INTRODUCTION
Tamarind (T. indica) is a fruit native to the African continent, typical of dry environments, and cultivated in many countries. It provides a wide variety of significant ecological and economic services in rural communities since, at harvest time, it constitutes an important source of income (Azad, 2018; Maia et al., 2021). The pulp of the fruit represents almost half the weight of the pod with a higher vitamin content; it also has mineral nutrients such as sodium, calcium, magnesium, potassium, phosphorus, and iron. In addition, it has diuretic, digestive, and depurative properties; plus, in traditional medicine in the Asian and African continent, it is used for the abdominal pain treatment, helminth infections, wound healing, malaria, fever, constipation, inflammation, cellular cytotoxicity, eye diseases, antibiotic, anticancer, and antidiabetic (Govindappa et al., 2021; Fandohan et al., 2019); in the Caribbean region of Colombia, it is used in cooking, juices, ice cream, sweets, fresh food, mainly in the dry season (López et al., 2016).
The tree is slow-growing, capable of reaching a height between 12-24m, and a circumference up to 7.5m. The foliage is bright green and feathery; it is composed of pinnate leaves, each one with 10-15 pairs of oblong leaflets 1.25 to 2.5cm long and 5-6mm wide that fold up at night. Its fruits are bulging and curved pods, which usually vary from 12.48 to 22.5cm long and 1.10 to 3.0cm wide, with fruit mass between 1.4 to 56.8g, pulp mass between 0.19 and 22g, and contain from 1 to 17 seeds (Bello & Gad, 2015). The trees begin to produce fruits when they are 8-12 years old and may be continued to produce fruits for 200 years (Bahru et al., 2014).
This species is distributed mainly in dry and semi-arid regions of northern Colombia; its cultivation as a commercial crop does not exist as such and it only proliferates as patio trees, as a consequence of the dispersion of seeds by anthropic action. There is an evident lack of knowledge on reproductive ecology, population genetics, biomass production, natural distribution, and seed quality, which prevents the production of excellent quality seedlings, since their demand is high in the international market (López et al., 2016); apart from having been prioritized for conservation and product development to support livelihoods in sub-Saharan Africa and other African countries (Okello et al., 2018).
Successful seed germination is vital in the propagation of the species, as T. indica seeds do not germinate when placed under conditions normally considered favorable for emergence, given that they can take up to 60 days, which makes the process slow and erratic, even through various pre-germinative treatments (Gomes et al., 2019), as it is the case of many legumes (Yousif et al., 2020). In this regard Bhattacharya & Suparna (2021) and Yousif et al. (2020) stated that the seeds of legumes such as tamarind have dormancy imposed by the hardness of the seed covers or their impermeability to water and immaturity of the embryo, a situation that makes the germination process slow and not uniform and, when the pulp is present, the concentration of tartaric acid inhibits germination.
The physiological quality, longevity and vigor of the seeds are determining factors, which ensure germination and seedling formation as well as the preservation of T. indicus y Dipteryx alata (Silva et al., 2021; Cordeiro et al., 2022). In this sense, many methods: germination test, biochemical test, and imagen test are used to evaluate the quality of the seeds and since in tamarind the conventional germination test requires up to two months for dormancy (Bahru et al., 2014), alternative methods are used such as the tetrazolium test, based on the staining of living tissues using 2,3,5-triphenyltetrazolium chloride solution. With this test, there is an embryo coloration due to the effect of the dehydrogenase enzymes involved in respiration (Carvalho et al., 2018), which represents a rapid option for seed viability control, which in 48 hours or less, allows knowing with a high degree of reliability the physiological quality of the seed destined for sowing or to evaluate its possibility of storage (Souza et al., 2018; Costa et al., 2018; Cordeiro et al., 2022).
Viability testing with tetrazolium has been employed in many species (Franca-Neto & Krzyzanowski, 2019). Recently its use has been reported in Carica papaya (Carvalho et al., 2018), Glycine max (Zuffo et al., 2018), Scaevola sericea (Liang et al., 2020), Psidium guajava (Guevara-Ohara et al., 2020), Phaseolus vulgaris (Besaye & Galgaye, 2022) and Tamarindus indica (Cordeiro et al., 2022).
Considering the importance of tamarind in the Caribbean region and that the conventional evaluation of its seed takes too long, the objective of this study was to evaluate seed viability by means of the tetrazolium test and reduce the time necessary to determine its physiological quality.
