Introduction
The perovskite Sr2SbMnO6 (SSMO), which belongs to the double perovskite family (Ortiz-Díaz, 2007) was synthesized and characterized by Ivanov (2009) (Cheah, et al ., 2006), who reported it as a ferroelectric material in the tetragonal phase space group (s. g.) I4/m. Majhi, et al. (2007) studied the structure of the polycrystalline ceramic SSM synthesized at room temperature. They found that this material crystallizes in a pure phase belonging to the I4mm space group. The material undergoes a structural phase transition from I4mm to Pm3m space group at about 725 K (Fesenko, 1972), and a second phase transition from ferroelectric in I4mm space group to paraelectric behaviour in the I4/mmm space group at 431 K (Foster, et al. , 1997; Politova, et al. , 1990).
Cheah, et al. (2006) showed that this perovskite had a tetragonal structure with I4/m space group and changed to a cubic symmetry above 550 ºC (space group Fm-3m). Furthermore, it has been reported that the crystal structure is tetragonal within the temperature range of 2 to 750 K (s. g. I4/m) and cubic above 750 K (s. g. Fm-3m) (Baral & Varma, 2009; Ivanov, et al ., 2009).
In the present work we report the synthesis and structural characterization of the Sr2SbMnO6 material using SrO instead of the commonly used SrCO3. With this sintering process we established that the suitable precursor for perovskite production was strontium oxide, as no impure phases were identified in the carbonate diffractograms. The X-ray diffraction (XRD) pattern showed that the experimental diffractograms fitted with three reported space groups. Therefore, there is need for a careful analysis of the structural arrangement of material at different temperatures, as well as refining each group, to clarify the real crystallization structure using the GSAS code for the Rietveld refinement. The analysis showed that for these three possible structural groups R [F2] and χ2 values were very similar, but the best fit of the primitive tetragonal structure was for P4/mnc space group (Figure 1).
The aim of this work is to report results of XRD studies of the SSMO complex perovskite at different temperatures, with careful refinements of experimental patterns, which were carried out by using the GSAS code, using a variety of models for space groups with the same extinction conditions.
Materials and methods
Polycristalline samples of Sr2SbMnO6 ceramic powder were prepared through the conventional solid-state reaction route. Stoichiometric amounts of SrO (99.9%), Mn2O3 (99.999%) and Sb2O3 (99.999%) were mixed and grinding in an agate mortar for 2 hours. Then, the mixture powder was pressed under 2.5 ton/cm2 to form pellets of 1 cm diameter. The pellets were then calcined at room temperature until reaching 1200 ºC for 10 hours. This temperature was maintained for 100 hours, and then reduced to 20 ºC within 12 hours. The evolution of the structural ordering of Sr2SbMnO6 was monitored with X-ray powder diffraction (XRD) using a X´Pert PRO PANalitycal diffractometer with CuKα radiation. Data were collected over the range of 15º < 2θ < 80º. The patterns of the thermal evolution were registered on a Phillips PW1710 diffractometer in the range between 25 and 1400 ºC. The XRD patterns were analyzed by means of the Rietveld refinement using the GSAS & EXPGUI software.
Results and discussion
We produced samples by three calcination processes at 1200 ºC for 100 hours, 1300 ºC for 60 hours and 1400 ºC for 60 hours. The structural characterization of each of the samples calcined by the Rietveld refinement method showed that the best temperature to produce the sample was at 1200 ºC. The diffraction pattern showed a better fit with space group P4/mnc. The structural parameters obtained in the Rietveld refinements for the three possible space groups are shown in Table 1.
Table 1 Comparison of the results obtained by the Rietveld refinement for three possible space groups to describe the Sr2SbMnO6 structure at room temperature using X-ray diffraction data.

The analysis of the Rietveld refinement allowed us to conclude that the ideal production method for the ceramic SSMO was the calcination at 1200 ºC resulting in a tetragonal crystal structure in the P4/mnc space group (# 128).
The Rietveld refinement for the X-ray diffraction pattern, recorded at room temperature with a tetragonal structure P4/ mnc, showed that the SSMO samples formed a single phase (Figure 2). Due to the various conflicting reports regarding the space group symmetry, we refined XRD data using a variety of models for space groups with the same extinction conditions, and we found that the diffraction pattern observed may be described in the P4/mnc model. Assuming that the assignment symmetry and structure determination were correct, we performed a structural analysis of the sample for a thermal variation in the temperature range between 25 and 1400 ºC.

