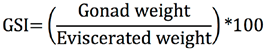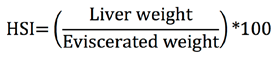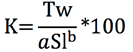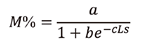INTRODUCTION
The state of Nayarit, located in the southeastern Gulf of California, has a 296 km coastline, which represents 2.7 % of the Mexican total (Conapesca, 2013). The fishery in the state uses a great diversity of fishery resources and natural ecosystems to carry out its activities, thus keeping a permanent pressure on fish, mollusks, crustaceans, and other aquatic organisms. The marine flake fishery consists of an important group of both marine and estuarine fish, specifically the species commonly known in the region as mojarra, the northern red snapper, chiuiles, the sea bass, sawfish, planiliza, and corvina, among others (Ramírez-Zavala and Bojórquez-Sauceda, 2006).
The striped weakfish Cynoscion reticulatus is distributed from the south of the Baja California Peninsula - including the Gulf of California - to Colombia, and it inhabits some coastal waters between 1 and 107 m deep (Robertson and Allen, 2008). It belongs to the corvina family, Sciaenidae, which includes 66 genera and approximately 291 species (Eschmeyer and Fong, 2013), distributed in temperate and tropical regions around the world and found in rivers, lakes, lagoons, and estuaries as well (Parenti, 2020). Corvinas or roncadores are regarded as predators with ecological and economic importance in the coastal systems where they are distributed (Chao, 1995). They are a eurythermal and euryhaline species, resistant to sudden changes in temperature (from 2 to 38 ºC) and salinity (from 5 to 39 g L-1) (Sabater-Pascual, 2012). There are few studies related to the reproduction of the genus Cynoscion in areas of the Eastern Pacific (Palacios et al., 2015). In general, it has been reported that sciaenid reproduction occurs between spring and summer, with adults usually moving along the coastline and grouping in large schools of fish to spawn in estuaries and coastal lagoons. Apparently, the family Sciaenidae includes iteroparous and gonochoric species, with partial and indeterminate spawning and asynchronous ovarian development (Cárdenas, 2012).
In San Blas, Nayarit, fishing activity is an important source of income for the population, and the striped corvina C. reticulatus is one of the main commercially used species. Due to its size and abundance, this sciaenid is caught throughout the year without restrictions, making it a very attractive resource for commercial coastal fishermen and sport fishing. Despite the above, there is not enough biological and/or ecological information that can be used to plan and implement fisheries management measures for sustainable fishing. In this way, this work aims to describe the reproductive biology of the striped weakfish C. reticulatus, as well as the existing relationship with the environmental factors (sea surface temperature (SST), chlorophyll, and photoperiod) of the area it inhabits, in order to generate information that provides elements for improving its sustainable utilization.
MATERIAL AND METHODS
Study area. The municipality of San Blas is located in the northern region of the state of Nayarit (21º 20’ and 21° 43’ N; 105° 02’ and 105° 27’ W), Mexico, to the southeast of the Gulf of California. It has an area of 849.78 km² and ranks twelfth in terms of territorial extension (3.0 % of the surface of Nayarit), and it is the municipality with the highest fishing activity in the state (Figure 1) (Buhaya-Lora and Ramírez-Partida, 2015).
Figure 1 Study area used to catch striped weakfish, Cynoscion reticulatus, by fishermen in San Blas, Nayarit in the southeastern Gulf of California.
Environmental factors. Photoperiod data were obtained from the Puerto Balleto Oceanographic Station, Nayarit, using the Mar V1.0 2011 software, at Centro de Investigación Científica y de Educación Superior de Ensenada (CICESE). The SST and chlorophyll were determined from satellite images. Monthly averaged images from an image composition of MODIS-Terra and MODIS-Aqua were used, with a spatial resolution of 1 km2 (Kahru et al., 2004).
Sample collection. Between 17 and 40 organisms per month (depending on availability) were collected from commercial coastal fishery in the port of San Blas, Nayarit, between January and December 2019. The fishing gear used for catching included gill nets ((mesh sizes 5.1, 6.3, 7.6, 8.9 and 10.2 cm (2, 2 ½, 3, 3 ½, and 4”)), shrimp nets ((mesh sizes from 5.1 to 8.9 cm (2 to 3½”)), and longlining with different hook sizes (#8 to #2) in order to obtain more size representativeness for the study.
