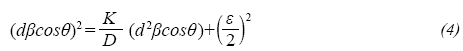1. INTRODUCTION
Air pollution is defined as the presence of substances in the atmosphere at a level high enough to affect human, animal, plant or microbial health. (Li, Lin, Liu, Guo, Ou, & Chen, 2015). Even though biofuels represent an opportunity to reduce contaminant emissions, the concentrations in which they are being used as blends with fossil fuels (B5, B8 and B10) are still too low to avoid the release of high amounts of pollutants to the atmosphere. Furthermore, there are some drawbacks regarding the use of biodiesel due to its lower heating value, and higher density, viscosity, fuel consumption, and NOX emissions compared with traditional fuels. Thus, the inclusion of additives in fuel blends becomes an essential topic in order to minimize these drawbacks (Rashedul, et al, 2014).
As a way to respond to the demand for higher quality fuels, researchers have been focused on finding substances that could improve the physicochemical properties of fuel blends and help reduce contaminant emissions. A number of additives in the form of metals and organic compounds, such as diglycol methyl ether, molybdenum, magnesium and poly-isobutylene have been reported in scientific literature (Shaafi, Sairam, Gopinath, Kumaresan, & Velraj, 2015) (Rashedul, et al., 2014). Additives are being used with biodiesel blends since they can counteract the typical issues commonly associated with biofuels. For instance, most of the oxygenated additives show higher brake thermal efficiency in blended biodiesel and decrease the brake specific fuel consumption, as compared to diesel fuel and biodiesel without additives. Moreover, the addition of metallic additives decreases the specific fuel consumption and improves the exhaust gas emissions (Rashedul, et al., 2014); (Shaafi & Velraj, 2015); (Mirzajanzadeh, et al, 2015). These additives can be obtained from organic and inorganic compounds, and can be modified with metals.
Compounds that are based on metals such as copper, iron, cerium and cobalt have been widely used as fuel additives. Considerable improvements in enthalpy and combustion quality have been observed when using this kind of materials (Shaafi, T., Sairam, K., Gopinath, A., Kumaresan, G., & Velraj, R., 2015). Increases in the total energy density of the fuel have also been achieved, which leads a reduction in storage volume.
However, since many of the energetic additives are at the micrometre-scale, several secondary effects could occur during combustion. These include ignition delay, low combustion rate, and incomplete combustion leading to the formation of metallic micro particles (Venkatezwarlu, et al, 2012; Shaafi et al., 2015).
Recent advancements in the development of nano-scale materials with innovative properties (electrical, chemical, physical, among others) have made them become highly promising tools that could improve many characteristics in fuels nowadays. Jones, Li, Afjeh, & Peterson, (2011) studied the application of alumina nanoparticles as hydrocarbon blend additive. They observed an increase in the combustion efficiency of biofuels like bioethanol, along with a higher heat emission when compared to the standard fuel (Jones, M., Li, C., Afjeh, A., & Peterson, G., 2011). Additional work conducted by Selvan, Anand, & Udayakumar (2009), focused on the application of Cerium Oxide (CeO2) nanoparticles-based additives. The authors indicated that the material was able to act as an oxygen donor catalyst in combustion systems, allowing the complete oxidation of carbon monoxide into carbon dioxide. They reported that nanoparticles could also reduce NOx compounds by oxygen adsorption. Furthermore, deposits of accumulated carbon in combustion chambers experienced burning stages that were attributed to the use of cerium oxide nanoparticles. This helped to reduce wear in moving parts of the engine and avoid the deposition of non-polar compounds into the combustion chamber (Selvan, M., Anand, R., & Udayakumar, M., 2009).
Many recent studies have been reported in the literature focusing on the use of nanomaterials as additives for hydrocarbon blends (Gan, Lim, & Qiao, 2012; Shaafi et al, 2015; Prabu & Anand, 2015). However, most of them do not specify which surfactant molecules are required to prepare nanoparticles suspensions in the fuel medium. There is no indication about the effect that the colloidal stability of these suspensions exerts on the physicochemical properties of blends either. Hence, this work presents a total interaction energy analysis for oleic acid-coated alumina nanoparticles suspended in fuel blends B10 (Diesel- Palm Oil Biodiesel), in order to verify the colloidal stability of this system and the effect of this nanostructured based additive on properties such as kinematic viscosity and flash point.
