Introduction
Sepsis is a clinical syndrome currently defined in the Sepsis-3 consensus1 as “a life-threatening organ dysfunction caused by a dysregulated host response to infection”, moreover its mortality varies from 20% to 50%2.
According to the World Health Organization Bulletin 2019, sepsis affects more than 30 million people each year, causing about 6 million deaths 2,3. In economic terms, in the United States, sepsis and septic shock are reported to cost $16,000 and $38,000 annually, respectively, resulting in 13% of the hospital budget4. In Ecuador, the picture is no different from global reality5.
The severity and mortality of sepsis can be predicted and estimated with validated scales, such as the SOFA and Lo gistic System of Organic Dysfunction (LODS)5,6; or by biologi cal markers such as procalcitonin7, interleukin 68-9, C-reactive protein10, among others. The infrastructure required and the high economic cost make many centers unable to use these tools routinely. It has motivated the search for simple and accessible alternatives; one of them is the comprehensive blood biometry, whose cell lines express modifications that could estimate the severity of the pathology, and generate therapeutic approaches that modify the course of the disea se11.
The systemic inflammatory impact on sepsis affects all he matological cell lines. The red series experiences a decrease in hematocrit, hemoglobin, and global infection, the erythro cytes maintain a constant volume, but morphologically they become spherical and rigid affecting their capacity for de formation and microcirculatory functionality. The erythrocyte distribution width (EDW) is a parameter of anisocytosis that tends to increase in sepsis, these changes have been detec ted in the first six hours of evolution according to experimen tal models, and have shown an association with mortality12.
The mean platelet volume, which reflects platelet activity, has been associated with unfavorable outcomes in several studies, such as mortality in sepsis, suggesting that platelets represent therapeutic targets in sepsis by combining their mechanisms of inflammation and coagulation13.
Other markers are the indices neutrophil/lymphocyte, mono cyte/lymphocyte, platelet/lymphocyte, mean platelet volu me/platelet count, which have shown diverse results, howe ver, their values tend to increase in non-survivors, and some of them have shown to be prognostic markers of mortality14).
Based on this premise, this study aimed to determine the uti lity of different cytometric parameters and indices as predic tors of mortality in septic patients.
Materials and methods
General description of the study
The study design consisted on a retrospective cohort. It was a census of all patients with sepsis from different infectious si tes, who were admitted to the Intensive Care Unit of the Pablo Arturo Suárez Hospital in Quito (Ecuador) over a 2-year period.
Selection criteria
We included adult patients (older than 18 years of age) diag nosed with sepsis using the SEPSIS criteria 3 (1). The sites of infection were abdominal, lung, urinary, soft tissue, central nervous system. We excluded patients with neoplasms or previous immunodeficiency states.
Variables
On admission, 48 and 72 hours of hospitalization in the Inten sive Care Unit, the cytometric parameters of the hemograms were recorded. These were erythrocyte distribution width, platelet mean volume, platelet count, neutrophil count, lym phocyte count, neutrophil/lymphocyte index (division bet ween the absolute number of neutrophils and the absolute number of lymphocytes) and MPV/platelet ratio (a division of platelet mean volume and the absolute number of platelets). The hemograms were performed by impedance in a Mindray BC-6800 equipment. We collected other parameters: lactate in mmol/L, central venous oxygen saturation in %, and pro calcitonin in ng/ml. Finally, the days of hospitalization in the Intensive Care Unit and the status of the discharge as alive or deceased were recorded.
Statistical Analysis
The analyses were carried out with the IBM SPSS version 25 statistical package, for which descriptive statistics were used, using tables and representing the absolute and relative va lues of the qualitative variables, as well as measures of central tendency and variability for the quantitative variables.
By analyzing the ROC curve, the cut-off point for the para meters of blood cytometry was determined; the areas under the ROC curve, sensitivity, specificity, PPV and NPV, and Odds Ratio (OR) were determined.
Bivariate analysis was performed to compare the clinical cha racteristics and blood cytometry parameters with the dischar ge condition of patients with sepsis. For categorical variables, the Chi-square test was applied for quantitative variables as they did not present normality, and the Mann-Whitney test of independent samples was used to compare means.
Multivariate logistic regression analysis with Wald’s test was used to determine the predictors of mortality in patients with sepsis using the cut-off points of blood cytometry parameters.
Statistical significance for comparing ratios and means was es tablished for p-value <0.05; the Odds Ratio was considered significant, observing the limits of the 95% confidence interval.
Ethical aspects: Secondary data from the hemograms were routine, so it was not necessary to apply informed consent.