Sample processing. The standard length (Sl) of the obtained specimens were measured with an ichthyometer, and the total weight (Tw), the eviscerated weight (Ew), the gonad weight (Gw), and the liver weight (Lw) with an electronic balance (± 0.01g accuracy). The gonads were fixed in 10 % formalin, saturated with sodium borate. For the quantitative and qualitative analysis of gonadal development, the conventional histological technique was applied (hematoxylin-eosin), and the samples were observed with an optical microscope (Zavala-Leal et al., 2022).
Sex ratio. The sex ratio was determined monthly (for each sampling) and the total sex ratio. Statistical significance was tested using a chi-squared (χ2) test, with a null hypothesis of one female per male (H0 = 1:1) (Zar, 2010). Once the sex of the organisms was known, a t-Student test was applied to determine the size and weight differences for each sex.
Morpho-physiological indices. The gonadosomatic and hepatosomatic indices, as well as the condition factor, were calculated. The gonadosomatic index (GSI) was obtained through the expression used by Vazzoler (1996):
The hepatosomatic index (HSI) through the Rodríguez-Gutiérrez equation (1992):
The condition factor (K) was determined with the equation proposed by Le Cren (1951):
Where: K = Condition factor, Tw = Total weight, Sl = standard length, a = intercept, b = slope.
Gonadal development. The gonadal developmental stages were identified in order to determine the reproductive cycle (as a function of the frequencies of the developmental stages) and thus the reproductive season. For females, six developmental stages were used: resting, initial development, advanced development, maturity, spawning, and post-spawning. Meanwhile, five developmental stages were used for males: resting, development, maturity, spermiation, and ejaculation. Relative frequencies were calculated monthly for the gonadal development stages in both sexes. The reproductive season was considered to be the period when 50 % of the organisms were observed with advanced gonadal development stages, both for females (advanced development, maturity, and spawning) and for males (maturity and spermiation). All this was done according to Sánchez-Cárdenas et al. (2011), with minor adjustments for this particular species based on our observations.
Relationship between reproduction and environmental factors. In order to determine whether there is a relationship between the gonadal development of the striped weakfish and the environmental conditions prevailing in the study area, the Spearman correlation analysis was performed. For this purpose, the monthly frequencies of gonadal development and the monthly averages of SST, photoperiod, and chlorophyll-a were used. The Spearman coefficient can take a value of +1 or -1, where +1 indicates a perfect (direct) association, a value of -1 indicates a perfect negative (inverse) association, and a value of 0 indicates no association. This analysis was performed using the StatSotf® Statistica 7.1 software.
Maturity size. This was defined as the length at which 50 % (L50) of the sampled fish were sexually mature (Sánchez-Cárdenas et al., 2011). Based on this concept, the relative and the cumulative frequencies were calculated for each sex per size interval of the organisms considered in reproduction. To obtain the L50, the data were adjusted to a logistic model using the CurveExpert 1.4 software.
Where M % = percentage of mature individuals, a = intercept, b = slope, c = constant, and Sl = standard length.
RESULTS
A total of 390 specimens of C. reticulatus were obtained. They showed an average size and weight of 27.9 cm Sl, and 344.4 g for both sexes. It was observed that females had greater length and weight than males (p= 0.0000 and p= 0.0010, for Sl and Tw, respectively). 264 females and 144 males were found. Females showed minimum and maximum sizes of 18.5 cm and 110 g in weight up to 40.3 cm Sl and 960 g, while males showed 19.7 cm and 120 g up to 38,6 cm Sl and 925 g.
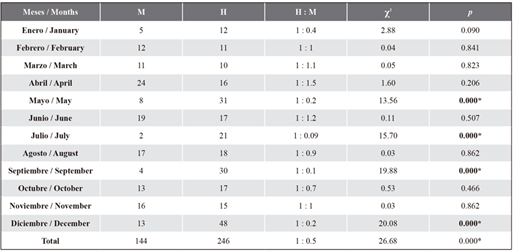
Sex ratio. According to the chi-squared test (χ2), the sex ratio was found different from 1:1 (M:H) for the whole sample. However, there were no significant differences between males and females in most months, except for May, July, September and December (Table 1).
The asterisk (*) denotes significant differences (p ≤ 0.05).
Morpho-physiological indices. The striped weakfish C. reticulatus showed a gonadosomatic index with high values from April to August, and low values for this index from January to March for both males and females (Figure 2). The hepatosomatic index showed the highest values from April to May and was relatively constant from June to December for both sexes (Figure 2). The Condition Factor also showed a pattern similar to GSI and HIS, with higher values in April (Figure 2).