2. EXPERIMENTAL DEVELOPMENT
Synthesis and characterization of oleic acid-coated alumina nanoparticles
Alumina nanoparticles were synthesized by sol-gel method using a 1M solution of nonahydrate aluminium nitrate Al(N03)3-9H20 in distilled water (Li, Pan, Xiang, Ge, & Guo, 2006). In a typical procedure, 200 mL of 1M citric acid were added to 200 mL of 1M Aluminium Nitrate solution. The reaction was carried out at 60°C for 2 hours and it was later heated up to 80°C for 1.5 hours, which led to the formation of a gel. Afterwards, this gel was dried at 200°C for 2 hours.
The synthesized nanoparticles were submitted to thermal treatments at 750 and 1000 °C to facilitate the formation of the α-Al203 phase, which presents a higher thermodynamic stability (Cava, et al., 2007). Size and crystalline structure of the alumina nanoparticles were determined by X-ray diffraction analysis using an X-Pert ProAnalytical diffractometer.
The physicochemical properties of the B10 (Diesel-Palm Oil Biodiesel) blend were determined as a function of the nano-additive concentration. For this purpose, alumina nanoparticles were coated with oleic acid to improve their dispersion in the fuel system. The oleic acid was selected as a surfactant because of its chemical affinity with the organic components in the fuel and the presence of a carboxylic group in its structure (Lee & Harris, 2006). This functional group can be coordinated on the nanoparticles surface during the modification stage. The concentrations of the nano-additives in the fuel blend were fixed at 10 and 20 ppm according to literature related to applications of nano-additives in biodiesel (Sajith, V., Sobhan, C, & Peterson, G, 2010; Prabu & Anand, 2015). For a typical preparation, the alumina nanoparticles (8 mg or 16 mg) were mixed with 20 mL of oleic acid. Then, this mixture was heated at 80°C under constant agitation for 1 hour, and it was subsequently submitted to ultrasound for 30 minutes. This procedure was repeated by adding 20 mL of oleic acid to the suspension of nanoparticles until it reached a total volume of 100 mL. Then, B10 fuel was added to nanoparticles suspension up to reach the selected concentrations.
Colloidal stability model
Colloidal stability of alumina nanoparticles coated with oleic acid and suspended in an organic medium was evaluated using the Deryaguin-Landau-Verwey-Oberbeek (DLVO) model. For this purpose, energetic interaction forces that govern the system, such as attraction forces (van der Waals interaction) and repulsive forces (osmotic and elastic) were estimated. The contribution of these forces was determined regarding potential energy experienced by two identical spherical particles as a function of their separation distance by Barrera, C, Herrera, A., & Rinaldi, C, (2009), (Park, Huang, Corti, & Franses, 2010).
For this analysis, the Van der Waals' attraction energy is expressed as a function that depends on the radius of the nanoparticles, the chain length of the surfactant molecule (Oleic Acid, 1.5 nm; Yang, Peng, Wen, & Li, 2010), and the Hamaker constants (Barrera etal, 2009), mathematical expressions and symbols are explained in the nomenclature table at the end of this paper.:
For this model, the organic solvent Dodecane was selected as the fuel oil solvent media, with a Hamaker constant of 4.35x 10-20 J (Van Oss, Chaudhury, & Good, 1988). Oleic acid acts as surfactant molecule, with a Hamaker constant value of 4.38x 10-20 J (Van Oss, C, Chaudhury, M., & Good, R., 1988). A value of 14.8x 10-20 J for the Hamaker constant was used for alumina nanoparticles (Bergström, 1997).
