores, the model average included only the percentage of coarse sand and nitrate with a positive effect and discharge with a negative effect. In contrast, the model for detritivores abundances was a full average model that included the percentage of coarse sand, silica, and the percentage of organic matter with positive effects and discharge and nitrate as negative effects (Table 2).
DISCUSSION
As we hypothesized, nematodes and protists dominated the community of meiofauna in Quebrada Prieta. We found that fluctuations in discharge were strongly related to meiofauna richness, abundance, and composition. We did not find any evidence of temporal variation in meiofauna richness, abundance, and composition.
MEIOFAUNA OF TROPICAL HEADWATER STREAMS
The richness of Quebrada Prieta seems lower than reports from temperate streams. In Lone Oak, stream richness was 142 morphospecies while Pant stream had 257 morphospecies (Reiss and Schmid-Araya, 2008). In Quebrada Prieta, we found 62 morphospecies. However, our methodology could have a bias towards specific groups, like Rhizaria, Amoebozoa, and Ciliophora since the Protargol staining was developed to identify larger protists (Skibbe, 1994), thus soft body meiofauna could be underrepresented (Garraffoni et al., 2017). Nevertheless, this staining has been used in meiofaunal studies focused on multiple taxonomic groups (Xu et al., 2010). However, the taxonomic details of other meiofaunal groups such as nematodes, rotifers, and flatworms, require different techniques in staining, microdissection, fixation, and microscopy. Still, our richness estimates are reasonable, as shown by the richness estimators.
The composition of meiofauna in our tropical study stream is similar to that reported for temperate streams. From the 13 major meiofaunal groups that could be found in freshwater environments (Robertson et al., 2000a), 11 groups were found in Quebrada Prieta. Dominant species in Quebrada Prieta had been reported as dominant in temperate streams, including nematodes and ciliates. In contrast, testate amoebae, which were abundant in Quebrada Prieta, are rarely reported in meiofaunal studies in temperate streams (Testate amoebae were not reported in these studies: Reiss and Schmid-Araya, 2008; Gaudes et al., 2010; Weber and Traunspurger, 2016). On the other hand, copepods were rare in Quebrada Prieta, especially compared to temperate streams in which these organisms are often abundant and species-rich (Strayer et al., 1997). In our study stream, there were no temporal patterns in the community structure, which is concomitant with aseasonality in the study site.
Similar to the richness, meiofauna abundances in Quebrada Prieta were lower than reports from temperate streams and continental tropical streams. This could be attributed to differences in sampling methodology, as we used a modified technique adjusted to our study site. However, we believe that methodology is not the main reason for lower abundances, since our study site is known to have generally low abundances of non-decapod macroinvertebrates (Ramírez and Hernández-Cruz, 2004). Peak abundances of meiofauna reached 227,500 and 743,400 individuals per m2 in Lone Oak and Pant streams in England (Reiss and Schmid-Araya, 2008). Meiofaunal abundances in continental tropical streams in Brazil have been estimated between 645,621and 1,329,938 per m2 (Araújo et al., 2017; Garraffoni et al., 2017). In Quebrada Prieta, the highest monthly mean abundance was reported in February, and it was 59,275 ± 58,870 ind per m2. Quebrada Prieta has lower abundances of aquatic insects (Ramírez and Hernández-Cruz, 2004) relative to continental tropical streams. Similarly, freshwater testate amoebae and microcrustaceans in mosses of Caribbean islands are less species-rich and abundant than mosses in temperate streams (Acosta-Mercado et al., 2012). These results highlight an island effect.
RELATIONSHIP BETWEEN ENVIRONMENTAL VARIABLES AND MEIOFAUNA
Discharge has been suggested as one of the strongest environmental variables affecting meiofaunal distributions (Whitman and Clark, 1984; Swan and Palmer, 2000). Periods of low flow are stable with little sediment movement and minimal organismal catastrophic drift. In Quebrada Prieta, the period with the lowest discharge was registered in February and this month was also when we found the highest abundance of meiofauna (Figure 2). Similar findings have been reported from temperate streams when discharge is low, meiofaunal abundances are high (Rader and Belish, 1999; Gaudes et al., 2010). For instance, higher densities of benthic invertebrates (meiofauna and macroinvertebrates) were observed during periods of low flows in the St. Louis Creek catchment in Denver, Colorado, USA. In this catchment, reaches with low discharge had the highest densities of copepods and chironomids. Increments of more than 3000 individuals were detected when comparing the highest discharge (0.638 m3s-1) with the lowest discharge (0.001 m3s-1) (Rader and Belish, 1999). A similar result was observed in a fourth-order stream in Illinois, USA, where densities of meiofauna during slow flow periods were approximately double than high flow periods (Smith and Brown, 2006).