MATERIAL AND METHODS
Plant material. The seeds of T. indica were collected from ripe healthy fruits of patio trees randomly taken from the rural area of Montería-Colombia (San Isidro village) at the beginning (January-February) of 2021, with coordinates 8.57833N and 75.8933W).
Location and Procedure. The study was carried out in the plant breeding laboratory of the Cordoba University - Colombia. The seeds were separated from the pulp and sieved to separate the damaged ones; they were air-dried, kept in plastic packaging, and stored at a temperature of 10°C (Bahru et al., 2014), until the commencement of the experiment four months later. Likewise, they were disinfected by immersion in 1% sodium hypochlorite for one minute, washed with abundant distilled water to remove the hypochlorite that could remain in the seeds and dried in the open air to avoid problems of damage to the internal anatomy and to optimize the tetrazolium test (Salazar et al., 2020). The moisture content of the seeds was determined with three samples of 9.82, 9.90, and 10.13g by the oven drying method (dehydrating them for 17 hours at 103°C), and they showed an average of 19.5%.
Preliminary tests were carried out to determine the ideal number of hours for the immersion of seeds, which would allow a longitudinal cut to directly expose the embryo to the tetrazolium salt solution, and thus, obtain an adequate coloration. Times of 24, 48, and 72 hours were tested at 40°C in dark conditions. Previously, the seeds were scarified with sandpaper N°100 all over the edge, except in the area where the micropyle is located. For the experiment, the seeds were divided longitudinally by the embryonic axis, with subsequent removal of the seed coats. The embryo of each seed was immersed in a solution of 2,3,5- triphenyl tetrazolium chloride, at concentrations of 0.50%, 0.75%, and 1.00% in the dark, at temperatures of 40ºC for 2, 4, and 6h, using four subsamples of 20 seeds per experimental unit inside 50ml beakers containing 15ml of the tetrazolium solution.
Seed viability. After subjecting the seeds to immersion times with the tetrazolium salt, the solutions were drained, and the seeds were washed with running water and individually evaluated using a stereoscope (Vista vision) to verify the presence or absence of red color, uniformity, location, and color intensity of embryonic tissues, being classified into two categories: 1) viable and 2) non-viable (Souza et al., 2018; Salazar et al., 2020).
Viable seeds were considered those whose embryos showed a uniform bright pink color, tissue with normal appearance, firmness, and cotyledon with more than 50% of its surface colored. The non-viable seeds were those with the embryo totally white (without coloration) or having part of the radicle without coloration; the presence of white tissues in the cotyledons indicates dead tissue. In order to know the physiological quality of the seed, these were evaluated through the traditional method and scarification of 1mm thickness with water sandpaper # 60 on both sides. The germination percentage (%G) was measured by counting the number of emerged seeds up to day 25 after sowing and the germination speed index (GSI) was measured simultaneously with germination using equation (1), recommended by Maguire (1962) .
Where, Pi is the number of normal seedlings germinated per day, Ti is e the number of days elapsed since planting, i is the number of counts, i=1,2,3 ........n
Experimental design. The experimental design was completely randomized with four replicates in a 3 x 3 factorial arrangement, three concentrations of tetrazolium salt and three immersion times, at a temperature of 40°C. One hundred seeds per replica constituted the experimental unit. Data were subjected to analysis of variance and Tukey's multiple comparisons at 5% for the viable seed variable. The results were analyzed by the statistical package SAS 9.4 (SAS, 2013).
The experimental design was completely randomized with two treatments and five replications, each experimental unit consisted of 20 seeds sown in plastic bags of 15 cm in length and 8 cm in diameter, with the following substrate mixture: sand, rice husk and alluvium in a 1:1:1 ratio, characterized by its high-water retention and aeration capacity. Irrigation was carried out every two days, between 11:00 am and 12:00 noon.
RESULTS AND DISCUSION
The initial moisture content of the seed, 19.5%, allows water to pass from the external medium of higher water potential to the interior of the seed, whose water potential is lower until reaching an equilibrium independent of its physiological state (Marcos-Filho, 2015). This allows the seeds to perform all their metabolic activities, including respiratory activities, which are necessary to carry out the tetrazolium test (Brito et al., 2020). The Analysis of Variance for tetrazolium concentrations, immersion time, and concentration-immersion time interaction, under controlled conditions at 40°C, is set out in Table 1.
