INTRODUCTION
The healing process comprises several intracellular and intercellular pathways, being activated and coordinated to restore tissue integrity and homeostasis. At the same time, cellular components of the immune system, blood coagulation cascade, and inflammatory pathways come into action; several cell types - immune cells, endothelial cells, keratinocytes and fibroblasts - undergo marked changes in the expression and phenotype of genes, leading to cell proliferation, differentiation, and migration [1].
In general, the goals of wound treatment are to prevent infections, reduce edema and inflammation, accelerate healing and minimize scar formation. However, the wound healing process can be aggravated by intrinsic and extrinsic factors, such as poor circulation at the wound site, comorbidities, and microbial infection [2].
Throughout history, using natural products is the basis for treating various diseases, being present in several cultures articulating empirical knowledge and health. Several natural products with healing activity have already been described in the literature, from popular use to commercially available formulations, for example, the species Orbignya phalerata (babassu coconut) has shown to accelerate the healing time in oral and topical treatment in models of induced lesions [3], Copaifera langsdorffi (Copaiba oil) was shown to have a healing effect from the topical treatment of induced skin lesions, with dose-dependent reduction in tissue repair time [4], and Dersani® (sunflower oil), a commercially available formula derived from a natural product, which favors the healing process in the topical treatment of wounds in elderly mice [5].
Likewise, the plant species Punica granatum L., popularly known as pomegranate, has been widely used as a medicine in many countries. In addition to its nutritional benefit, the fruit is also used empirically for treating several diseases such as acidosis, dysenteria, microbial infections, diarrhea, helmintiasis, hemorrhage, and respiratory pathologies [6, 7]. In vitro and in vivo studies have sought to scientifically validate its therapeutic potential for wound treatment [8, 9].
In this context, this study aimed to conduct a literature review on in vivo therapeutic effects of different P. granatum extracts in the wound healing process, synthesizing data on extract production, combinations of different formulations, animals and experimental models frequently used, and the main results described in these studies, in addition to presenting clinical studies that used this natural product and perspectives for future study.
METHODS
This study was conducted through the following stages: guiding question elaboration, search for primary studies, assessment of primary studies, data extraction, analysis and synthesis of results, and presentation.
The guiding question elaboration was based on PICO strategy, in which "P" refers to the study population (experimental models in vivo); "I" refers to the intervention studied or the variable of interest (use of Punica granatum extracts as a healing agent); "C" refers to comparison (comparison between different in vivo models); "O" refers to the outcome of interest (healing action of in vivo extract) [10]. Thus, the guiding question of this integrative review was: does the Punica granatum extract have healing activity on in vivo models ?
For the survey of articles in literature, a search was conducted in the PubMed, Scopus, Science Direct and Science Citation Index Expanded (Web of Science) databases. The following controlled descriptors, keywords and synonyms with Boolean operators were used for database crossing: "Wound healing" OR "Healing, Wound" OR "Healings, Wound" OR "Wound Healings" AND "pomegranate" OR "pomegranates" OR "punica granatum".
We included articles that assessed the healing activity of P granatum extracts on in vivo models, without language restriction or period of time. Articles that analyzed experimental models other than in vivo, as well as theses, dissertations, literature review and duplicate articles were excluded. A total of 176 primary studies were identified. Of these, 30 were excluded because they were duplicated; 131 were excluded after reading their titles and abstracts; 1 was excluded for not meeting the guiding question. The final result consisted of 14 studies, 12 studies in experimental models and 2 human studies. The study selection is presented in the flowchart (figure 1), as recommended by the Prisma group [11].
RESULTS AND DISCUSSION
From the established criteria, 14 studies were selected for this review. These studies were reviewed to extract data such as: pharmacogen, complete product ingredients (in combined preparations), animal and experimental model. The results were searched for differences between the test group and the control group in wound contraction, neovascularization, collagenization, inflammatory reactions and local tissue biomarkers (tables 1, 2).
Table 1 Preparations based on Punica granatum extracts for wound treatment.
