1. INTRODUCTION
Piezoelectric materials are very important for the electro-electronic industry; they are used in ultrasound devices, electro-mechanical energy converters, and clean energy generators. However, some of the main components of such materials are elements that cause human health problems and environmental damage. For instance, lead zirconate titanate (PZT), one of the most commercially used piezoelectric materials, contains around 60% lead by weight [1]. People who suffer from lead poisoning can experience headaches, constipation, nausea, anemia, and reduced fertility. Moreover, prolonged exposure to lead can cause more serious symptoms, such as severe damage to nerves, kidneys, and brain [2]. Consequently, we must find possible lead substitutes. A promising candidate to replace PZT is sodium potassium niobate ((K,Na)NbO3, KNN), a ceramic material that possesses excellent piezoelectric properties [3][4]. Further, said ceramic host might be doped with rare earth ions, transforming KNN into a multifunctional ceramic with two or more desirable properties, e.g., piezoelectricity and photoluminescence, which make it suitable for manufacturing sensors and actuators [5]. However, the production of 𝐾0.5𝑁𝑎0.5𝑁𝑏𝑂3 involves many difficult conditions that include the volatility of alkaline elements, the high influence of stoichiometry on the physical properties, and problems with densification processes [6], [7].
KNN production by means of solid-state synthesis requires several hours of reaction and high temperatures, which hampers said production due to the instability of the precursors and the volatility of alkaline metals [5]. In order to solve the densification problem, different advanced processes have been used to promote sintering, such as spark plasma sintering [8] and hot pressing [9].
In spite of those disadvantages, different factors that affect the obtained powders-such as the selection of precursors, the chemical heterogeneity of calcined powder, calcination temperatures, and grain size are still studied and investigated in full detail [7], [10], [11], [12].
The fragmentation mechanism of micrometer particles induced by the grinding process may be a key element to obtain one or another particle shape of micrometric size [13]. This is very important to obtain 𝐾0.5𝑁𝑎0.5𝑁𝑏𝑂3 powders with structural and microstructural properties that allow the continuation of the densification process.
This work presents a comparison and optimization of different conditions to prepare ceramic powders of 𝐾0.5𝑁𝑎0.5𝑁𝑏𝑂3 in order to determine the effect and efficiency of using a horizontal grinder and a planetary ball mill grinder. Such powders were obtained by the solid-state reaction method, which offers several advantages over the techniques mentioned above, e.g., a simple synthesis route, low synthesis cost, and ease of commercial application [14][15][16]. We also studied the reaction conditions that do not alter the elemental composition; additionally, we obtained the specific morphologies, uniform particle sizes, and crystalline phases, which allow the densification and/or doping of the obtained powders.
2. MATERIALS AND METHODS
Ceramic powders of 𝐾0.5𝑁𝑎0.5𝑁𝑏𝑂3 were prepared via solid-state reaction (oxide mixing) using niobium pentoxide, Nb2O5 (Aldrich, purity 99%); sodium carbonate, Na2CO3 (Aldrich, purity 99%); and potassium carbonate, K2CO3 (Alfa Aesar, purity 99%). The powders were mixed according to the stoichiometric formula (1) and calcined according to the diagram in Fig. 1.

Source: Authors’ own work.
Fig. 1 (Color online) Sequence diagram of the synthesis of KNN in the solid state.
Initially, each reagent was macerated and thermally treated separately for two hours at 250°C. This happened because the hygroscopic nature of the carbonates from alkaline metal precursors to eliminate water and guarantee the weight of the powders. Then, the carbonates and the oxide were mixed in two different grinders: a planetary ball mill grinder and a horizontal grinder. The horizontal grinder consists in a cylindrical chamber that contains balls and rotates around its horizontal axis. Balls are swept along by the wall and then fall onto the load. Subsequently, particle fragmentation results from the compression of the particles on the bed by the balls. In the planetary ball mill grinder (PM400 - Retsch), the grinding chamber rotates on an orbit around the center. This rotational movement is the self-rotation of the grinding container superimposed. The resulting centrifugal and acting acceleration forces lead to strong grinding effects. Furthermore, there are forces working according to the coriolis acceleration. The result is an intensive grinding effect between the grinding balls and the sample.
The grinding process was carried out for 2h at 200 RPM. Afterward, the resulting samples were dried at 110°C for 48 h to remove water and obtain dry ceramic powder for characterization. The reaction sequence of the powders was investigated through thermogravimetry and differential scanning calorimetry (TG and DSC, respectively) at an incremental rate of 10° C/min, starting at room temperature and up to 1200° C, in an air atmosphere, using a Netzsch Simultaneous Thermal Analysis system (STA 409 ET). Such analysis allowed us to establish 800°C, 850°C, and 900°C as the calcination temperatures. The phase formation of the calcined samples was identified via x-ray diffraction, with a scan from θ=10º to θ=60º, using a Rigaku diffractometer with CuKα radiation at room temperature. The size, agglomerate formation, and morphology of the particles of the calcined powders were studied via scanning electron microscopy using a Jeol JSM 5800 LV microscope.
3. RESULTS AND DISCUSSIONS
Figure 2 shows the TG/DSC of the non-calcined powders of the material obtained after drying and maceration in the horizontal grinder (the TG/DSC results of the powder obtained in the planetary grinder exhibited a similar behavior, but they are not included here). With respect to weight loss, two regions can be clearly distinguished. The first one is a 15.1% weight loss that can be attributed to water evaporation. The presence of water in the carbonate-oxide powder mixture is due to the hygroscopic nature of both carbonates, particularly K2CO3, which easily absorbs in a normal atmosphere where the manipulation of the precursor powder takes place. The second region begins at around 850°C, and it is possibly caused by the loss of carbon dioxide (see products of Equation (1)). These results allow us to establish that, for this work, 800°C, 850°C, and 900°C are the calcination temperatures, which are usually employed for the calcination of 𝐾0.5𝑁𝑎0.5𝑁𝑏𝑂3 powders obtained by oxide mixing [17].