The steric repulsion energy is obtained from the sum of osmotic and elastic forces. Osmotic forces depend on the free energy of the solvent-ligand interactions because of the ligand molecules solvation, while the elastic forces are the result of entropy lost due to compression between the ligand chains and the surface of adjacent particles (Park, Y., Huang, R., Corti, D., & Franses, E., 2010). Osmotic repulsion and elastic force can be estimated as follows (Barrera et al, 2009), (Herrera, Resto, Briano & Rinaldi, 2005)
Based on the mentioned model, a total interaction energy analysis was developed to evaluate the feasibility of the suspension process of alumina nanoparticles in the organic B10 media.
Estimation of kinematic viscosity and flash point
Physicochemical properties, such as kinematic viscosity and flash point for fuel blends with alumina nanoparticles (10 and 20 ppm) were determined according to ASTM D445 and ASTM D93 regulations. In addition, a sample of fuel without additives (0 ppm) was characterized to determine the effect of alumina nanoparticles on the selected properties of BIO fuel blends.
3. RESULTS AND ANALYSIS
Size and crystal structure of the alumina nanoparticles
Powder X-ray diffraction is a powerful analytical technique widely used to study the structure, crystallite size, and morphology of nanoscale materials (Withfield & Mitchell, 2004). It is based on the principle that the wavelength of X-rays is comparable to the distances between atoms in condensed matter (Giannini et al., 2016). It is a simple and non-destructive technique used to determine the physicochemical properties of nanomaterials and their composites with minimum invasion (Ingham, 2015). The accuracy, relevance, sensitivity and availability of X-ray diffraction increase its roles in several fields, such as geology, pharmaceuticals, polymers, medicine, nanotechnology, among others (Das, Ali & Hamid, 2014). Figure 1 shows the X-ray diffraction patterns of the alumina nanoparticles after calcined at 750°C and 1000°C. It was observed that the sample that was calcined at 75 0°C showed the presence of both γ -Al203 and α -A/203 phases, with γ -Al203 at a smaller proportion. This is in agreement with the results reported by Cava et al., (2007), which indicated that at this temperature it's not possible to achieve a complete arrangement of the crystal lattices. After calcination at 1000°C, only peaks corresponding to α-Al203 phase were observed, suggesting a total transition of the γ -Al203 phase in the alumina sample and the presence of a hexagonal structure, which are characteristics of alumina nanoparticles (Feret, Roy, & Boulanger, 2010).
The average crystal size of the alumina nanoparticles was calculated from the X-ray diffraction patterns and the size-strain plot method (SSP) (Khorrami, Zak, & Kompany, 2012). The size-strain plot method distinguishes the width of the peaks that are induced by particle size and stresses in the crystal lattice, which are caused by distortions and imperfections in the crystal (Khorrami, Zak, & Kompany, 2012). This method works under the assumption that the size-induced widths (βD) and the stress-induced widths (βS= 4εtanθ) are additive parameters of a total peak width β. The SSP equation is given by (Khorrami, G, Zak, K., & Kompany, A., 2012):
where d corresponds to the distance (in nanometers) between two adjacent planes, K is a constant that depends on the shape of the particles (a value of 0.75 is used for spherical particles) and ε is a measure of deformation of the lattice parameters due to crystal defects such as vacant, grain boundaries, crystal dislocations, among others.
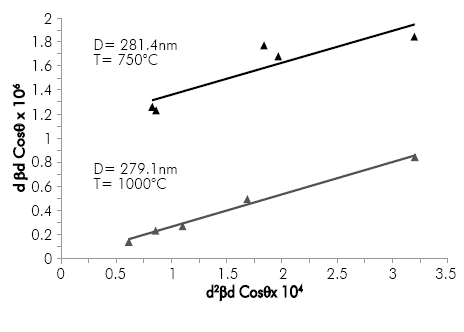
The terms (d2(βcosθ) and (d(βcosθ)2 in equation 4 correspond to the abscissa and ordinate of a straight line, respectively. Figure 2 shows the fitting line obtained after plotting these terms from the diffraction patterns that were obtained for alumina nanoparticles calcined at 750 and 1000°C. From this analysis, it was estimated an average crystal size of about 280 nm, which was not dependent on calcination temperature. This result compares very well with the value estimated by Mirjalili, Mohamad & Chuah, 2011, which determined an average crystal size ranging from 250 to 300 nm for α-Al203 nanoparticles synthesized by the sol-gel method (Mirjalili, F, Mohamad, H., & Chuah, L., 2011).