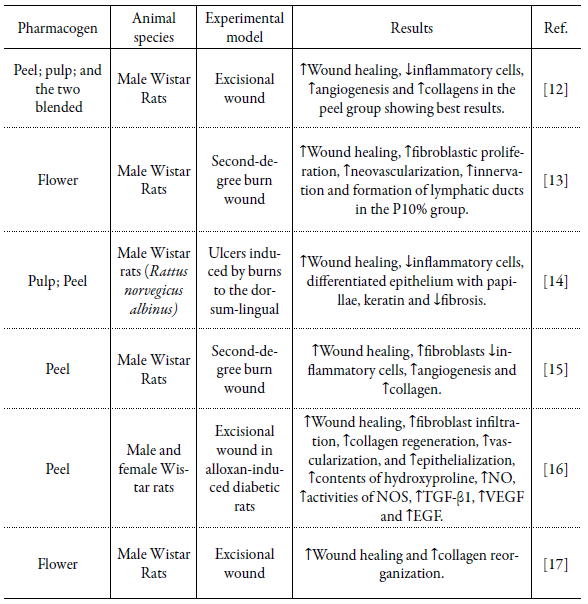
P10%, cream containing a concentration of 10% of P. granatum flower extract; NO, nitric oxide; NOS, nitric oxide synthase; TGF-ß1, growth factor-ß1; VEGF, vascular endothelial growth factor; EGF, epidermal growth factor; ↑, increase in mentioned variable; ↓, decrease in mentioned variable.
Table 2 Multi-herbal preparations with Punica granatum used for wound treatment.

T4, Powder of standardized pomegranate extract (SPE) with 40% ellagic acid with a concentration of 10% SPE; P4, pomegranate extract standardized to 40% ellagic acid with a concentration of 7,5%; ↑, increase in mentioned variable; ↓, decrease in mentioned variable.
Punica granatum Linnaeus (Lythraceae), commonly known as pomegranate, is a plant native to Iran, and can be found throughout the Mediterranean region and also in Brazil, with wide use worldwide as a food and medicinal source [24, 25].
Its whole fruit was shown to have anti-inflammatory and antioxidant effects [26], the oil of its seeds was able to decrease blood sugar levels of fasting patients [27], while extracts from the peel exerted anti-hyperglycemic effect, hypolipidemic, hepatoprotec-tive, antioxidant [28], promoting wound healing [18].
When analyzing several pomegranate cultivars originating in Iran, higher levels of total tannins, phenolics and antioxidant activity in pomegranate juice were identified. The peels had higher content of total flavonoids, anthocyanin and ascorbic acid [29]. This shows that the choice of a given pharmaceutical can influence the biological effect produced due to its chemical composition, and it is necessary to identify the mechanisms of action of these bioactive substances for their medicinal application according to the pathology to be treated. In this study, it was identified that the part of the most used plant was peel and flowers, followed by the extract of the whole fruit, pulp or peel/pulp combined, only one study did not indicate which part of the plant was used for extract preparation.
Pomegranate has been known for its food and medicinal value. About 50% of the total weight of the fruit corresponds to the peel, which is a rich source of bioactive phytochemicals such as tannins (punicalin, pedunculagin, punicalagin, gallic acid, gallic acid and glucose ellagic acid esters) and other phenolics including flavonoids [29, 30]. The peel is generally discarded as residue, being little explored by the food industry, which makes it the ideal part for medicinal use.
Using different parts of the plant in healing models also demonstrated distinct effects. The extract of the upper layer of the peel was able to significantly improve wound healing, while the pulp (membrane between the arils) showed no promising effects, with healing percentages of 94.83 % and 78.17 % on day 14, respectively. The ointments made with the peel extract or with the peel and pulp extracts mixed were effective in visual improvement of the wounds, as well as in histopathological parameters, reducing the number of cells of the immune system - polymorphonuclear (PMN) and macrophages - accelerating the second stage of healing and increasing fibroblast migration to the injured tissue [12].
The healing properties of P. granatum (PG) extracts are closely linked to the presence of tannins, phenols, and flavonoids. Another important factor is the concentration that these compounds are in the formulations used. In this study, concentrations of PG extract ranging from 2 % [8] to 30 % [16] were identified in the formulations, and the use of formulations that had a concentration of ~10 % of PG extract was more frequent.
Some authors who used experimental groups with formulations that had different concentrations pointed out that those that contained 10% of the extract in their formula obtained better results [13, 19]. After using variations in PG concentration between 2.5 and 7.5 % in its formula, it was identified in a higher concentration (7.5 %) than collagen deposition, infiltration of polymorphonuclear neutrophils (PMN), angiogenesis and degree of fibrosis were significantly different from the other groups, including those with lower concentrations of extract (2.5 % and 5 %), showing a dose dependent response [20]. Identifying the concentration of compounds used in these formulations is extremely important for the development and standardization of a masterful formula for medicinal and commercial use.
In the search for an ideal formula, some studies seek to mix different substances - natural or synthetic - in order to potentiate the effects of these compounds. The combination of bioactive substances is promising in treating complex pathological processes, such as healing, because it allows simultaneous action of these substances on several targets, ensuring greater efficacy and less chance of resistance to treatment [31].
