INTRODUCTION
Plants are exposed to different phytopathogenic agents and insects that could affect their normal development, to respond the attacks they have implemented local and systemic defense mechanisms such as structural barriers that could block the infection and colonization processes (Yadeta & Thomma, 2013). Additionally, plants continuously respond to complex interactions between biotic and abiotic factors through effectors, elicitors, and resistance (R) or avirulence (Avr) genes, their interaction could cause response to disease (Meng & Zhang, 2013; Gururani et al. 2012).
Tomato historically went from being a merely ornamental plant to being one of the most important vegetables today in terms of consumption and production (Abdallah et al. 2016), as well as, is a model organism to genetic and molecular studies related whit plant defense response (Andolfo et al. 2014). Vascular wilt is one of the most important diseases of tomato (Solanum lycopersicum L.) cultivation, the causative agent Fusarium oxysporum f. sp. lycopersici (Fol) (Sacc) W. C. Snyder and H. N. Hans, is present on all continents and distributed by races that make possible the presence of this and the disease in almost all regions of the world (Hernández-Martínez et al. 2014).
This review describes the economic importance of tomato cultivation in the economic, social and nutritional fields, its origin, distribution and its relationship with Fol, for which a comprehensive description is given of relevant aspects of the fungus such as morphology, races and explanation of the infection and colonization processes, as well as the known defense mechanisms that are used by the plant to respond to the infection. The knowledge and understanding of the tomato-Fol interaction will allow an integral vision even from the genetic part, which is essential to know and explore, especially with the isolates specific to the producing areas of the country to determine their true identity and lead new and more effective management strategies that reduce the losses caused by the pathogen, all this taking advantage of the fact that the tomato is a short cycle crop that allows studies and inferences to be made in a short period of time.
MATERIALS AND METHODS
A search for articles was carried out through the internet in scientific databases such as Google Academic, Dialnet, Jstor, Science Direct, PubMed and Academic Microsoft. “Fusarium oxysporum”, “Fol”, “tomato”, “Solanum lycopersicum”, “Lycopersicum sculentum”, “plant pathogen interaction”, “tomato interaction” and “tomato resistance” were used as the keywords to search titles, abstracts, and author keywords from 2005 to 2020. Author keyword and word cluster analysis were made using Microsoft Excel (version 2010). The Global Citation Scores (GCS) and Local Citation Scores (LCS) were acquired by using HistCite. The impact factor values from Journal Citation Reports (JCR) were also added for the corresponding identified journal titles.
RESULTS AND DISCUSSION
The best known and accepted model for understanding the interaction between plants and pathogens is the zig-zag model (Jones & Dangl, 2006); however new models have emerged such as the invasion model where immunity molecules are given a role beyond pathogenicity (Cook et al. 2015) and the multi-component model that includes, in addition to interaction, activation and modulation (Andolfo et al. 2016). However, the process of interaction between plants and pathogens is still in progress and is supported by new technological tools that facilitate understanding the complex process.
The host. The tomato (Solanum lycopersicum L.) is a dicot plant, belonging to the Solanaceae family which comprises more than 3000 species; among these, the tomato is one of the most cultivated vegetables in the world (Srinivas et al. 2019). Apparently, its origin is not well elucidated. For many years it was thought that the wild Cherry tomato, (S. lycopersicum var. cerasiforme), was the ancestor of the cultivated tomato (Cheema & Dhaliwal, 2005); however, it is clear today that the latter is a mixture of wild and cultivated tomatoes (Bergougnoux, 2014). In this regard, some authors indicate that it may have originated in Andes mountains, specifically in Ecuador, where a large yet unexplored genetic diversity is found (Morales et al. 2014). This vegetable has been a model of study in various areas such as physiology (Rodríguez-Ortega et al. 2019), biochemistry (Trong et al. 2019) and biotechnology (Ali et al. 2014; Murillo-Gómez et al. 2017); it has also been described as an agronomic model in genetic engineering and an important resource in tissue culture (Gerszberg et al. 2015).
It is also listed as one of the most important vegetables in terms of commercial use, cultivated areas, production and yield (Hernández-Martínez et al. 2014; Tampoare et al. 2012). In terms of consumption, it is only surpassed by potatoes (Bergougnoux, 2014), and it is also an important source of lycopene, beta-carotene, and flavonoids (Gerszberg et al. 2015). China is currently the main producer with 31% of world production and 20% of the total cultivated area (Rodríguez-Ortega et al. 2019). In Colombia, production in 2019 was 556,692 tons with a yield of 65,66t/ha with a total area 8,478ha (AGRONET, 2021).
A valuable resource that must be used intelligently is the wide genetic diversity of wild varieties; this plurality makes it very suitable for evolutionary studies and domestication processes, managing to identify characteristics of interest in the germplasm that, through crosses, can be introduced into commercial materials (Blanca et al. 2015; Ranjan et al. 2012).
In this regard, the authors of this review have participated in evaluations with different genotypes from the gene-bank of the Universidad Nacional de Colombia, which have been tested, evaluating aspects such as their genetic diversity (Ceballos-Aguirre et al. 2017), production and quality of the fruit (Ceballos-Aguirre & Vallejo, 2012; Herrera et al. 2015). Also, the characterization of pathogenic and non-pathogenic isolates of Fol isolated from commercial tomato crops in the Colombian Andean zone (Carmona et al. 2020), and field evaluation of tomato introductions produced via micrografting, which were also tested with a pathogenic strain of Fol race 2 have been our study topics.