Colloidal stability analysis
The total interaction energy between two oleic acid-coated alumina nanoparticles that are suspended in a diesel/biodiesel blend was modelled as the sum of the interactions forces that control the stability of the system: attraction forces (equation 1) and repulsion forces (equation 2 and 3). The advantage of an additive that is based on fatty acids relies on the fact that they promote modification of the nanoparticles surface, thus allowing their suspension in an organic solvent (Khalil, Yu, Liu, & Lee, 2014). Figure 3 shows the total interaction curve that was obtained for alumina nanoparticles with a diameter of 280 nm coated with oleic acid and suspended in the organic media at 25 °C. A local minimum can be observed at a separation distance of approximately 3 nm; it is followed by a notorious increment in steric stabilization forces. This behaviour can be attributed to overlapping of oleic acid chains that are adhered to the surface of the particles, which prevents their agglomeration and causes repulsions with stabilizing tendencies in the fuel blend (Barrera etal, 2009).
Figure 4 shows the results obtained for the modelling of the total interaction energy between nanoparticles sizes ranging from 20 to 300 nm. This energy was compared to the minimum value of the system (-1.5kBT), with the purpose of determining the limit at which spontaneous agglomeration might occur as a result of solvent agitation. This analysis considers that if the minimum value of the total interaction curve is greater than or equal to Boltzman's stabilization energy limit (-1.5kBT), the particles will have a higher energy that will allow their dispersion in the organic medium. If the opposite happens, and the value is lower than Boltzman's stabilization energy limit, nanoparticles will precipitate (Barrera et al., 2009), (Park et al., 2010). From this analysis, and according to Figure 4, a minimum nanoparticle size of 60 nm is required to obtain a completely stable colloid. This suggests that systems with alumina nanoparticles sizes that are greater than 60 nm will produce colloids with limited stability and a tendency to coalescence. This low stability was displayed for the synthesized alumina nanoparticles in the form of precipitation after a few hours of being coated with the oleic acid and suspended in the fuel blend at a concentration higher than 100 ppm.
Despite the fact that the oleic acid molecule has suitable features as a surfactant agent for suspension of nanoparticles in organic media, the shortness of its chain (1.5 nm) does not provide enough steric repulsion to keep a high concentration of alumina nanoparticles suspended in the fuel during long periods of time. Thus, for the preparation of the fuel blends with nano-additives, low concentrations of 10 and 20 ppm were selected to ensure the availability of these nanoparticles during the determination of the kinematic viscosity and flash point properties of the biofuel media, since no nanoparticle precipitation was observed after 24 h of addition of alumina nanoparticles to the B10 fuel mixture at these settled concentrations.
Physicochemical properties
In this work, physicochemical properties such as kinematic viscosity and flash point were evaluated for palm oil biodiesel / regular diesel blends in order to establish the effect in these properties due to a nanostructured based additive. The kinematic viscosity and flash point were determined according to ASTM D-445 and ASTM D-93 standards. Table 1 presents the obtained results for the physicochemical tests of fuel blends with and without additives.
These results show a significant change in viscosity when increasing the additive concentration in the fuel blend. On the other hand, there is no substantial change in flash point.
Table 1.Results of the physicochemical tests for fuel blends with and without additives

* ASTM D-445 requires a maximum deviation of 0.11%
** Legal values regulated by the Colombian Government regarding commercialization of fuel blends for diesel engines. Resolution 182087 from 2007.
According to Table 1, a change in the kinematic viscosity of about 10% was observed, which can be attributed to the addition of oleic acid and alumina nanoparticles. It is important to note that the volume of oleic acid was constant for blends preparation. On the other hand, the amount of alumina nanoparticles was increased from 8 to 16 mg, thus, the difference in the kinematic viscosity values at 10 and 20 ppm could be attained to the presence of alumina nanoparticles.