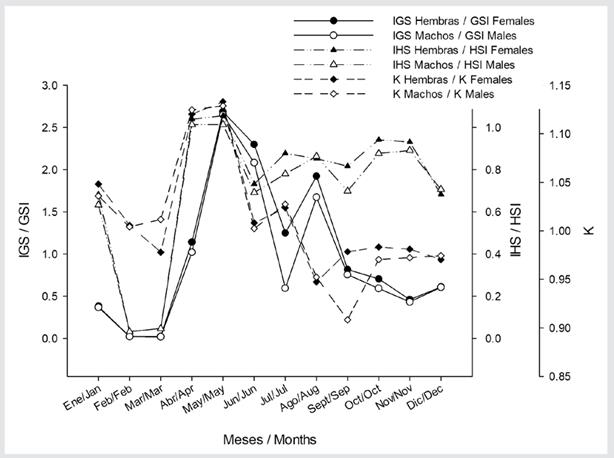
Figure 2 Morpho-physiological indices in striped corvina Cynoscion reticulatus from the southeastern Gulf of California.
Gonadal development. The corvina shows an asynchronous gonadal development. Females show a concentric lamellar organization, whereas, in males, the seminiferous tubules are arranged longitudinally. Six developmental stages were described for females (resting, initial development, advanced development, maturity, spawning, and post-spawning) (Table 2), while five stages were described for males (resting, development, maturity, spermiation, and ejaculation) (Table 2).
Table 2 Gonadal developmental stages of striped weakfish C. reticulatus from southeastern Gulf of California.

Regarding the reproductive cycle, it was observed that females show a high proportion of organisms in advanced developmental, mature, and spawning stages from May to August, while females in resting and initial developmental stages are predominant from October to March. Males are mature and in spermiation from April to September, and a high percentage of organisms in resting and developmental stages is observed from October to March (Figure 3).
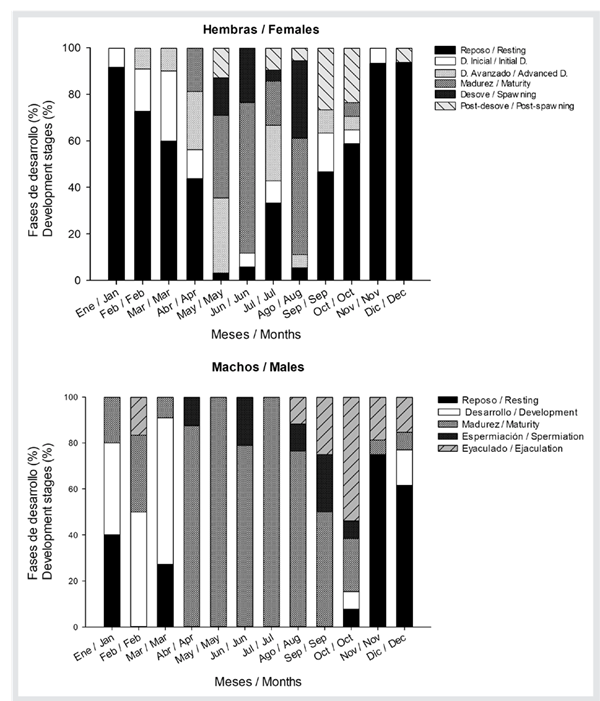
Figure 3 Gonadal developmental stages of striped weakfish C. reticulatus from southeastern Gulf of California.
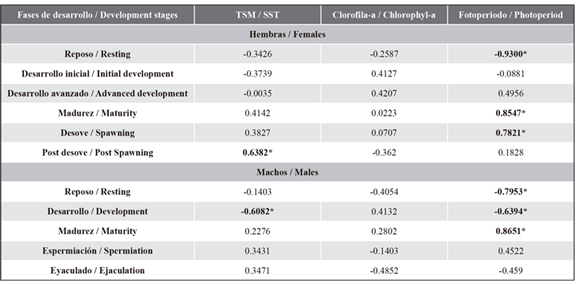
Relationship between reproduction and environmental factors. The Spearman correlation analysis showed that the gonadal development of C. reticulatus was related to the SST and the photoperiod, but not to the chlorophyll-a concentration. Specifically, a positive correlation between the SST and the spawning stage was observed in females, while the photoperiod maintained a positive correlation with the maturation and spawning stages, as well as a negative one with the resting stage. In males, only a positive correlation between the photoperiod and the maturity stage was observed, whereas the resting and development stages showed negative correlations with the photoperiod and the sea surface temperature (Table 3).