Figure 2 X-ray diffraction pattern for Sr2SbMnO6 ceramics after Rietveld refinement of the crystal structure at room temperature.
Figure 3 shows the X-ray patterns of the Sr2SbMnO6 sample as a function of temperature, in a range from 100 to 1000 ºC. The figure shows that as temperature increased, several Bragg peaks began to overlap; the systematic absence of these reflection peaks is indicative of a cubic space group Fm-3m. All patterns obtained between 800 and 1000 ºC were refined in the cubic space group.
The Rietveld refinement of the XRD data from 100 to 600 ºC with space group P4/mnc gave the best fit. The best quality of fit for the tetragonal structure P4/mnc was obtained at a temperature of 600 ºC (Figure 4). At this temperature we observed that the P4/mnc structure was maintained.
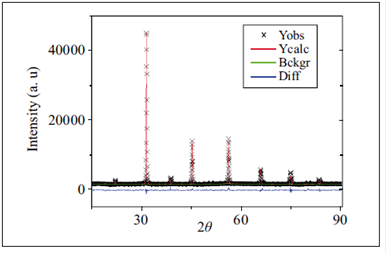
Figure 4 Observed, calculated and difference plots of the X-ray diffraction pattern for Sr2SbMnO6 after Rietveld refinement at 600 ºC.
In Figures 2, 3 and 4 it is possible to observe how from 700 ºC on the structure lost its monophasic character due to the structural transition of the Sr2SbMnO6 system. Before reaching 700 ºC, the system obtained using Rietveld refinement appeared clearly tetragonal (Figures 3 and 4).
Figure 5 shows the cubic character of the Sr2SbMnO6 system wherein the diffraction peaks become simple peaks. The results at 900 and 1000 ºC exhibit the same behavior. The data corresponding to 700 ºC are not ideal, as the structure began the transition phase at this temperature.
Table 2 shows the structural parameters obtained with the Rietveld refinements at 600 ºC (tetragonal structure) and 800 ºC (cubic structure).
Table 2 Lattice parameters, atomic positions and bond distances of Sr2SbMnO6 obtained by the Rietveld refinement of X ray diffraction at different temperatures. In P4/mnc SG, Sr occupied 4d(0 ½ ¼), Sb occupied 2a(0 0 0 0) and Mn occupied 2b(0 0 ½). In Fm - 3m SG, Sr occupied 8c(¼¼¼), Sb occupied 4a, and Mn occupied 4b. Agreement parameters are also included.

For temperatures above 1000 ºC the system exhibited diffraction peaks showing how it gradually lost the monophasic cubic structure (Figure 6). In this temperature range the system lost the unique phase, and other phases were observed (arrows).

Figure 6 Diffractograms obtained for the temperature range from 1100 to 1400 ºC. The arrows show the presence of other structural phases.
Figure 7 shows that as temperature increased a progressive reduction of the tetragonal structure has place, and above 700 ºC the material turned cubic. The values of lattice parameters were amplified for 20.5 in order to simplify the comparison. A rapid change in the lattice parameter was evident with increasing temperature.
Conclusions
Our results showed a space group at room temperature and a transition temperature different from those reported in other studies. Diffraction patterns for the thermal evolution of the sample showed a tetragonal structure with space group P4/mnc above 600 ºC, and a cubic structure from 800 to 1000 ºC with space group Fm-3m. Rietveld refinement results for these two temperatures evidenced that at 600 ºC, χ2 = 1.455 with R [F2] = 0.0854, and at 800º C, χ2 = 1.437 and R [F2] = 0.091. The results of the structural analysis of the diffractograms of the samples differ from the crystal structure and the transition temperature of the Sr2SbMnO6 system proposed by other authors (Yamagata, et al ., 2017; Yamagata, Inoue, & Koyama, 2017).


