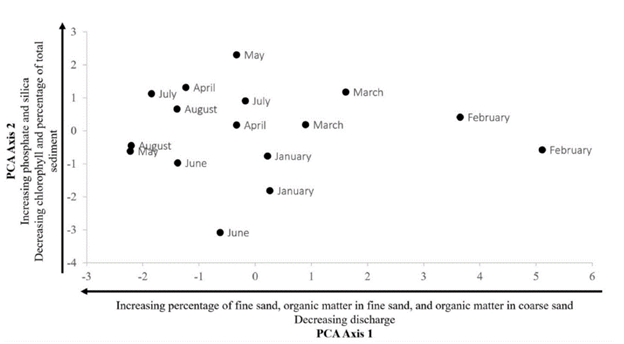
Figure 2 PCA of environmental variables January 2017 to August 2017 in Quebrada Prieta, Luquillo Experimental Forest, Puerto Rico. Axis 1 explains 41 % of the variation, and Axis 2 explains 18 % of the variation.
Sediment, a robust environmental variable affecting meiofauna in temperate streams (Silver et al., 2002; Radwell and Brown, 2008), also affected meiofauna in our study. However, only the percentage of coarse sand was related to meiofauna richness and abundance. Available information on meiofauna comes from streams that are longer and wider than our Caribbean Island streams; thus, higher sediment accumulation might be present. Sediments generate microhabitats that are suitable for meiofauna, which is not necessarily present in mountain Caribbean islands due to the limited sediment depth. Streams with sediment depths of at least 10 cm have been found to have high meiofauna abundances (Radwell and Brown, 2008; Reiss and Schmid-Araya, 2008; Majdi et al., 2017). However, the relative abundance of meiofauna has also been related to coarse sand in second-order streams in England (Reiss and Schmid-Araya, 2008). Other sediment sizes measured were not related to meiofauna abundances in our stream.
In our tropical study stream, sediment characteristics (percent fine sand, organic matter in fine sand, organic matter in coarse sand), discharge, phosphate, silica chlorophyll, and sediment percentage explained environmental variability in Quebrada Prieta. However, no relationship between sediment characteristics (other than the percentage of coarse sand) and meiofauna was found. We attribute this to consistently shallow sediments in Quebrada Prieta. Quebrada Prieta's geomorphology is dominated by boulders with some patches of sand; however, it lacks large areas of sediment accumulation. Most sediment accumulations are shallow and vulnerable to discharge disturbance and high background macro-consumer activity (e.g., shrimps), which reduces the quantity of organic and inorganic sediments (Pringle et al., 1999). In contrast, temperate studies in streams with high sediment depth report a clear relationship between sediment and meiofauna. In temperate streams in Arkansas, United States, the type of particle size of the substratum is a predictor of the abundance of meiofauna. However, in these Arkansas streams, the depth of sediments was at least 10 cm, which was the size of the core sampled (Radwell and Brown, 2008). Quebrada Prieta drains an aseasonal forest and has little seasonality in sediment characteristics, with the two tributaries showing very few differences where the maximum depth of sediment is between 1 to 2 cm (the depth that our PVC tube could be inserted). Nevertheless, the percentage of coarse sand is a strong estimator of meiofauna in our stream. This sediment variable was related to the mean abundance and richness of meiofauna. Even when looking at dominant groups, coarse sand was related to the mean abundance of taxonomic groups Rhizaria (testate amoebae) and Nematoda, and the functional groups bacterivores and detritivores.
Shrimps are dominant in our tropical stream. However, we found no evidence of meiofauna and shrimp interactions. This could be because shrimp densities are more or less constant in Quebrada Prieta; therefore, any effect could be overshadowed. Shrimps are potentially playing an important role in structuring meiofauna by removing inorganic sediments through bioturbation and ingesting algae and fine particulate organic matter (March et al., 2002). Shrimp grazing activity in pool environments has been shown to significantly reduce the biomass of upper story algae and inorganic sediments (Pringle et al., 1999) and to enhance the quality (C:N) of the remaining understory biofilm and organic matter. This results in an enhanced abundance of mayfly insect grazers (March et al. 2002). Therefore, shrimp exclusion studies could detect the role of shrimps in structuring the meiofauna.
Our findings indicate a potentially important role of macroinvertebrates in determining meiofaunal community composition. Macroinvertebrates in Quebrada Prieta are dominated by shredders, collectors, and filter-feeders and are composed of insects and shrimps (Ramírez and Hernández-Cruz, 2004). These organisms consume coarse organic matter transforming it into fine organic matter (Anderson and Sedell, 1979), which represents organic matter available for small organisms, such as meiofauna and bacteria. Therefore, it is not surprising that most meiofauna recorded in Quebrada Prieta were mainly bacterivores or detritivores. Low richness and abundance of algivorous or predator meiofauna were recorded. Also, shrimps remove small fine particles (March et al., 2002; Macias et al., 2014), which could benefit meiofauna by reducing the number of small particles in the streambed, thus promoting mayflies which could facilitate the enhanced fine organic matter. Thus, we hypothesize that macroinvertebrates have positive effects on meiofauna via organic matter consumption.
CONCLUSIONS
This is the first study on lotic meiofauna in the headwaters of Luquillo Experimental Forest, El Yunque, and, as far as we are aware, one of the firsts in the tropics. The meiofauna in Quebrada Prieta is similar in composition to temperate streams when taxa are classified by phyla. However, our tropical meiofauna is dominated by testate amoebas, ciliates, and nematodes. As in temperate streams, discharge and sediment (coarse sand only) have a strong effect on meiofauna abundance and richness. However, macroinvertebrate abundance is likely to play a role in regulating the abundance of meiofauna in our study.