Some studies that used PG were identified in association with others natural products, the combination of henna extract (a dye prepared from Lawsonia inermis), pomegranate and myrrh (a natural gum or resin extracted from a tree species of the Commiphora genus) were able to increase contraction and decrease wound time of epithelization. The ethanolic extract of PG flowers alone also obtained total wound closure on the same day of the mixed group [21]. A different result was observed when a mixture of ethanolic extracts from PG and Malva sylvestris flowers, Amygdalus communis leaves, Arnebia euchroma roots, and Scrophularia deserti stems showed healing activity in second-degree burn model in diabetic rats. When applied alone, PG extract was not able to close the wound during the 35 days of observation [22]. This difference in activity using the same pharmacogen and plant product can be justified due to the material collection location or influence of seasonality, since the amount and sometimes even the secondary metabolites' nature is not constant throughout the year. It is necessary to detect the conditions and times for cultivation or collection of plant raw material with desirable concentrations of active ingredients. Another alternative would be the investment in biotechnological production studies and genetic improvement of the plant instead of using wild plants, leading to obtaining uniform raw materials with quality control.
A combination of PG, honey and bee venom in scaffolds - Manuka Honey/ Pomegranate Peel Powder extract/Bee Venon (MH/PPP/BV) or Lyophilized Multiflora Honey/ Pomegranate Peel Powder extract/Bee Venon (LH/PPP/BV) or Manuka Honey/ Pomegranate Peel Powder extract (MH/PPP) - applied to an animal model of excisional wound showed complete healing on the tenth day of the experiment, compared to the treated and untreated control group that showed delayed healing and complete wound closure was observed only on days 13 and 14, respectively. Histological data showed that the MH/PPP/BV group had the best performance in terms of healing on days 5 and 10, followed by LH/PPP/BV and MH/PPP. All treatment groups provided reduction of the inflammatory phase, allowing the early formation of granulation tissue and epithelialization. In the MH/PPP/BV group, the wounds revealed accelerated healing with complete epithelium and minimal infiltration of inflammatory cells, with hair follicle growth [18].
Another association identified was a PG extract standardized with 40 % of ellagic acid (meaning that there is 40 % of ellagic acid and 60 % of non-ellagic acid content) used in two studies, in an animal burn model and another of excisional lesion. The first has histological characterization with high collagen density with good arrangement accompanied by complete and mature epithelium, low number of inflammatory cells - polymorphonucleate (PMN) - and angiogenesis [19]. The second is characterized by well-organized collagen deposits, a PMN count of <40, mild angiogenesis as well as moderate degree of fibrosis [20]. However, these studies did not have experimental groups that presented the substances separately, making it difficult to visualize synergism, antagonism or any difference in compound combination. The association of different natural products is used quite frequently, presenting itself as an alternative for the development of a formulation richer in bioactive compounds or even optimizing their distribution; however, the previous or concomitant identification of the action of the compounds separately should be performed to understand the role that each substance plays in the repair process.
PG extracts have demonstrated great healing activity in in vivo models, even in wounds with a tendency to more serious complications such as bacterial infections. Some authors have described antibacterial activity concomitant with healing, scaffolds composed of pomegranate/honey/bee venon, inhibited bacterial growth from 90-98 % to Staphylococcus aureus and Escherichia coli, while significantly increasing the percentage of wound closure on days 3, 5 and 10 of experimentation [18]. Other results using the plate diffusion method revealed that all formulations (pomegranate, henna, myrrh and the three mixed) showed excellent activity against Gram-negative bacteria such as E. coli ATCC 25218 (21 mm), Gram-positive bacteria, as S. aureus ATCC 29213 (20 mm), MRSA ATCC 29213 (20 mm), Bacillus subtilis ATCC 10400 (21 mm) and antifungal activity against Candida albicans ATCC 10231 (29 mm), although no formulation has demonstrated any activity against Pseudomonas aeruginosa ATCC 15442 [21].
In a second-degree burn model in non-diabetic rats, antibacterial results revealed that the minimum inhibitory concentrations for S. aureus, P. aeruginosa and E. coli were 6.25, 12.5 and 6.25 mg/mL. Regarding lesion repair, the skin burns of rats treated with PG showed reduction of inflammatory cells and appearance of fibroblasts and granulation tissue in the seventh day; on day fourteen, collagen fibers increased, inflammatory cells decreased strongly and many fibroblasts and granulation tissue appeared at the lesion site. On day 21, angiogenesis was observed and the presence of fibroblasts, inflammatory cells disappeared and were replaced by new granulation tissue, epithelialization progressed rapidly in PG-treated rats [15]. It is believed that the presence of tannin and flavonoid compounds, among others, may be responsible for the antibacterial effect for both Gram-positive and Gram-negative bacteria as well as the acceleration of tissue repair [32, 33].