Table 3 Correlations between the reproductive events of striped weakfish C. reticulatus and the environmental factors in the southeastern Gulf of California
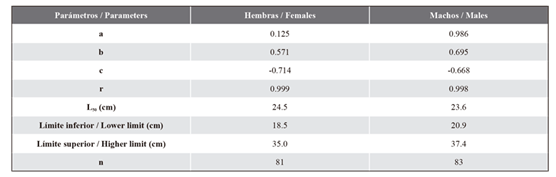
Sexual maturity size. The striped corvina showed a relatively smaller mean size at maturity for females than for males. For females, L50 was 24.5 cm of Sl, whereas, for males, it was 23.6 cm (Figure 4). The maturity intervals and adjustment parameters of the applied sigmoidal model are shown in Table 4.
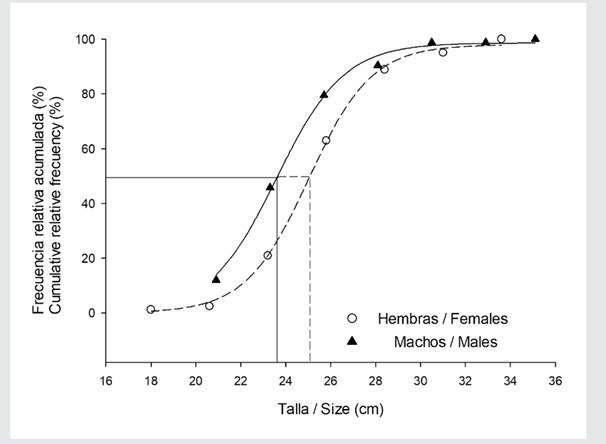
Figure 4 Sexual maturity size (n=164) of the striped weakfish C. reticulatus from southeastern Gulf of California. The lines indicate the intersection of the cumulative relative frequency and the organisms’ size.
DISCUSSION
Determining the size composition of a fish population is of the utmost importance, as it allows knowing the different biological stages they go through and differentiating the population groups of a species in a fishery (Longart et al., 2011). According to the results regarding the size of each sex in the striped weakfish C. reticulatus, females were larger than males. Ortiz et al. (2021) report very similar sizes and weights for this species in Guatemala, (Ẋ = 32.48 cm and 340.05 g). In a study conducted in 2016 on the composition of the ichthyofauna caught in the artisanal fishery of Isla Isabel, Nayarit, Mexico (Torrescano-Castro et al., 2016), larger individuals of C. reticulatus were found (Ẋ = 40.8 cm and = 619.4 g). The difference between the sizes stated in the aforementioned work and the other two can be attributed to the different fishing gear used, since this work reports the use of gill nets with mesh sizes of 8.9, 10.2 and 12.7 cm (3.5, 4 and 5”) (for 96.4 % of the total catch), while Ortiz et al. (2021) report the use of gill nets with mesh sizes of 5.1 and 7.6 cm (2 and 3”). In this study, 2 to 4” mesh sizes were used. Furthermore, unlike the study area reported by Torrescano-Castro et al. (2016), which is 35 km away from the nearest coast, the other two studies were conducted in the estuary and adjacent coastal areas, which have a high fishing pressure in both Nayarit and Guatemala (Ramírez-Zavala and Bojórquez-Sauceda, 2006; Ortiz et al. 2021). Another factor to consider in the size difference could be the fact that, according to information from the NOAA Weather Forecast Center (NOAA, 2022), the study was conducted in a representative year, whereas that of Ortiz et al. (2021) was conducted in the midst of La Niña. Our study was conducted during part of El Niño.
As for the sex ratio, it is a parameter used as a basis to interpret the composition of exploited stocks and the variations in their abundance (Punt et al., 1993). In this study, the chi-squared test showed significant differences (χ2= 26.68; p< 0.05) in the sex ratio between females (63 %) and males (37 %). Ortiz et al. (2021) report similar data for the same species, with a higher sex ratio for females, as is the case of the golden croaker Micropogonias altipinnis. The differences observed in the sex ratio (1:1) can be attributed to differences in growth rate between males and females, resulting in greater or smaller catch of individuals of either sex. It may also be due to the different feeding behavior of the sexually mature fish, as well as to the selectivity of the fishing gear (Tello-Martín et al., 1992). In turn, Jaramillo-Londoño (2010) argues that, depending on the behavior of the species, a greater number of females can be mostly observed as the sexual maturity and spawning months approach, which coincides in some aspects with the conducted study. In the gulf weakfish Cynoscion othonopterus, a sex ratio of 1:1 (H : M) has been reported (Acosta-Valenzuela, 2008), but also a higher proportion of males (1:2) (Román-Rodríguez, 2000). According to Nikolsky (1963), the sex ratio can vary considerably between species, between populations and even from one year to another within the same population due to changes in environmental factors and food availability, among others.
