INTRODUCTION
Mangrove ecosystems are complex and dynamic, particularly the radical submerged system of Rhizophora mangle, which forms an intricate network spanning from the upper level to the sublittoral. The almost cylindrical shape of the root constitutes a substrate with a wide contact surface that is available for the colonization of sessile organisms, among which floral algae stand out. These organisms, along with diverse groups of invertebrates, can establish communities with high species richness (Ellison and Farnsworth, 1992; Hogarth, 2007).
Within mangroves, macroalgae play an important role since they act as primary producers of these ecosystems’ complex trophic chain. They also have great ecological value, as they build natural filters that retain sediments and contribute with large amounts of organic matter in the form of enriched detritus, which is transported from the mangrove to the neighboring ecosystems, representing its biomass. This constitutes a significant contribution in the production of organic carbon. In the same way, macroalgae are also environmental indicators of physicochemical changes caused by natural or anthropogenic events (Steinke and Naidoo, 1990; Laursen and King, 2000).
In the Caribbean, numerous studies on macroalgae associated with mangrove environments have been carried out, most of them focusing on taxonomic or inventories works. Cordeiro-Marino et al. (1992) elaborated the first compilation for the region, reporting 109 macroalgae species. Later, in the Mexican Caribbean, Collado-Vides et al. (1995) identified 52 macrophyte species associated with mangroves in a lagoon system, In Cuba, Martínez-Daranas (2005) elaborated an inventory, reporting 163 macroalgae associated to R. mangle roots and indicating the predominance of pantropical and cosmopolitan species.
In the Panama Caribbean, Ríos et al. (2019) described the vertical zoning of macroalgae in the roots of R. mangle, identifying 11 species of the phylum Rhodophyta, out of which Bostrychia montagnei, B. moritziana, B. tenella, Polysiphonia howei, P. scopulorum, Acanthophora spicifera, and Caulerpa caespitosa were the most abundant; while, in the Colombian southwestern Caribbean, 36 species have been reported which are associated to the R. mangle root system (Salazar-Forero et al., 2021), constituted by 21 Rhodophyta, 11 Chlorophyta, and four Phaeophyceae. Nevertheless, these last studies highlight the need to generate more knowledge on the biodiversity, distribution, and seasonality of the phycofloristic community associated with red mangrove, in order to better understand the dynamics and ecological services that it provides to mangroves in the Caribbean.
In Venezuela, the first works on macroalgae in mangroves were carried out by Post (1936; 1963), who studied the association of red algae of the genera Bostrychia and Caloglossa. Hammer and Gessner (1967) identified macroalgae present in mangroves of the eastern coasts of Venezuela. Díaz-Piferrer (1970) elaborated an inventory that mentions some species associated with red mangrove roots in the Gulf of Venezuela, the Maracaibo Lake, and the coasts of the states of Anzoátegui, Sucre, and Nueva Esparta. González (1977), Lobo and Ríos (1985), Albornoz (1988), González and Vera (1994), and Vera (2004) contributed with lists of macroalgae for Falcón and particularly the Morrocoy National Park.
López et al. (2009) identified 40 macroalgae species in R. mangle roots in the Paraguaná Penninsula (Falcón), being this work the one that mentions the largest number of species for this substrate in the country. In eastern Venezuela, two papers stand out: Barrios et al. (2004) in the Gulf of Santa Fe and Barrios and García (2013) in Caño Mánamo-Delta Amacuro. These works report the association of Bostrychietum that is typical in mangroves, where there is a predominance of rhodophytes with numerous adaptation mechanisms for desiccation and fluctuations in salinity and temperatures. In light of the need to generate more knowledge on macroalgae in R. mangle roots and considering their importance as a basis for supporting the primary and secondary productivity of these environments, a phycofloristic survey was conducted with the purpose of assessing the spatial-temporal composition of macroalgae associated with R. mangle roots in two coves of the southeastern Caribbean of Venezuela.