Results
We analyzed 159 patients with sepsis, of which 25.16% (40 patients) had died at discharge; the average age of all pa tients was 59.47 years; regarding the location of infection, there are no significant differences between the groups (p=0.496); SOFA presented significant differences between non-survivors and survivors with p-value 0.000. On the other hand, differences were observed in the initial APACHE II va lues with a p-value of 0.000 (Table 1).
Table 1 Clinical characteristics of patients with sepsis by discharge condition.

SD: Standard deviation.
SOFA: Sequential organ failure assessment.
APACHE: Acute physiology and chronic health evaluation II.
With regard to blood cytometry parameters, significant diffe rences were observed for the erythrocyte distribution width (EDW), the MPV, platelets, and the MPV/ platelet ratio with p-values <0.05 when compared to the discharge condition of patients with sepsis. Baseline MPV, platelets/mm3, MPV/ platelets, neutrophils/mm3, lymphocytes/mm3, and other pa rameters are different between the groups. (Table 2)
Table 2 Comparison of blood cytometry parameters of patients with sepsis by discharge condition.

MPV: Mean platelet volume.
SD: Standard deviation.
EDW: Erythrocyte ditribution width.
For the blood cytometry parameters that presented statisti cal significance in the bivariate analysis with the exit condi tion, mortality cut-off points were determined using the ROC curve (Figure 1)
The estimate of the area under the curve was significant in predicting mortality with p-values <0.05, for EDW at baseli ne (0.708) and 48 (0.696) hours, WMV at baseline (0.666), 48 (0.699) and 72 (0.728) hours, and for the WMV/platelet ratio at 48 (0.651) hours. (Table 3)
Table 3 Test for the area of the ROC curve to predict mortality in patients with sepsis according to blood cytometry parameters
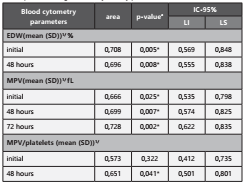
EDW: Erythrocyte distribution width. MPM : Mean platelet volume.
SD: Standard deviation.
The cut-off points were determined on the ROC curve using the Youden index; for the EDW at baseline and at 48 hours the cut-off point was ≥14,50%, where the sensitivity was 62.50% at baseline and 69.57% at 48 hours, specificity 61.34% at ba seline and 58.93% at 48 hours, PPV 32.21% at baseline and 25, 81% at 48 hours, NPV 82.95% at baseline and 90.41% at 48 hours, plus patients with baseline or 48-hour EDW ≥14,50% are 2.65 and 3.68 times more likely to fail to survi ve than those with <14.50% values. The MPV at baseline, 48 and 72 hours presented a cut-off point of ≥8,45fL, where the sensitivity was 67.50% at baseline, 78.26% at 48 hours and 72.22% at 72 hours, specificity 65.55% at baseline, 57.14% at 48 hours and 59.22% at 72 hours, PPV 39.71% at baseline, 27, 27% at 48 hours and 23.64% at 72 hours, NPV 85.71% at baseline, 92.75% at 48 hours and 92.42% at 72 hours, plus patients with baseline, 48 or 72 hour MPV ≥8,45fL are 3.95, 4.80 and 3.78 times more likely to not survive than those with <8.45 fL values. Finally, the ratio WMP/platelet presented a cut-off point at ≥4,75, with sensitivity of 60.87%, specificity 64.29%, PPV 25.93%, NPV 88.89%, where patients with WMP/ platelet ≥4,75 are 2.80 times more likely to not survive than those with values <4.75. (Table 4)
Table 4 Statistical predictors of mortality in patients with sepsis by cut-off points of blood cytometry parameters.

PPV: Positive predictive value. NPV: Negative predictive value.
OR: Odds ratio. CI: Confidence interval.
MPV: Mean platelet volume. EDW: Erythrocyte distribution width.
The multivariate logistic regression model was used to deter mine the relationships with mortality using the cut-off points of the blood cytometry parameters. The results obtained showed that the initial EDW with p-value 0.005 and the initial MPV with p-value 0.004 are predictors of mortality, where for initial EDW ≥14,5, the probability of not surviving was increa sed by 5.25 times, while for MPV ≥8,45 fL the probability of not surviving was increased by 5,28 times. (Table 5)
Discussion
In our study, we evaluated the predictive usefulness of cyto metric indicators for mortality in critically ill patients with sepsis; EDW, MPV, platelet count, MPV/platelet ratio were as sociated with mortality, while neutrophil count, lymphocyte count, and neutrophil/lymphocyte ratio were not significant. In the multivariate model with logistic regression, the mea surement of EDW and MPV at admission were independent predictors of mortality, i.e., an EDW value greater than or equal to 14.5% and an MPV greater than 8.45 fL, are 5.25 and 5.28 times more likely, respectively, to not survive.
