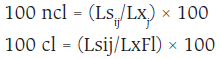Introduction
The cultivation of cowpeas (Vigna unguiculata (L.) Walp) is of great economic and social importance in developing countries and areas with low human development indexes (Moussa et al., 2011) because this crop is a source of income for small producers, and its seed is the main source of protein and minerals for many people living in these regions (Ahamefule & Peter, 2014; Boukar et al., 2011).
Although cowpeas are economically and socially important, this crop still has a low production yield. The main causes for this low productivity are the limited technology used by producers, the use of varieties not adapted to the edaphoclimatic conditions and with low productive potential, phytosanitary problems, and inadequate management of fertilisation and soil fertility (Aliyu & Makinde, 2016).
To address the problem of low productivity, more productive varieties and varieties that are adapted to regional conditions should be identified and selected (Teixeira et al., 2010). Regarding the management of the fertility of different varieties, the use of nitrogen- fixing is recommended (Farias et al., 2016; Haro et al., 2018).
Bradyrhizobium japonicum is a nitrogen fixing bacterium that has been widely studied in soybeans (Glycine max), and tested in cowpeas (Wongphatcharachai et al., 2015). Previous studies have reported increases in cowpea yields of up to 70 % when plants are inoculated with strains of this bacterium (Haro et al., 2018).
One way of evaluating the performance of varieties of agricultural crops under different conditions is by using a crop life table. Crop life tables make it possible to identify and quantify the loss factors and the different production components (Pereira et al., 2017). By using crop life tables, the critical production components and key loss factors of agricultural crops can be identified.
Knowledge of critical components and limiting factors enable the development of management strategies aimed at reducing crop losses. Thus, it becomes feasible to plan and develop breeding programmes, make fertility recommendations for cultivation, and identify varieties and strains that are better adapted to local edaphoclimatic conditions. It is worth mentioning that the present work was carried out in Brazil, there was no assessment of losses, and the data was only summarised in the production report.
Considering the many factors that may lead to low cowpea productivity in tropical regions, the present study aimed to identify and quantify the losses in the critical production component and the key loss factors for three varieties of cowpea in the presence or absence of inoculation with B. japonicum.
Materials and Methods
Study site
This study was conducted in the Amazon biome, at the Federal University of Western Pará, located in Santarém, Brazil (02°24'52"S and 54°42’36"W). The climate is classified as Am tropical (Peel et al., 2007). During the experiment, the plants were grown protected by a 50 % shading screen.
Experimental design
The experimental design consisted of randomised factorial blocks (2 inoculation levels x 3 cultivars x 30 randomised blocks), with a total of 180 plots. The cowpea varieties used were buttermilk, milk, and vinegar, and the treatment consisted of the inoculation with B. japonicum for some of the plants, while others remained inoculated.
The varieties chosen were those that producers in the Amazon prefer for growing. The buttermilk variety has an early cycle (65 to 75 days), belongs to the colour class and butter subclass, and the grain has a cream colour. The vinegar variety has a medium cycle (71 to 90 days), belongs to the colour class and vinegar subclass, and the grain has a red colour. The milk variety has a medium cycle (71 to 90 days), belongs to the white class, and the grain has a white colour (Ribeiro, 2002). The inoculant used was Bradyrhizobium japonicum, isolate BR 3262.
Substrate
The substrate used was a Yellow Latosol with a clay texture, collected at a depth of 0.0-0.2 m from the soil surface at the UFOPA Experimental Unit in Santarém, Brazil.
The chemical characteristics of the soil were as follows: phosphorus (P) = 7.0 mg dm-3; potassium (K) = 1.8 mmolc dm-3; pH (CaCl2) = 3.9; calcium (Ca) = 13.0 mmol dm-3; magnesium (Mg) = 5.00 mmolc dm-3; H + Al = 84.0 mmolc dm- 3; SB = 20.0 mmol dm-3; CTC = 104.0 mmolc dm-3; m = 0 %; V = 19.0 %; organic matter = 30.0 g dm-3.
The substrate was adjusted by increasing the base saturation to 60 % (Melo, 2007). Subsequently, phosphorus pentoxide (P2O5, 40 kg ha-1) and potassium oxide (K2O, 20 kg ha-1) were applied according to the results of the chemical fertility analysis, following the recommendations for fertilising cowpeas (Melo, 2007).
Seeding
The B. japonicum inoculant was mixed with the seeds in the inoculation treatment. Then, the seeds were inoculated one day before planting, with B. japonicum, isolate BR 3262. Following inoculation, four seeds were sown per pot and, 10 days after emergence, thinning was carried out.
Data collection
Data was collected daily during the reproductive stage of flowering. The total number of flowers and aborted flowers per plant was counted.