MATERIALS AND METHODS
Study area
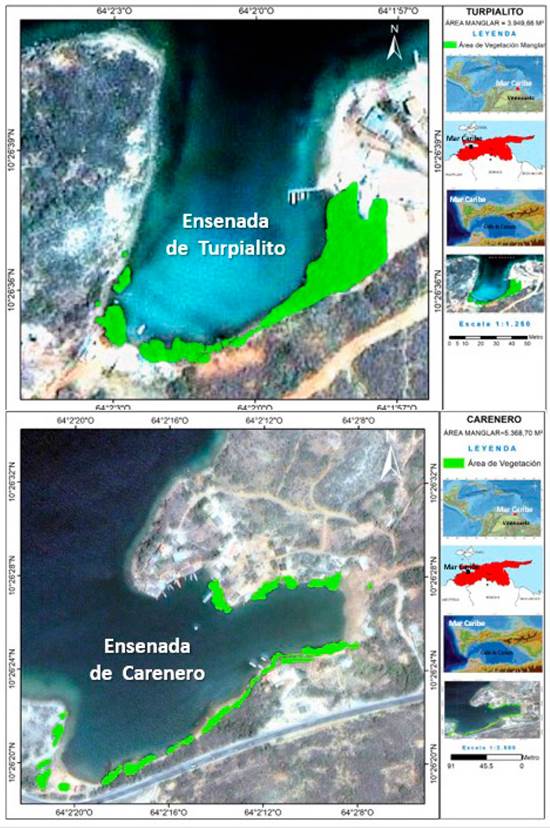
The coves of Turpialito (10º 26’ 5” N, 64º 02’ 56” W) and Carenero (10 º 26’ 42” N, 64° 2’ 16” W) are located in the southern coast of the Gulf of Cariaco, in the southeastern Caribbean of Venezuela. The mangroves that surround Carenero cove are constituted by R. mangle, Laguncularia racemosa C.F. Gaertn, Conocarpus erectus, and Avicennia germinans. The mangrove composition of Turpialito is similar, except for the latter. R. mangle is the dominant mangrove in both coves, occupying an area of 5368 Figure 1.70 m2 in Carenero and 4001.43 m2 in Turpialito. The mouth length of these coves varies between 118.54 and 175.97 m, and it is greater in Turpialito
Sampling design
The evaluated coves have similar marine environments, with coral and Thalassia testudinum patches, as well as substrates lacking vegetation and rocky areas associated with mangroves. Based on these different environments and the red mangrove cover, four stations were established in each cove, which were distributed as follows:
Carenero cove
Stations 1 and 2 have well-develop roots entirely submerged to depths of 1.0 to 1.2 m, respectively, with T. testudinum and coral patches. Station 3: the mangrove is less developed. Its roots are submerged to an average depth of 0.5 m, and they are subjected to tidal changes and little presence of T. testudinum. Station 4: is located in front of the cove mouth, adjacent to a rocky zone and exposed to moderate currents throughout the year, with less developed roots at an average depth of 0.45 m.
Turpialito cove
Stations 1 and 2 are close to T. testudinum patches, with average depths between 0.7 and 1.0 m. Station 3: the red mangrove is little developed. It is adjacent to a very fine sand bank, which is why its roots are little submerged (0.55 m). Station 4: the mangrove is located in front of the cove mouth in a rocky zone. It is exposed to moderate currents, with an average depth of about 0.5 m.
Environmental factors
With the purpose of understanding whether there are spatial-temporal changes, temperature and salinity records were obtained in triplicate for each cove using a 6600v2 multiparametric probe (model YSI6600).
Obtaining the phycoflora
Macroalgae were collected bimonthly from November 2012 until September 2013. In each cove, three R. mangle roots from each station were randomly selected. The algae were manually collected by scraping the root, with the purpose of extracting them with their holdfast structures. The samples were placed in previously labeled polyethylene bags and wrapped in paper, so that they maintained enough humidity and remained in good shape for a longer time. In the laboratory, each sample was carefully washed and placed in containers with a 4 % formaldehyde solution in seawater. For the taxonomic identification of the algae down to the lowest possible taxon, the most distinctive morphological features were observed through histological cuttings made by freehand, with which semi-permanent plates were prepared, using glycerin at 30 % as a means of mounting, according to the modified Womersley technique (Ramírez, 1995). The observation was carried out using a Labomed LX400 bright-field microscope. The collected samples were herborized and incorporated to the Phycological Herbarium of the Oceanographic Institute of Venezuela, Universidad de Oriente.
Taxonomic identification
The works by Taylor (1960), Chapman (1961) , 1963), Joly (1967), Lemus (1979); 1984), Aponte (1985), Littler and Littler (2000), and Dawes and Mathieson (2008) were used for taxonomic identification. The classification and distribution of macroalgae was corroborated according to the catalogs by Ganesan (1989), Wynne (2017), Carballo-Barrera et al. (2020), and Guiry and Guiry (2020). The different morpho-functional types of the macroalgae were determined according to the criteria proposed by Steneck and Dethier (1994).