According to the frequency analysis of the gonadal development stages, the striped weakfish showed a broad period of reproductive activity. In this species, a high proportion of mature females and males was observed between April and June. This coincides with high GSI values, which also shows that the reproduction in this area takes place from May to August. In the Central Pacific in Guatemala, this species was found to have a broad reproductive period with two distinct peaks, one of lower intensity from March to April and the most representative from July to August (Ortiz et al., 2021). According to Lowerre-Barbieri et al. (2011), the duration of the spawning season is fundamental for reproductive success and can be affected by fishing issues. Regarding the population, the spawning seasonality varies in duration and in the degree of synchronization between individual spawning periods and the season in which they occur (fall-winter or spring-summer). It has been reported that an extended or expanded spawning period is more common in warm seasons (Pavlov et al., 2009). As for other sciaenids in México such as the gulf weakfish (C. othonopterus), it has been reported to show a higher reproductive activity in April and May, during its migration towards the upper Gulf of California (Ríos-Medina, 2012). Valdez-Muñoz (2010) and Landeros (2016) report that the totoaba (Totoaba macdonaldi) reproduces at the mouth of the Colorado River from the end of February until June, observing that the highest reproductive peak occurs from April to June. For other species of the genus Cynoscion in the Gulf of Montijo, Panama, the reproductive season usually varies between species. Robles-Pineda (2007), reports that the queen weakfish (C. albus) shows mature organisms from October to May, at the end of the rainy season and in the middle of the dry season, while the cachema weakfish (C. phoxocephalus) has two distinct seasons, the most intense being in January and the weaker in June. For the scalyfin weakfish (C. squamipinnis), mature organisms were observed throughout the year, mainly in the rainy and dry seasons. It has been reported that, for some sciaenids, there are migrations with reproductive purposes up to the river mouths and estuarine lagoons, coinciding with the rainy seasons (Cárdenas, 2012; Cotero-Altamirano et al., 2018).
The influence of abiotic factors has been extensively studied in fish, with the aim to learn their reproductive biology and be able to control different biological aspects for achieving their reproduction. Temperature and salinity are among the main controlled factors. However, there are other environmental variables such as the photoperiod which can influence the development of fish (Aragón-Flores et al., 2014). In this study, it was observed that the striped corvina showed a positive correlation with the sea surface temperature and the photoperiod. Palacios et al. (2015) report that the continuous increase and decrease in temperature, with the appropriate number of light hours, have an effect on the diameter and maturity degree of the oocytes of the captive C. phoxocephalus. It has been widely demonstrated that annual changes in the photoperiod are responsible for the onset of gonadal maturation, activating the endocrine reproductive axis of fish with the consequent use of energy resources by the reproductive tissues. Instead, the temperature is a synchronizing factor that indicates whether the conditions are suitable for reproduction and induces the final maturation of the gametes and the spawning (Estrada-Godínez et al., 2014).
Regarding the sexual maturity size, a slightly higher size was reported for this same species in the Guatemalan Pacific (Ortiz et al., 2021). This difference can be attributed to the way in which the measurements were taken, as these authors report total length, while this study reports standard length. In the gulf wakfish C. othonopterus, variations in maturity size have been reported in the same location for different years (2008: L50= 50.2 cm, 2013 = 55.9 cm, 2014 = 49.9 cm and 2015 = 48 cm) (Mendivil-Mendoza et al., 2018). According to these authors, the differences in size at maturity can be attributed to the estimation method used, to bias in fishing (sampling), and to the fishing pressure that the population can withstand during a given period.
CONCLUSIONS
This is the first work aimed at understanding the reproductive biology of Cynoscion reticulatus in the southeastern Gulf of California. The striped corvina shows a wide reproductive period ranging from mid-May to August. This species was found to be a partial spawner because it shows an asynchronous gonadal development. Furthermore, the reproductive event was found to be directly related to the sea surface temperature and the photoperiod. Finally, the mean size at sexual maturity is 24.5 cm Sl for females and 23.6 cm for males. This information can be useful as a basis for approaching fishery management.











 text in
text in