The variability in the size of erythrocytes or anisocytosis is assessed by the EDW; its normal ranges are 12 to 13%. The causal relationship of increased EDW is not well understood, but it is a prognosis of clinical severity and death in different acute and chronic diseases, sepsis among them11.
The MPV is a marker of severity in sepsis; when proinflam matory cytokines are increased, platelets change morpholo gically from discoid to spherical with pseudopodia, increase their size15 and can express receptors for immunoglobulin G (IgG), that is to say, they participate in adaptive immunity16 and Toll type for molecular pattern recognition to pathogens (PAMPs) (11. Young platelets are larger than old platelets, are more metabolic and enzymatic functional, and have a greater prothrombotic effect due to increased synthesis of throm boxane A2, p-selectin, and glycoprotein IIIa.17,26 Nishimura et al. (18 identified poor prognosis when platelets are young and large, due to a lower organization of their microtubules.
Regarding the EDW, several studies have determined the as sociation and prediction of mortality. Wang et al.19 in 602 cri tical patients with a cut-off point of 14.8%; Cavusoglu et al.20 in the United States with data collected from critical patients with sepsis in 10 years with a cut-off point of 14.4%; Braun et al.21 in community pneumonia with a cut-off point of 14.5%, and finally Hunziker et al. (22, in 17922 critically ill patients de monstrated that it is an independent predictor of mortality and by associating it with the Simplified Acute Physiology Score (SPAS II), it improved its prognostic performance and stratification capacity, bringing the area under the curve (AUC) from 0.746 to 0.774 (p < 0.001) for hospital mortality and 0.793 to 0.805 (p < 0.001) for ICU mortality. In our re sults, the EDW at admission and at 48 hours was significantly associated with mortality and with a cut-off point similar to the studies cited of 14.5% (AUC 0.708 CI 0.569-0.848 p: 0.005) and, at admission, was an independent predictor of mortality with an ORadj of 5.25 (p: 0.005).
Several studies have demonstrated the predictive usefulness of MPV in sepsis, including one by Kim et al. (23, which found it to be an independent predictor of mortality at 28 days in 345 critical patients, and Zampieri et al. (24, which found it to be a strong predictor of mortality at 72 hours in 84 patients; Vardon-Bounes F., et al. (25, who studied the kinetics of MPV and determined the significance of 90-day survival of MPV with HR 3.79 and two meta-analyses, one performed by Ta jarernmuang P. et al. (26, in 11 studies (n=3724), which aimed to determine whether there is an association between MPV and mortality in critically ill patients, found a significant as sociation with day three values and identical results were re plicated in the meta-analysis by Vélez JL et al. (27, where they analyzed ten studies (n: 1845) with critically ill patients diag nosed with sepsis. In our study, we found an association with mortality of WMV from admission to 72 hours, but in the multivariate model of logistic regression, only MPV > 8.45 fL of admission had an independent predictive value for mor tality with an ORa of 5.28. An interesting fact is that in the current SARS-Cov2 pandemic, the MPV / platelet count ratio has been shown to be a useful index to predict severity of viral pneumonia28.
Other cytometric indicators such as the absolute number of neutrophils and lymphocytes and their neutrophil/lympho cyte ratio, the latter promising under the physiopathological reasoning that in bacterial sepsis the neutrophil count in creases and the lymphocyte count decreases, therefore the value of the same increases the greater the severity of the picture, were not useful in predicting mortality, this contrasts with some studies29-31 and coincides with the results of a pre vious study by the same authors32.
The strengths of this study are the concordance of its results with the world literature and that it starts from a low-cost analytical examination to obtain its results; in addition to the low probability of measurement bias by using the same equipment to measure the variables used in the cytometric parameters and indices, but, being single-centered and with a relatively small sample makes it necessary to carry out stu dies with a greater number of patients to determine if the results are replicable.
In conclusion, as our research has demonstrated, cytometric parameters and indices are a tool to determine severity and mortality in sepsis; our study determines that and positions the EDW and initial MPV as independent predictors of mortality; they should be used in association with scales and other validated bio markers to optimize management and therapy of patients with sepsis and would be an alternative in centers where only blood cytometry is available as an analytical test. The clinical significan ce of our results relies on cytometry as an alternative in centers where only blood cytometry is available as an analytical test.
















