The study of fungi (especially marine ones) has recently undergone development. According to the studies by Jones et al. (2015), 1,112 species have been identified (in 472 genera), out of which 805 are Ascomycota, 21 are Basidiomycota, and 26 are Chytridiomycota, in addition to other related phyla. They can produce alkaloids, lipids, enzymes, pigments, and compounds with medical applications, among others, in roles such as biotransformation, biodegradation, and bioremediation (Masís-Ramos et al., 2021). This includes marine yeasts as an alternative for phytopathogen control and disease treatment (Hernández Montiel et al., 2015), which has led to records of 138 Ascomycota and 75 Basidiomycota yeasts (Jones et al., 2015).
In particular, the diversity and distribution of endophyte fungi in plants have been studied for seagrasses in temperate and tropical areas, given the usefulness of said microorganisms in diverse applications such as the production of bioactive metabolites and bioinoculants for disease control in plants and animals (Mata and Cebrián, 2013; Abdel-Wahab et al., 2021; Ettinger et al., 2021). In the Caribbean, studies have focused on searching for bioactive compounds in endophyte fungi (Rodríguez, 2008), on comparing the fungal communities of different meadows (Mata and Cebrián, 2013), and on producing secondary metabolites within seagrass meadows with and without symptoms of the wasting disease (Castro-González et al., 2022). This last study showed a differential metabolic response in the analyzed seagrasses of the Colombian Caribbean, even at the regional level, which posed several questions: What fungi are present in the leaf tissue of T. testudinum where symptoms of the disease caused by Labyrinthula spp. Are observed? Does said mycobiota vary between seagrass beds? Hence, the objective of this research was to determine, by means of meta-taxonomic analysis, whether the diversity of the endophyte mycobiota of T. testudinum leaves from two geographically distant seagrass meadows with different anthropogenic influence (Providencia Island and the Tayrona National Natural Park, PNNT) also varied.
The seagrass samples were obtained from stems in the collection of the Makuriwa Marine Natural History Museum of Colombia, which had been collected by Invemar in two areas of a meadow of the Old Providence McBean Lagoon National Natural Park in Providence Island (81° 22’ W - 13° 20’ N), identified as samples M1 and M2, and in Chengue Bay (sample M3) of PNNT (74° 11’ W - 11° 17’ N). Epiphyte organisms were eliminated by washing the leaves of T. testudinum with distilled water before and after submerging them in HCl 0.5 % for 30 min. Then, the leaves (five for every sampling site) were pulverized in liquid nitrogen for DNA extraction using the Invisorb kit while following the manufacturer’s instructions. The amount of DNA was measured with the Quant-iT PicoGreen dsDNA kit. The DNA extracts (10 ng/µl) were sequenced via Ilumina’s MiSeq platform, based on the amplification of the ITS ribosomal locus with the ITS3F - ITS4R oligonucleotides (White et al., 1990), generating 300 pb sequences. Sequence quality and rarefaction analyses, as well as the classification of operational taxonomic units (AND) of the fungi, were carried out using the Mothur platform (version 1.39.5) and the UNITE database (version 04.02.2020), respectively. The ITS sequences were deposited in GenBank with the BioProject ID PRJNA694701.
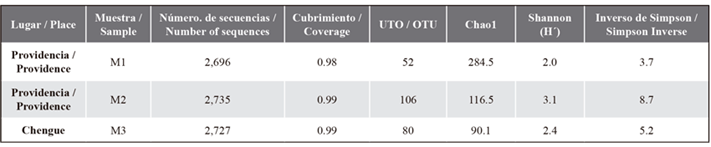
The results of the rarefaction analysis indicated that the sampling was sufficient for the samples processed in the fungal diversity analysis, with a coverage value > 98 %. A total of 8,158 sequences and 238 AND were obtained, out of which 66 % were found in meadows of Providence Island (M1 and M2) and 44 % in Chengue meadows (M3). The richness and diversity analysis showed variations between the M1 and M2 samples taken in Providence Island, with a greater proportion (Simpson) or richness and uniformity of AND (H’ Index) in the M2 sample. In addition, a greater richness of rare OTUs was observed in M1, followed by M2 and M3, according to the Chao1, as described in Table 1.
Table 1 Number and diversity of operational taxonomic units (OTUs) of T. testudinum endophyte fungi.
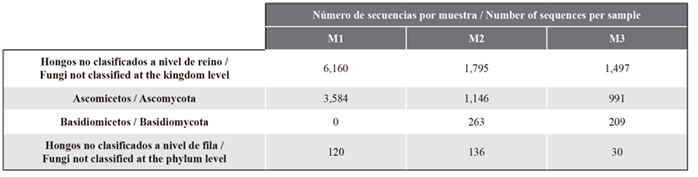
According to the taxonomic categories, the sequence classification and relative abundance analysis showed that 60 % of the total sequences could not be classified at the kingdom level. These sequences represented the highest number in each sample, suggesting high diversity and novelty, which does not allow them to be compared against sequences currently stored in public databases. In the analyzed samples, it was observed that, at the phylum level, the highest number of sequences corresponded to Ascomycota (35.9 %), followed by Basidiomycota (2.9 %) and fungi not classified at this level (1.8 %). This is mainly due to the presence of amplicon sequence variant complexes in seagrasses, which also cannot be identified given the lack of information in current databases (Ettinger and Eisen, 2020). The data showed a larger number of Ascomycota sequences in Providence seagrasses (M1 and M2) with respect to those in Chengue (M3), as well as a similar number of Basidiomycota at both sampling sites, with the exception of M1, where this group was not identified (Table 2).