Floristic composition
Ecological indices were calculated, such as Specific Richness (S) by counting the total number of species identified in each cove and sampling station (Margalef, 1986) and Specific Constancy (C), which was obtained using the formula defined by Krebs (1985), for which three categories are established: C = Constant (species in more than 50 % of the samplings), A = Accessory (species in 25-49.9 % of the samplings, and Acci = Accidental (species in less than 24.9 % of the samplings).
Statistical analysis
To compare the temperature and salinity values between the sampling months and stations, a Kruskal-Wallis non-parametric analysis of variance was performed (Siegel and Castellan, 1994) using the Statgraphics Centurion 5.1 statistical software.
RESULTS
Environmental factors
The average temperature values showed significant differences for both coves (Carenero: KW P = 0.02, Turpialito: KW P = 0.003), with no significant variability between stations (Carenero: KW P = 0.88, Turpialito: KW P = 0.58). In Carenero (Figure 2 a-b), the temperature oscillated between 29.4 ºC (November/12) and 23.3 ºC (September/13), whereas, in Turpialito (Figure 3 a-b), this factor fluctuated between 29.8 ºC (November/12) and 25.5 ºC (January/13).

Figure 2 a) Bimonthly and b) spatial water temperature variation in the Carenero cove (Gulf of Cariaco) in the southeastern Caribbean of Venezuela.

Figure 3 a) Bimonthly and b) spatial water temperature variation in Turpialito cove (Gulf of Cariaco) in the southeastern Caribbean of Venezuela.
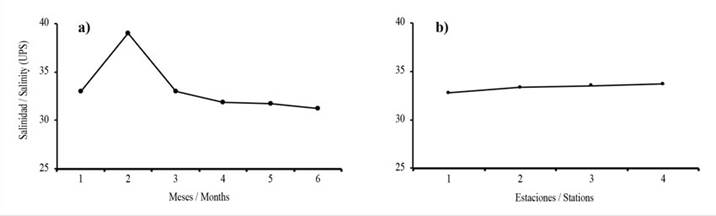
Salinity showed significant temporal differences (Carenero: KW P = 0.001; Turpialito: KW P = 0.003), but not between stations (Carenero: KW P = 0.85; Turpialito: KW P = 0.90). In Carenero (Figure 4 a-b), this parameter oscillated between 31 in November/12 to 36.2 in January/13, whereas, in Turpialito (Figure 5 a-b) a greater variation was recorded, with an oscillation between 33 and 39 for the same months.

Figure 4 a) Bimonthly and b) spatial water salinity variation for Carenero cove (Gulf of Cariaco) in the southeastern Caribbean of Venezuela.
Taxonomic identification
A total of 23 macroalgae species were identified, grouped in 12 families -Chlorophyta (11), Ochrophyta (7), and Rhodophyta (5)- and distributed according to their functional morphotypes in seven foliaceous, seven siphonal, four corticated macrophytes, three filamentous, and two articulate calcareous (Table 1).
Floristic composition
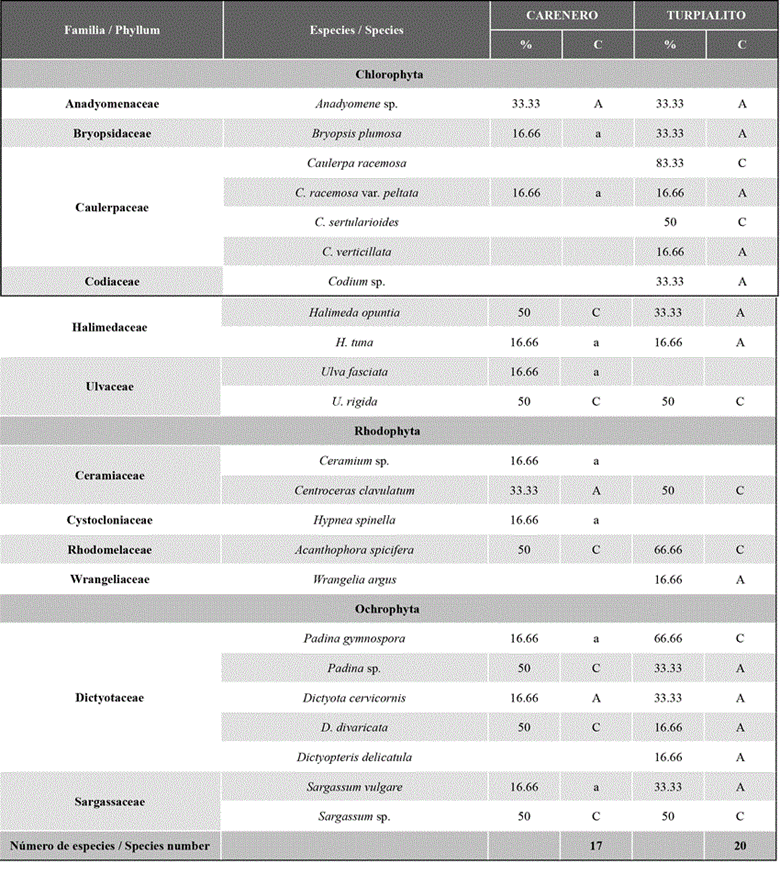
The number of species identified by cove was 20 in Turpialito and 17 in Carenero, with 14 common species for both coves. No algae were recorded during November 2012 in any cove. In Carenero, temporal changes were observed in the number of species collected, with the greatest richness values in March (8), May (9), and September (10) 2013; whereas stations 2 and 4 showed the greatest richness (9 and 11 species, respectively). In Turpialito, The number of identified species showed no noticeable changes, with the greatest richness being recorded in March (10); whereas, in the remaining months, eight or nine species were identified. The same behavior was observed between stations, where richness showed little variation, with values oscillating between 12 and 13 species (Figure a-b).