After the physiological maturation of the first pod, they were harvested separately for each plant twice a week.
The commercial grains, unfertilised eggs, malformed grains, grains damaged by cowpea weevils Callosobruchus maculatus (Fabricius 1775) (Coleoptera; Bruchidae), and grains damaged by other cowpea pests were counted for each plant and pod.
The cowpea pests include species of the Pentatomidae family, like brown stinkbugs, Euschistus heros (Fabricius 1798) (Hemiptera: Pentatomidae), redbanded stinkbugs, Piezodorus guildinii (Westwood 1837) (Hemiptera: Pentatomidae), and southern green stinkbugs, Nezara viridula (Linnaeus, 1758) (Hemiptera: Pentatomidae) (Ribeiro, 2002). The number of remaining malformed pods separated from the plant was also counted.
At the end of the cycle, the commercial grains were separated by plant, weighed, and their moisture content was determined. Seed analysis was conducted using the direct greenhouse method (Brasil, 2009). After determining the grain moisture, the weight of the grains was adjusted to 12 % moisture.
Creation of the crop life tables
The cowpea crop life table was adapted from the models proposed by Pereira et al. (2017). The productivity estimate (kg ha-1) in each production component was calculated as follows:
Actual productivity after creating the crop life tables was calculated as follows:
The loss estimates for each factor were calculated as follows:
Where, regarding productivity estimates, LxFl = Estimated productivity at the beginning of flowering (kg ha-1); LxPo = Productivity of the pod component (kg ha-1); LxGr = Productivity of the grain component (kg ha-1); LxGh = Real productivity (kg ha-1); Pl = Total number of plants; Fl/Pl = Total number of flowers per plant; Grt/Po = Total number of grains per pod, where Grt = Sum of commercial grains and grains with (i) factors, i = Loss factors such as non-fertilisation of the eggs, malformation of the grains, grains damaged by cowpea weevils, or grains damaged by other pests, and Po = Sum of commercial pods with malformed pods; Pgr = Average grain weight (kg); F = Conversion factor from productivity to 1 ha; Po/Pl = Total number of pods per plant; Pon/Pl = Number of commercial pods per plant; and Grn/Po = Number of commercial grains harvested from the total number of pods.
Regarding loss estimates for each factor, Fa = Loss from abortion of flowers (kg ha-1); Pol = Estimated grain loss for each factor (kg ha-1); Grl = Estimated grain loss for each factor (i) (kg ha-1); Nfa = Number of aborted flowers; NPo = Number of malformed pods; and NGr = Number of grains lost due to (i) factors.
Production losses (Dfx ij.) (kg ha-1) were calculated using the formula Dfxij'1 = Pdij - Pdj+1, where j = Production component'1 (flower, pod+, and grain), and i = Loss factors such as flower abortion, non- fertilisation of eggs, malformation of grains, grains damaged by cowpea weevils, and grains damaged by other cowpea pests, among others; Dfxij = Estimated loss for each factor (i) in each component (j) of production (kg ha-1); Pdij = Estimated productivity (kg ha-1) for each factor (i) in each component (j) of production; and Pdij = Estimated productivity (kg ha-1) in the subsequent production component (j).
The losses (100 ncl) and cumulative losses (100 cl) in percentages were calculated using the following formulae:
Where Ls = Estimated losses for each factor (i) in each component (j) of production; Lxj = Estimated productivity (kg ha-1) in each component (j) of production; and LxFl = Estimated productivity at the beginning of the reproductive phase in the flower production component (kg ha-1).
The partial loss factor (kij) for each factor (i) in each component (j) of production and the total loss factor (K) were calculated using:
Statistical analysis
In this experiment, means and standard errors for productivity and losses (kg ha-1) and non-cumulative losses (%) were calculated. Based on the calculated means and standard errors, graphs and crop life tables were created for each cowpea variety in treatments with and without inoculant.
All analyses were performed at a 5 % significance level. The productivity (kg ha-1) and cumulative loss (%) data did not meet the assumptions of the analysis of variance (ANOVA), as tested by the Shapiro-Wilk and Levene tests. For that reason, we opted for Friedman’s nonparametric ANOVA with block design, and the Wilcoxon test was used for comparisons between means.
Data on partial (kj) and total (K) losses met the assumptions of ANOVA, as tested by the Shapiro- Wilk and Levene tests. As such, parametric statistical tests were used, such as path analysis.
In this study, the coefficients of the path analysis were estimated to identify and quantify the effects of partial losses (kij), and the effects of treatment with and without inoculant on total loss (K). Path diagrams were created for each variable studied. The significance of each trail coefficient was calculated separately using standardised linear regressions.