Table 1 Mean squares of the Analysis of Variance for the characteristic percentage of viable seeds subjected to different concentrations of tetrazolium and immersion times.
| Variation source | DF | Viable seeds (%) |
|---|---|---|
| Tetrazolium | 2 | 17.59NS |
| Time | 2 | 1989.81** |
| Tetrazolium x Time | 4 | 212.03* |
| Error | 18 | 54.62 |
| Total | 26 | 224.85 |
| C.V. (%) | 10.61 | |
| Mean | 69.62 | |
| R2 | 0.83 |
*: significant with p<0.05; **: highly significant with p< 0.001; NS: not significant with p<0.05; C.V.: coefficient of variation.
There were no significant differences between the different concentrations of tetrazolium salt while estimating embryo viability, so any concentration is reliable to obtain accurate topographic interpretations since they allowed the assessment to identify healthy tissues in viable seeds and alterations of non-viable seed tissues.
The use of tetrazolium solutions at low concentrations allows for a higher yield of tetrazolium salt during analyses. In this regard, Cordeiro et al. (2022) evaluated tetrazolium concentrations of 0.075%, 0.1%, and 0.5% and established that 0.1% for 6 hours allows efficient evaluation of viability in T. indica seeds. Regarding the immersion time, highly significant differences were detected (Table 2, Figure 1), highlighting that the longer the time in the tetrazolium salt solution, the higher the percentages of determinations of viable embryos of this species, with values ranging from 81.66 ±4.40 to 83.33± 6.00.
This enabled us to affirm that six hours of immersion is sufficient for the cells of the embryo to react with tetrazolium salt, and accurate topographic interpretations can be achieved and used to estimate viability. Similar findings have been reported across various species, including Zuffo et al. (2018) and Franca-Neto & Krzyzanowski (2019) . Additionally, similar results were obtained in studies on Phaseolus vulgaris by Besaye & Galgaye (2022) and T. indica by Cordeiro et al. (2022) .
Table 2 Mean percentages of viability of Tamarindus indica seeds obtained at different concentrations of tetrazolium (TZ) and immersion times.
| Time (hours) | |||
|---|---|---|---|
| TZ concentration | 2 | 4 | 6 |
| 0.50 | 60.00±5.77 b | 63.33±4.40 b | 81.66±4.40 a |
| 0.75 | 73.33±4.40 b | 46.66±1.66 c | 83.33±1.66 a |
| 1.00 | 63.33±3.33 b | 61.66±4.40 b | 83.33±6.00 a |
Different letters between columns indicate statistical difference between means (p<0.05) according to Tukey's test.
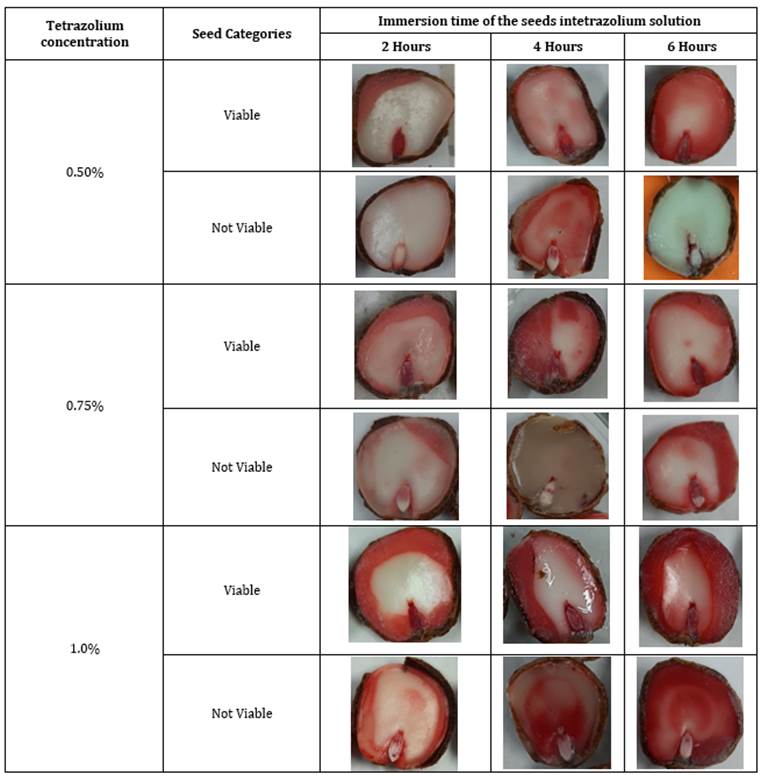
Figure 1 Seed tissue staining patterns under different tetrazolium concentrations and immersion times.
