Introduction
Gallstone ileus manifests as a mechanical obstruction of the gastrointestinal tract, frequently preceded by cholecystitis. It is responsible for 1%-3% of total cases of obstruction, occurring in 0.3% to 0.5% of all patients with cholelithiasis1.
It is caused by the passage of a stone and its subsequent accommodation in the lumen of the GI tract2 due to the formation of fistulas between the gallbladder and the intestine. The most common fistulas are cholecystoduodenal fistulas, followed by cholecystojejunal, cholecystocolonic, and cholecystogastric fistulas. Gallstone ileus is less common due to the passage of stones >2 cm from the ampulla of Vater to the GI tract. When the stone lodges in the duodenum and obstructs the gastric outlet, it is called Bouveret syndrome. In contrast, when it hits the ileocecal valve, it is called Barnard syndrome3, and the most common sites of obstruction are the ileum, jejunum, duodenum, colon, and stomach4.
The clinical picture manifests in acute, subacute, and chronic forms. In the formers, there is a sudden intestinal obstruction with nausea, vomiting, abdominal pain, distension, absence of feces, and flatus. In the subacute form, symptoms of intestinal obstruction occur, but flatus is present. In the chronic form (called Karewsky syndrome), there are intermittent periods of obstruction followed by asymptomatic periods caused by the passage of the stone through the intestinal lumen until its expulsion in the feces or final impaction. In older patients, the Mordor triad (intestinal obstruction, history of biliary disease, and signs of cholecystitis) occurs5.
Clinical case
This is an 86-year-old male patient with a history of cerebrovascular disease, atrial fibrillation, and a pacemaker. He presented with a 24-hour clinical picture of abdominal pain in the right upper quadrant, associated with fever and emesis of bile content. On physical examination, he was found to be dehydrated, jaundiced, and with a positive Murphy’s sign. Paraclinical tests showed leukocytosis, neutrophilia, and altered hepatobiliary biochemistry. A diagnosis of Tokyo 2 cholangitis with intermediate risk for choledocholithiasis was made, and antibiotics were started according to management guidelines. An ultrasound of the bile ducts was taken, reporting a gallbladder with gas interposition and a significant acoustic shadow suggestive of a gallbladder calculus mold corresponding to a positive Wes sign. Due to the history of pacemaker use, cholangioresonance was ruled out, indicating an abdominal tomography with double contrast.
During hospitalization, his general condition deteriorated, and sepsis of biliary origin was suspected, so they decided to take him for management in the intensive care unit (ICU). The abdominal tomography report did not allow an adequate definition of the gallbladder; it showed target- and string of pearls-like images, suggestive of mechanical intestinal obstruction, associated with intussusception and intestinal ischemia with significant distention of thin loops (Figure 1). Additionally, an image of intrahepatic pneumobilia was observed (Figure 2), for which an exploratory laparotomy was performed. An intestinal obstruction was found 170 cm from the angle of Treitz due to an ogival gallstone measuring 6 * 4 cm (Figure 3). An enterolithotomy was performed without correction of the cholecystoenteric fistula in the first surgical stage, requiring the placement of the vacuum-assisted closure (VAC) system secondary to evisceration due to significant distention of intestinal loops.
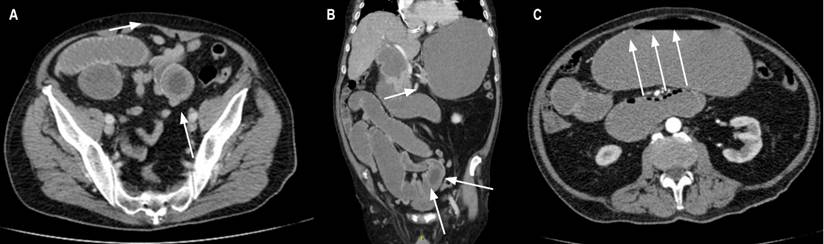
Figure 1 A and B. Target-like image from abdominal tomography suggestive of intestinal obstruction due to intussusception. C. String of pearls-like image suggestive of intestinal obstruction. Images owned by the authors.
In the second surgical stage, the dysfunctional VAC system, kinking of the enterorrhaphy, and an intraluminal clot proximal to it that obstructed the intestinal lumen were found, so the second plane of enterorrhaphy was removed with permeabilization of the intestinal lumen and skin closure.
A flap closed the fascia and the skin in the third surgical stage. During the procedure, no leakage through the enterorrhaphy was observed. Subsequently, the patient was discharged without post-surgical complications.
Discussion
The diagnosis is made by X-ray, ultrasound, or abdominal tomography imaging. The X-ray showed pneumobilia or Gotta-Mentschler sign, distention of the intestinal loops, and radiopaque stone (Rigler’s triad) pathognomonic of the disease when at least two criteria are present. However, it is only found in 15-53% of cases. It becomes Rigler’s tetrad when a change in stone position is located on serial X-rays. Additionally, if an X-ray with contrast medium is performed, Forchet’s sign (intestine with a snake-shaped silhouette surrounded by a halo of radiolucent stones) and Petren’s sign (passage of contrast medium into the gallbladder through the fistula) are described3,6,7. Abdominal ultrasound has better diagnostic performance in detecting radiopaque stones, pneumobilia, and the fistula site; however, it is operator-dependent, and the patient’s clinical status may alter the examination results. Abdominal tomography is considered the gold standard for diagnosis since it has a sensitivity of 93% and a specificity of 100%, in addition to determining the viability of the intestinal segments8,9.
Initial management involves intestinal rest, decompression of the GI tract using a nasogastric tube, volumetric resuscitation with crystalloids, and electrolyte replacement10, followed by surgical management for deobstruction by removing the stone1. The procedure is performed with a clipping of the affected intestine, a longitudinal cut at the antimesenteric border, stone extraction, and transverse closure of the intestinal wall to prevent stenosis6.
The optimal management in these patients is discussed, describing three types of procedures: enterolithotomy, enterolithotomy plus fistula closure in a single surgical stage, or enterolithotomy plus delayed fistula closure8. The procedure is chosen individually according to the patient’s condition: in critically ill patients, only enterolithotomy is performed, while in patients with less severe forms of the disease, the fistula is closed in a single surgical stage. Delayed fistula closure is chosen in patients with a persistent clinical picture in whom only enterolithotomy was performed since the risk of recurrence is 5% to 33%, and spontaneous fistula closure occurs in up to 50% of cases5,9. It has been reported that the obstruction is managed by removing the stone by open and laparoscopic means in cases of stone enclavation at the small and large intestine levels or less commonly by endoscopic means when the obstruction occurs in the sigmoid colon8,11.
Conclusions
We presented a clinical case of intestinal obstruction secondary to gallstone ileus with the typical characteristics of intestinal obstruction preceded by cholecystitis, successfully managed with enterolithotomy in the first surgical stage without closure of the cholecystoenteric fistula. Subsequently, he required multiple surgical reinterventions secondary to bleeding and kinking of the enterorrhaphy. However, he had a favorable outcome on discharge. The findings of the clinical picture are compatible with what has been reported in the literature. The surgical approach is controversial since a consensus has yet to be reached on the surgical technique of choice due to the small number of reported cases.











 text in
text in 