Path analysis involves multiple regressions and allows the inclusion of direct and indirect interactions. For each path diagram, the goodness of fit (Gfi) was calculated. This parameter measures the degree of adjustment of the path model.
The non-cumulative losses with the highest mean were considered the critical production component. The critical production component that included only one loss factor was considered the key loss factor. On the other hand, for critical components that included more than one loss factor, the path coefficients were compared. The partial loss factor with the highest significant value was considered the key loss factor.
Results
Significant differences in yield were observed between cowpea varieties (x2 = 16.03, Gl = 2, p < 0.01), but there were no differences between treatments with and without inoculant (x2 = 0.40, Gl = 1, p = 0.52). However, the vinegar (1410.02 kg ha-1) and milk (1377.28 kg ha-1) varieties were more productive than the buttermilk variety (1064.18 kg ha-1) (Figure 2). Using the Wilcoxon test, we found that there was no significant difference between the yields of the vinegar and milk varieties (Z = 1.14, Gl = 1, p = 0.26). However, there were significant differences in productivity between the vinegar and the buttermilk varieties (Z = -3.14, Gl = 1, p < 0.01), and the milk and buttermilk varieties (Z = 3.71, Gl = 1, p < 0.01).
Significant differences in total losses were observed between the three cowpea varieties (X2 = 33.10, df = 2, p < 0.01), but no significant differences were observed between treatments with and without inoculant (x2 = 7.51, df = 1, p = 0.06).
The largest losses occurred in the milk variety (89.38 %), followed by the buttermilk (84.73 %) and vinegar (81.76 %) varieties (Figure 1). However, in the buttermilk variety, inoculation with B. japonicum did result in differences between yield losses of treatments, and in non-inoculated cowpeas the total loss was 84.53 %, whereas in inoculated cowpeas it was 87.28 % (Figure 1).

Figure 1. Yield and Yield loss (mean ± standard error) of cowpie bean cultivars with or without inoculation. Note: Means followed by the same lowercase letter (for comparison between cultivars) or uppercase in the column (for comparison between inoculated and non-inoculated) did not differ among themselves. Wilcoxon test at p < 0.05. N = 180.
According to the Wilcoxon test, the losses of the vinegar variety differed significantly from those of the milk variety (Z = 7.21, Gl = 1, p < 0.01) and the buttermilk variety (Z = 3.97, Gl = 1, p < 0.01). The productivity of the milk variety was different from that of the buttermilk variety (Z = -2.97, Gl = 1, p < 0.01). Losses varied between the production components. Thus, the greatest losses were observed in the flower component, followed by the grain and pod components (Tables 1, 2, 3).
Table 1 Table of yield components of losses of buttermilk cultivar with or without inoculation.
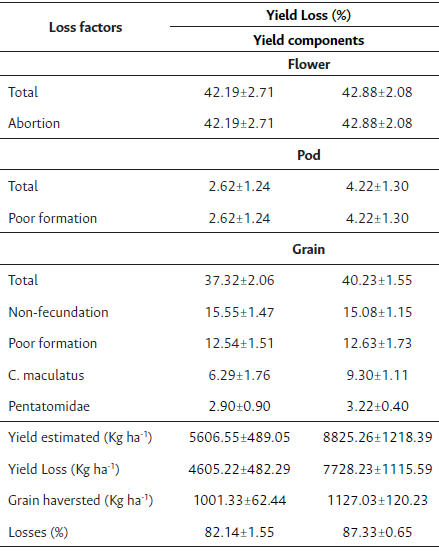
Note: N = 60.
Table 2 Table of yield components of losses of milk cultivar with or without inoculation.
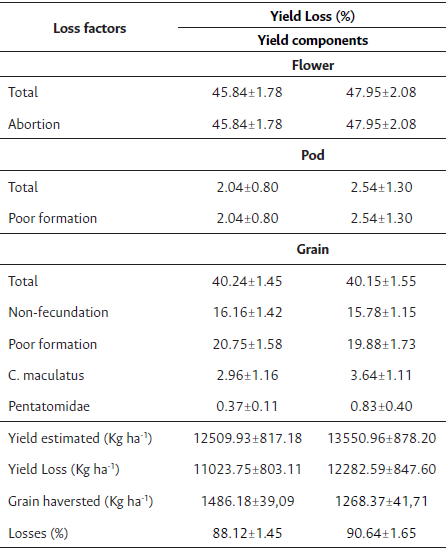
Note: N = 60.
Table 3 Table of yield components of losses of vinegar cultivar with or without inoculation.

Note: N = 60.
All path diagrams of each variety had a good model adjustment, with Gfi > 0.90 (Figures 2, 3, and 4). In all three cowpea varieties (buttermilk, milk, and vinegar), the flower component was considered a critical production component. Regarding the flower component, flower abortion was the only loss factor (Figures 2, 3, and 4).