The tetrazolium concentration-immersion time interaction registered significant differences (Table 1), indicating that the viability of the seed varies according to the concentration of the tetrazolium salt and the immersion time in the solution (Table 2). However, it was observed that the longer the immersion time, the greater the red coloration in the viable tissues of the seeds due to the activity of the dehydrogenase enzymes of respiration. Similarly, when the physiological activity is deficient, the tissues remain discolored or exhibit abnormal coloration since they release small amounts of H+, which are insufficient for a normal reaction to occur with the tetrazolium salt (Marcos-Filho, 2015).
Studies on Cucumis anguria seeds reported by Paiva et al. (2017) , who evaluated different concentrations of the tetrazolium salt and immersion times at temperatures of 30 and 40°C, respectively, found that the treatment of 0.050% for six hours at 35°C or four hours at 40°C are sufficient to identify viable seeds. Therefore, the immersion time of the embryos in the tetrazolium solution and its concentration generate variations in the staining patterns as can be seen in Figure 1 (Paiva et al., 2017; Pereira et al., 2020).
Topographic or staining patterns related to seed viability were identified according to the response of different tissues to the tetrazolium salt reaction. The activity of dehydrogenases allows the release of hydrogen ions, which reduces the colorless tetrazolium solution to red formazan, which stains living cells red, while dead cells remain uncolored. The seeds of Figure 2a are viable while those of Figure 2b are considered non-viable. Since the key structure that should present staining is the radicle, a light pink non-homogeneous color in the radicle or lack of embryo coloration results in the classification of the seed as non-viable (Mancipe-Murillo et al., 2018).
The results obtained in this research effectively provide the procedure for the determination of tamarind seeds viability, thereby constituting the tetrazolium test as a potential alternative for quickly quantifying the germinable or non-germinable state of the embryo. It is important to bear in mind that the tetrazolium test has some disadvantages, such as being a biochemical method that requires some experience and skill to perform and analyze.

Figure 2 Viability of the embryo of Tamarindus indica seeds when subjected to the 0.50% tetrazolium solution a) Viable seeds b) Non-viable seeds.
The analysis of variance for the variables germination percentage and germination speed index is shown in Table 3, indicating the absence of significance for %G between treatments and highly significant differences between them for GSI. Seed quality measured through the %G were 67 and 50%, respectively for scarification and conventional, acceptable values and lower than those reported by Bello & Gad (2015) , who using concentrated sulfuric acid reported a germination percentage of 95%, since the acid softens the teguments and facilitates imbibition and accelerates germination, with the disadvantage of the handling of this acid by the operator. Regarding GSI, scarification was superior with a value of 1.78, which benefits vegetative development in the nursery stage with better quality seedlings; while the conventional value of 0.74 reflects the physical dormancy imposed by the teguments.
Table 3 Mean squares of the analysis of variance of two characteristics associated with physiological seed quality.
| Variation source | DF | %G | GSI |
| Treatments | 1 | 722.5ns | 2.70** |
| Error | 8 | 166.25 | 0.11 |
| Total | 9 | 228.05 | 0.39 |
| C.V. (%) | 22.04 | 26.32 | |
| Mean | 58.5 | 1.26 | |
| R2 | 0.35 | 0.75 |
DF: degrees of freedom; G: germination;GSI: germination speed index; C.V.: coefficient of variation, **; highly significant with p< 0.001; NS: not significant with p<0.05
CONCLUSIONS
Our findings revealed that the viability of tamarind seeds can be effectively assessed by subjecting them to a 0.50% tetrazolium concentration for a duration of six hours at 40°C, under laboratory conditions. The scarification of the seed with sandpaper allows obtaining seedlings of better quality regarding their growth and vegetative development. The tetrazolium test serves as an efficient alternative to conventional germination tests for evaluating the quality of tamarind seeds in terms of cost and time.
