According to ASTM D6751, the acceptable range for biodiesel viscosity is 1.9-6.0 mm2/s. The viscosity of biodiesel blends out of the standard regulations can affect the performance of engines. Low viscosities do not provide sufficient lubrication, while high viscosities can generate deposits resulting in poor combustion and increasing emissions of greenhouse gases (Nwadike, Yahaya, O'Donnell, Demshemino, & Okoro, 2013). Thus, the kinematic viscosity is an important parameter required in the study of biodiesel production processes (Tat & Van Gerpen 1999).
Table 2 presents a comparison with other studies that have reported the use of nanostructured additives for fuel blend applications. So, Table 2 shows that the use of fatty acids (oleic and abietic) as stabilizing agents produce greater changes in the kinematic viscosity of fuel blends. This can be attained to the viscous nature of these organic molecules, as a viscosity of 13.2 cP has been reported for oleic acid at 49°C (Noureddini, Teoch & Clements, 1992). It is important to note that the amount of oleic acid used in this research for the additive preparation did not represent any kind of drawback for the legal values of the kinematic viscosity allowed for B10 fuel blends. Nevertheless, the observed increase of this property could represent a beneficial effect for engines, since low viscosity fuels result in wearing and power loss in some injection pumps, because of their insufficient lubricant capacity.
Table 2 Comparative analysis of physicochemical properties of fuels blends after application of nanostructured additives

** Equivalent concentration of particles in the fuel blend.
Regarding the flash point property, an increase of 1°C was observed after adding nanoparticles at concentrations of 10 and 20 ppm to the B10 fuel blend, indicating no significant effect in the concentration of alumina nanoparticles used in this work. This response is similar to the one that was reported by Sajith, Sobhan, & Peterson, (2010) when using a nanoparticle concentration of 20 ppm cerium oxide (Sajith et al, 2010). These variations can be attributed to the use of surfactant agents during modification stage. Such molecules (like oleic acid) are capable of modifying the medium properties and promoting phase contact (nanoparticles-oleic acid- biodiesel), thus increasing the attraction forces between the particles and reducing the flow from liquid to gas phase. It should be noted that fuels with high enough flash point are easy to handle when it comes to transportation and storage (Sajith et al, 2010).
4. CONCLUSIONS
The alumina nanoparticles that were synthesized by sol-gel method exhibited particles size of approximately 280 nm (compact hexagonal structure) according to X-ray diffraction analysis and SSP method. Because of its large size, the oleic acid molecule that is used as part of the additive was not the most suitable surfactant agent to suspend the nanoparticles in the fuel blends, as it was observed in the colloidal stability analysis. The use of additional stabilizing agents with properties that could offer a better suspension of nanoparticles in the additive (affinity with the fuel, chain length, etc.) is therefore recommended. Blend formulations with higher concentrations of nanoparticles could be achieved in this way. These types of colloidal stability studies are important for those researches that involve systems with lyophobic additives, as a tool of previous evaluation and feasibility of such systems. It is important to highlight that reported previous publications regarding the use of nano-additives in fuels did not perform any prior analysis of colloidal stability for the nanoparticles. Therefore, this paper evidences how necessary it is to evaluate the use of some fuel systems in engines, which could guarantee their continuous effect during the entire process.
In addition, it was observed that the obtained physicochemical properties (flash point and viscosity) are within the quality requirement range for diesel fuel and its blends with biofuels in order to be used for engines. Regarding the influence of alumina nanoparticles in the physicochemical properties of blends it was more substantial for the kinematic viscosity. Therefore, it can be expected that higher alumina nanoparticles concentrations could show a significant effect in these properties. The results showed that application of nano-additives in biodiesel blends offers a potential opportunity to improve physicochemical properties in fuel blends, however long chain surfactants need to be evaluated to guarantee a higher colloidal stability for future application in industrial process.