Nine Ascomycota families were found, among which the most abundant was Saccharomycetales fam Incertae sedis (1,245 sequences), represented by the genus Candida and the species C. parapsilosis, followed by Saccharomycetaceae (794), represented by the genera Kazachstania, and the families Phaeosphaeriaceae (225) and Aspergillaceae (194). We detected a high number of sequences that could not be classified in this phylum (1,859). For the Basidiomycota, six families were found, with the most abundant being Sporidiobolaceae (235), 77 % of which is made up of the genus Rhodotorula, found at both sampling sites (Figure 1). A variety of genera was also found for Ascomycota and Basidiomycota endophyte fungi, with a greater richness of the latter in Providence Island (17 genera), which were mostly different from those found in Chengue (Table 3).
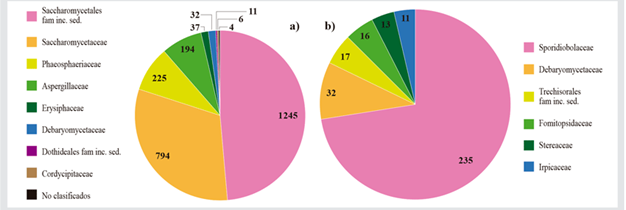
Figure 1 a) Ascomycota and b) Basidiomycota families detected in T. testudinum. The numbers correspond to the amount of sequences per family.
Table 3 Fungi genera found in seagrasses of Providence Island (M1 and M2) and Chengue (M3). Parentheses indicate the number of sequences for the most abundant genera, and those in bolt are shared between sampling areas.

With this methodology, several fungi genera were identified which complement the large amount of information on endophyte fungi that have been isolated from seagrasses in the family Hydrocharitacea by means of traditional isolation techniques (Newell and Fell, 1980; Mata Cebrián et al., 2013), including T. testudinum, which mostly correspond to Ascomycota as previously recorded (Supaphon et al., 2017). Some of them are common in the rhizomes and leaves of seagrasses such as Penicillum and Aspergillus (Rodríguez, 2008; Supaphon et al., 2013; Venkatachalam et al., 2015; Ettinger and Eisen, 2020), which have also been isolated in waters of the Colombian Caribbean (Santos-Acevedo 2018). Others, although they are plant pathogens, have been recorded in marine environments, such as Steccherinum, Bipolaris, Beauveria, Cercospora, and Alternaria (Manamgoda et al., 2014; Jones et al., 2015), are parasites of fungi (Papiliotrema), or have been recorded in estuarine palms such as Hyphoderma (Loilong et al., 2012). However, several Basidiomycetes typical of land and saprophytic environments were detected which are not reported in marine environments, as is the case of Lyophyllum, Coprinellus, Ceratobasidium, Amyloporia, Postia, Trametes, Sistotremastrum, Rynchogastrema, Steccherinum, and Mycena.
As for yeasts, several of the genera found herein, i.e., Preussia, Kazachstania, and Meyerozyma (Cheng et al., 2009; Jones et al., 2019; Singh et al., 2020), Hanseniaspora (Cheng et al., 2009; Abdel-Wahab et al., 2021), Hortaea (Ettinger et al., 2021; Abdel-Wahab et al., 2021), and several Candida species including C. parapsilopsis (Wang et al., 2008; Arora et al., 2021), in addition to Pichia and Rhodotorula (Hagler et al., 1981; Hirimuthugoda, 2006; Ettinger and Eisen, 2020), have already been recorded as ubiquitous in marine and coastal environments in water, sediments, and animals (Kutty and Philip, 2008), as well as in mangroves and/or seagrasses.
The differences between meadows regarding fungi genera suggest variations andn local environmental conditions or in the host plant’s genotype, which determine the endophyte composition (Wainwright et al., 2019). In this regard, the study by Castro-González et al. (2022) recorded differences in the nutrient concentration (ammonium and phosphorous) of the water column and in the total organic carbon and organic matter in the sediments of both meadows, which may influence the fungal community. However, it is necessary to broaden studies in order to corroborate this assertion. These results suggest that the diversity of endophyte fungi in these seagrasses might be key in their protecting role against T. testudinum pathogens such as the protist Labyrinthula spp., whose presence was evidenced in symptoms of the wasting disease for the sampled meadows, with an incidence < 15 % (Castro-González et al., 2022), even though this protist has caused ‘natural’ mortality in seagrasses of different coastal areas around the world (Garcias-Bonet et al., 2011; Burge et al., 2014).
However, in the near future, it is necessary to extend research to the microbiome associated with seagrass meadows and evaluate whether it varies between healthy meadows and those affected by disease, as well as its possible role in plants’ defense system against pathogens and/or physicochemical changes in waters of the Colombian Caribbean on different spatial-temporal scales, as recent studies indicate that the composition of the phylloshpere’s microbiome varies with both the incidence of disease and geographical distance, as well as with temperature to a lesser extent (Beatty et al., 2022). On the other hand, considering the previous information on these organisms, their potential is fundamental, especially for countries with high marine biodiversity, as is the case of Colombia, regarding their development in the field of biotechnology for useful products and processes. This is why delving into the understanding of their bioprospection constitutes the foundation of scientific and technological research in the short term.











 text in
text in