Regarding constancy, in the Carenero cove, six species were constant, three accessory, and eight accidental; in the Turpialito cove, seven were constant, seven accessory, and six accidental (Table 2).

Figure 6 a) Bimonthly and b) spatial variation in the richness of macroalgae associated with the roots of the red mangrove R. mangle at the sampling stations of the Carenero and Turpialito coves (Gulf of Cariaco) in the southeastern Caribbean of Venezuela.
DISCUSSION
The algal composition of the Carenero and Turpialito coves is similar to that reported by Barrios et al. (2004) for R. mangle in the Gulf of Santa Fe. Out of the species found, U. fasciata, C. sertularioides, A. spicifera, S. vulgare, C. clavulatum, and C. chemnitzia were also identified in this study. Vera (2004) identified 30 macroalgae species associated with red mangrove in the Morrocoy National Park, while López et al. (2009) reported 40 species in the Paraguaná Penninsula. Out of the macroalgae identified in this work, seven are new records for the Gulf of Cariaco: Anadyomene sp., C. verticillata, H. tuna, U. rigida, H. spinella, D. implexa, and D. delicatula.
The high diversity and abundance values of algae associated with R. mangle roots in the Caribbean are related to the stability of factors such as salinity, water transparency, irradiation, temperature, and the moderate amplitude of the tides, in addition to the greater availability of hard substrates, among them corals, limestones, and mangrove roots, which generate dynamics that favor the richness of phycoflora associated with red mangrove roots (Ríos et al., 2019; Salazar-Forero et al., 2021). The composition of algae associated with red mangrove roots in the Carenero and Turpialito coves is characteristic of mangroves in the Caribbean (Taylor, 1960; Cordeiro-Marino et al., 1992; Salazar-Forero et al., 2021), as it is made up of species that generally inhabit spaces protected by calm and shallow waters.
In November 2012, no algae were found in the sampled roots, which is related to the strong rainfall that took place in that period, which generated an important runoff water flow, high turbidity, and below-average salinities (25.5) in the area. After this period, an important recovery of the phycoflora was observed in the red mangrove roots. The number of species identified was similar in both coves. In Turpialito, the floristic composition showed no spatial-temporal changes, despite the variations in temperature and salinity. The greatest algae richness was collected in the distal region of the root, even though the distribution of many macroalgae species generally encompasses the root as a whole, including its upper area, as is the case of the genera Ulva, Bryopsis, and Centroceras, which suggests that these species can endure cyclic emersions and desiccations.
On the contrary, the algal flora of Carenero showed spatial-temporal fluctuations, and it was recorded mainly in the distal portions of the root, albeit in the form of plumes. A community of invertebrates associated to them was observed in the roots, which was visibly greater to that observed in Turpialito. The composition of macroalgae species present in the mangrove roots depends, to a great extent, on the nature of the pioneer organisms that colonize the substrate.