In general, the mechanisms by which PG promotes the healing process acceleration are not fully understood. Huan et al., 2013 [16], verified the increase in hydroxyproline content, a marker that indicates an increase in the amount of collagen in wound tissue, while ELISA tests showed that the expression of TGF-ß1 was significantly higher than the control group on days 4, 7 and 14 post-injury; these results are confirmed by qRT-PCR. After the injury, there is an increase in TGF-ß1 levels released by platelets, which is a fundamental mediator of collagen synthesis and degradation as well as promotion of fibroblast migration and proliferation during wound healing.
In human studies, the pharmaceutical, solvent, healing model, study design, number of patients, interventions, treatment control group and main results were also described (Table 3).
Table 3 Human studies on Punica granatum used for wound treatment.
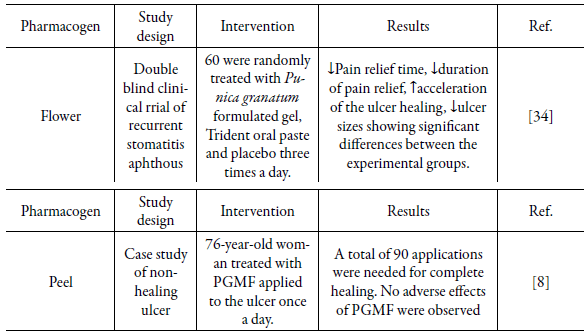
PGHF, P. granatum peels ethanolic hydrogel-based extract formulation; ↑, increase in mentioned variable; ↓, decrease in mentioned variable.
Research focused on using natural products has gained prominence in recent years, as it is believed that they can provide subsidies for developing new pharmacological agents with broad biological activity. However, there is still much to be known about the role of plants in wound treatment. In this regard, the main limitation is the scarcity of clinical studies in humans and the complexity in the identification of all chemical constituents.
Using PG in humans was identified in only two studies, a double-blind clinical trial of 60 patients with aphthous stomatitis, showing that the duration of ulcer healing in the treatment group was significantly shorter than in the placebo group and also in the control group. In this study, PG extract was prepared by a formulation with mucoadhesive gel that presented greater durability over ulcers. Moreover, the formulation of plant origin presented fewer side effects, being considered an effective substance in support treatment for controlling recurrent oral aphthous stomatitis [34].
Another human study, described as a case study, analyzed the effect produced by a masterful formulation of ethanolic extracts of PG peels in a chronic ulcer; despite rigorous medical measures and frequent use of topical treatments with antimicrobial agents, corticosteroids, debridement and ulcer dressing, a patient had a recurrent wound on the left leg with an area of 23.52 cm2. In six weeks, the ulcer decreased to a quarter of its original size and healed completely six weeks later after a total of 90 daily applications of masterful formulation on the wound. In this investigation, a formulation of ionic hydrogel with concentration of 2 % PG ethanolic extract presented an average punicalagin content of 17.9 mg. The authors believe that formulations containing no less than 0.49% of total punicalaginas, determined by liquid chromatography and calculated with reference to the masterful formulation may be effective in wound healing therapy [8].
Although many studies in experimental models demonstrate the healing activity of PG, there are few data available on the standardization and stability of these formulations. Clinical trials are still scarce and advances in animal studies are needed for human studies.
The probable mechanism of pomegranate action in tissue repair should be studied and clarified, toxicological and pharmacological studies focused on pharmacokinetics and bioavailability should be carried out to ensure the safety and efficacy of treatment and consequent development of a commercial drug. This identification of the healing activity of PG extracts in different in vivo models can contribute to implementing a protocol in future studies, in order to promote direction for researchers who wish to work with this natural product.
CONCLUSION
The mechanism by which Punica granatum extracts modulates the healing process is not fully understood, but the results suggest that it can act at all stages of healing with great repair potential. Further studies should be conducted to clarify the mechanism of action behind this biological activity, including the search for isolated substances that may be indicated as an active ingredient responsible for tissue repair. Human studies are still scarce, and more research is needed to ensure safety in its therapeutic application and in the development of new medicines.