Source: Authors’ own work.
Fig. 2 (Color online) TG/DSC of non-calcined powders obtained by horizontal grinding
Figure 3 presents the x-ray diffraction patterns of 𝐾0.5𝑁𝑎0.5𝑁𝑏𝑂3 powders mixed in a planetary ball mill grinder at 200 RPM for 2h and calcined at different temperatures (800°C, 850°C, and 900°C) for 2h. At the three temperatures, the expected peaks of K0.5Na0.5NbO3 can be seen (PDF card #32-0822), but with the presence of secondary phases and precursors that do not completely react (K2CO3, Nb2O5, and K3Nb6O17), as reported by Mahdi et al. [18].

Source: Authors’ own work.
Fig. 3 (Color online) X-ray diffraction pattern of K0.5Na0.5NbO3 using a planetary ball mill grinder at different calcination temperatures (800° C, 850° C, and 900° C)
Note that, in the case of the powders produced with the planetary ball mill grinder, the spectrum with the least percentage of secondary phases was obtained calcinating at 850°C. Characteristic peaks of the K0.5Na0.5NbO3 phase can clearly be seen at 2θ=45-47, revealed by the relative intensities at the (200) and (002) diffraction peaks, and 2θ=51-53, which indicate a coexistence of tetragonal and orthorhombic phases of the perovskite structure[19][20].
The x-ray diffraction patterns of calcined powders obtained by horizontal grinding at 800° C, 850° C, and 900° C for 2h can be seen in Fig. 4. As in the case of the powders obtained in the planetary ball mill grinder, the characteristic peaks of 𝐾0.5𝑁𝑎0.5𝑁𝑏𝑂3 can also be identified. Additionally, the powders in Fig. 4 exhibit a lower percentage of secondary phases than those in Fig. 3 (the secondary phases of Nb2O5, and K3Nb6O17 can be located in Fig. 4). It is also clear that calcined powder obtained by horizontal grinding at 900° C for 2h is the most suitable option to be calcined and/or doped because it presents a lower quantity of secondary phases. It has been reported that the doping process decreases and/or eliminates the formation of secondary phases in the 𝐾0.5𝑁𝑎0.5𝑁𝑏𝑂3 system.
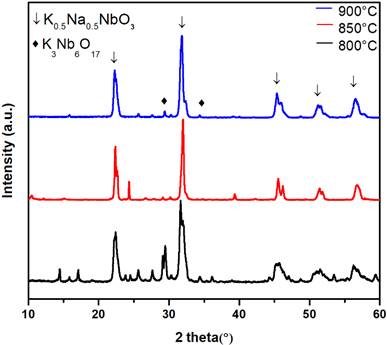
Source: Authors’ own work.
Fig. 4 (Color online) X-ray diffraction pattern of K0.5Na0.5NbO3 using a horizontal grinder at different calcination temperatures
Fig. 5 shows the micrographs of the two calcined KNN powders: (a) ground in a planetary ball mill grinder and calcined at 900°C and (b) horizontally ground and calcined at 900°C for 2 h. The micrographs of the powders obtained with the other calcination temperatures are not shown here because of the presence of multiple secondary phases. Parallelepipeds and other cubic morphologies can be seen in Fig. 5, as well as fractured particles and the formation of agglomerates. At said calcination temperature (900ºC), the horizontal grinder produces better results than those reported in the literature-homogeneous particle sizes (1-5 µm), lower formation of agglomerates, and fewer secondary phases-which can be confirmed with the diffractograms [21].
4. CONCLUSIONS
The horizontal grinding system enabled us to obtain, by solid-state reaction, powders of the 𝐾0.5𝑁𝑎0.5𝑁𝑏𝑂3 system with a lower percentage of secondary phases and particle size, as well as a morphology suitable for the fabrication of piezoelectric ceramics with KNN and/or doping the powders for the synthesis of multifunctional systems.
