In Carenero, the dominant species were A. spicifera, H. opuntia, and U. rigida, while C. racemosa, A. spicifera, and U. rigida were dominant in Turpialito. Martins et al. (1999) point out that most of the species in these genera can tolerate wide salinity ranges, hence their abundance in mangroves. The dominance of Caulerpa racemosa, C. racemosa var. peltata C. sertularioides, C. verticillate, and A. spicifera could be related to their tolerance to intertidal changes (Dawes and Mathieson, 2008), while the morphological and physiological adaptability of many microalgae species allows them to have a wide bathymetric and spatial distribution that favors their presence in different environments (Marín and Peña, 2016).
Acanthophora spicifera is an indicator of high organic material contents and tends to be located at the ends of R. mangle roots (Barrios et al., 2004). Given its cespitose structure and the development of a basal disk, it can take advantage of the ends of the roots to grow, which, given their reduced area, constitute areas of less competition with fouling organisms that need more surface to settle. Nevertheless, the stoloniferous growth and abundance of rhizoidal projections of Caulerpa allow it to adhere along the mangrove root. In the case of Ulva, the representatives of this genus are widely distributed worldwide and tend to be opportunistic organisms that successfully colonize vertical substrates such as mangrove roots. It can be added that U. fasciata could be an indicator of some degree of contamination in Carenero, as this species, and all Ulvales in general, are characteristic of eutrophic areas (Van den Hoek et al., 1998; Lotze and Schranm, 2000).
The percentage of constant species in both coves was similar, but, when comparing Carenero and Turpialito, a greater presence of accidental species was observed in the latter. Barrios et al. (2004) point out that it is possible to find untypical or accidental species in algae communities associated with mangroves. The availability and viability of propagules and spores, transported by the currents and waves, as well as the adequacy and type of substrate, are factors that influence the distribution and connectivity of sessile organism populations (Gaylord et al., 2006). Another determining factor of this difference is that, in Turpialito, there is a greater variety of environments close to mangroves, such as soft sandy and muddy-loamy bottoms, coral patches, T. testudinum pastures, rocky areas, anthropogenic structures such as docks and crop systems, which allow a greater incidence of species that manage to colonize mangrove roots.
Among the species associated with R. mangle roots in the Turpialito cove, macroalgae were identified which are characteristic of nearby reefs and soft bottoms, such as C. sertularioides and Halimeda opuntia, which suggests that the richness of macroalgae associated with red mangrove could be the product of the incorporation of species from other environments. Collado-Vides et al. (1995) found, in the lagoon system of Nichupté (Mexico), 17 species exclusive to R. mangle roots and 35 macroalgae species both on the roots and on sandy bottoms consolidated with marine grasses. Their study highlighted that there are morphological elements in many macroalgae that do not limit their distribution, which allows them to colonize both substrates.
The closeness of coral patches and rocky areas and particular physical and biotic factors interact to produce the phycofloristic composition associated with roots that was observed in the aforementioned coves. In a study conducted in Colón Island, in the Bocas del Toro province of the Panama Caribbean, most macroalgae were distributed in the medium and distal areas of the roots, which were always submerged, thus suggesting that many of the species present there, such as A. spicifera and H. spinella, among others, colonize the R. mangle roots growing in protected spaces (Ríos et al., 2019).
However, it must be considered that not all macroalgae species that are common in these environments may be represented in the roots of these mangroves, either because of the high contents of tannins and other secondary products of the roots (Conde et al., 1995), the competition is generated for space with other organisms during the colonization process, or the frequent shading caused by plant cover, which reduces the available light. Ogden and Gladfelter (1986) indicate that coastal mangroves are a functional component of mangrove-marine grass-coral reef complexes, and even though the interactions between these ecosystems are not clearly defined, the complex root system, macroalgae, and other organisms that colonize them reduce the impact of runoff and resuspension sediments on coral communities.
Some global environmental phenomena taking place within the Gulf of Cariaco, such as coastal upwelling, which occurs between December and April and May (Okuda et al., 1978), together with other characteristics of each cove, such as the width, depth, the structural complexity of the mangrove roots, bivalve collection (e.g., oysters) by fishermen -which upsets the dynamics of the communities on the roots- and the presence of other ecosystems in neighboring areas play an important role in the spatial-temporal distribution of the phycoflora associated with mangroves in Carenero and Turpialito. The topographic characteristics, structure, and disposition of mangroves within both coves, together with the closeness and biological exchange with different marine environments, are fundamental to the composition and assembly of the phycoflora associated with R, mangle in Carenero and Turpialito, which, despite being close and sharing environmental similarities, exhibit different algal compositions.











 text in
text in