Figure 2. Path diagram of buttermilk cultivar with a yield loss of components, factors, and inoculation effect. Note: The path diagram indicates the results of the goodness of fit (Gfi). Path (direct effects) and correlation coefficients are indicated for each interaction. The effects of one variable on another are indicated by a one-headed arrow; Correlation is indicated by a double-headed arrow. Solid lines denote direct effects, and dashed lines denote indirect effects. * indicates path significance at 5 % of probability.

Figure 3 Path diagram of milk cultivar with a yield loss of components, factors, and inoculation effect. Note: The path diagram indicates the results of the goodness of fit (Gfi. Path (direct effects) and correlation coefficients are indicated for each interaction. The effects of one variable on another are indicated by a one-headed arrow; the correlation is indicated by a double-headed arrow. Solid lines denote direct effects and dashed lines denote indirect effects. * indicates path significance at 5 % of probability.

Figure 4 Path diagram of vinegar cultivar with a yield loss of components, factors, and inoculation effect. Note: The path diagram indicates the results of the goodness of fit (Gfi). Path (direct effects) and correlation coefficients are indicated for each interaction. The effects of one variable on another are indicated by a one-headed arrow; the correlation is indicated by a double-headed arrow. Solid lines denote direct effects and dashed lines denote indirect effects. * indicates path significance at 5 % of probability.
The partial losses of the grain component significantly contributed to the total losses. The milk variety had the highest trail coefficient, followed by the buttermilk and vinegar varieties (Figures 2, 3, and 4). The factor that contributed the most to the partial losses of the grain component for the milk variety was poor grain formation. In the buttermilk and vinegar varieties, the non-fertilisation of eggs contributed the most to the partial losses of the grain component (Figures 2, 3, and 4).
Inoculation increased yield losses due to increased rates of flower abortion in the buttermilk and milk varieties, but did not affect yield losses in the vinegar variety (Figures 2, 3, and 4). Inoculation also increased the rate by which cowpea plants were damaged by cowpea weevils in the buttermilk variety. Indirectly, inoculation increased the total losses in the three varieties studied (Figures 2, 3, and 4).
Discussion
The vinegar and milk cowpea varieties had higher productivity than the buttermilk cowpea variety under the experimental conditions described above, which may be due to the adaptability of these varieties to local edaphoclimatic conditions. Thus, it is necessary to pay attention to the climatic variables of the región, as these may affect the performance and productivity of different cowpea varieties (Santos, 2011).
Abortion of flowers was the key factor behind losses in the critical flower component for the three varieties studied. Flower abortion is common in some plant species. In such plants, a large number of flowers are formed, after which the flowers with greater adaptive power are selected to be fertilised and the others are aborted (Burd, 1998; Kozlowski & Stearns, 1989).
Losses in the grain component were due to poor grain formation and non-fertilisation of eggs. These losses are also affected by climatic variables. Stress caused by high temperatures, for example, can compromise the viability of eggs, cause infertility in the female and male reproductive system, and affect grain filling (Hedhly, 2011; Snider et al., 2011).
Inoculating cowpea plants with B. japonicum did not increase the productivity of the tested varieties, however, it increased the rate of abortion of flowers in the buttermilk and milk varieties. Indirectly, inoculation increased the total losses in the three cowpea varieties studied. It may be that the strain of inoculant used was not adapted to these cowpea varieties. There are several Bradyrhizobium strains that are well-adapted to cowpeas, including the
INPA 03-11B, UFLA 3-84, BR3267, UFLA03-153, and UFLA03-164 strains (Farias, 2016). Inoculation with B. japonicum affected the rate of damage caused by cowpea weevils in the buttermilk variety. Cowpea weevils are an important insect pest of stored grains, and resistance to this insect pest varies among different cowpea varieties (Lima et al., 2001).
Conclusions
The vinegar and milk cowpea varieties were found to be more productive and better adapted to the local conditions of our study site than the buttermilk variety. The yield losses were greater in the milk variety, followed by the buttermilk and vinegar varieties. The critical production component for the three varieties was found to be the flowers, and the key loss factor was found to be the abortion of the flowers. The grain components of the three varieties affected the total losses. The factors that contributed the most to the loss of the grain components for the three varieties were the malformation of the grains and the non-fertilisation of the eggs. Inoculation with B. japonicum directly affected losses due to the increased abortion of flowers in the buttermilk and milk varieties. In addition, inoculation also affected the rate of cowpea weevil infestation in the buttermilk variety. Indirectly, inoculation with B. japonicum affected the total losses in the three cowpea varieties studied.