Historically, based on the ability of wild tomatoes can cross with cultivated materials, a classification was established and were called: 1. Sculentum complex, that include its genotypes considered a source of resistance to biotic and abiotic factors due their ability to hybridize and, 2. Peruvianum complex who were recognized for their potential for crop improvement for its wide diversity. Later, through phylogenetic studies, it was divided into three groups in which the existence of the cultivated tomato and 12 wild relatives was recognized (Bergougnoux, 2014).
It is well known that available tomato varieties with a report of resistance to soil pathogen as Fusarium oxysporum do not show a successful response to local races (Palacio et al. 2014), so it is necessary to use the genetic resources as an alternative to improve the specie. In this way, the wild species of the Lycopersicum section constitute a specific case of genetic diversity that, due to its special characteristics such as resistance to pests and diseases, tolerance to drought, salinity, frost, better physical appearance of the fruits and high lycopene contents, are highly suitable for inclusion in breeding programs (Morales et al. 2014). In this group, according to Ceballos-Aguirre et al. (2017), S. lycopersicum var. ceraciforme and Solanum pimpinellifolium are the most promising due to the ease with which crosses are obtained and the wide variability of characteristics found in them.
A good example is the red Cherry tomato IAC391, belonging to S. lycopersicum var. cerasiforme, which highlights the morphological composition of the plant and its fruits (Agudelo et al. 2011), the organoleptic properties, quality standards of the fruits (Ceballos-Aguirre & Vallejo, 2012; Franco et al. 2018) and economic viability (Herrera et al. 2015).
The pathogen. The Fusarium genus has approximately 300 species (Rampersad, 2020) and Fol is included among the causative agents of the more than 200 diseases that affect tomato (Djidonou et al. 2016), alone or together with other pathogens whose natural habitat is the soil (Singh et al. 2017). The origin of Fusarium dates back to approximately 91.3 million years, which coincides with the appearance of woody and flowering plants and has been recovered from native soils in different parts of the world (Koyyappurath et al. 2015). The fungus species can be found in the tropics, temperate zones, desert zones, alpine and arctic zones where adverse climatic conditions prevail (Okungbowa & Shittu, 2014). It is an ascomycete facultative soilborne parasite (Sutherland et al. 2013), the most Fusarium species are harmless and abundant in the soil microbial community (Koyyappurath, 2015), and together with the pathogenic strains make up a complex of including the endophyte Fo47 used to prevent vascular wilt in tomato caused by Fol (Wang et al. 2020).
Some species within the Fusarium genus produce meiotic spores and also three types of asexual spores that have nuclei derived mitotically from the hyphae (Gordon, 2017), microconidia, macroconidia and chlamydospores; however, not all species produce the three types of spores and less than 20% have known sexual cycle (Ma et al. 2013).
The anamorphic state of the fungus is made from macroconidia derived from producer cells called phialides, which in turn are grouped into a pad-shaped structure called sporodochium (Kant et al. 2011). Microconidia are generally uninucleated, 5-12µm long by 2.5-3.5µm wide and germination ranges from 1% to 20%. Macroconidia are multinucleate, translucent, canoe-shaped. They have from 3 to 5 septa (Kant et al. 2011), a conical apical cell and the basal cell with a foot-shaped end; they measure from 27 to 46μm in length by 3-4.5μm in width, and their germination is rapid, which is an advantage for the spread of the pathogen (Leslie & Summerell, 2006). Chlamydospores are spores that result from the modification of hyphal segments and have thick cell walls; their main function is soil survival, and they can occur in isolation, in pairs, in clusters or in chains. According to the position they occupy in the hypha, they can be: intercalary, if they are inside the hypha; sessile, or terminal, when they are at its end. They are also characterized by their high resistance to unfavorable environments (Leslie & Summerell, 2006) and can be kept alive indefinitely in infested soils, even in the absence of a host (Khan et al. 2017).
The life cycle in Fusarium species can be divided into three stages: latent, which includes the inhibition and subsequent germination of resistance structures; parasitic, by which the pathogen penetrates, colonizes, moves through the xylem, induces the appearance of symptoms and causes the death of the host, and the saprophytic stage characterized by the formation of new resting structures on the residues of the dead host (Okungbowa & Shittu, 2014).
Contrary to non-pathogenic ones, the pathogenic strains of Fusarium oxysporum have the ability to penetrate the root and cause tracheomycosis by invading the vascular tissue of the host plants (Koyyappurath, 2015), causing the vascular wilt (Andolfo et al. 2014), as well as crusts, blight, cankers and rot stem or fruit (Rampersad, 2020; Edel-Hermann & Lecomte, 2019; Okungbowa & Shittu, 2014). Additionally, some species of the genus produce mycotoxins such as fumonisins, fusaric acid and trichothecenes that facilitate the host invasion process, especially in cereals, with important effects on human and animal health (Koyyappurath, 2015).
The F. oxysporum species are morphologically diverse (Botero et al. 2018) and, due to its economic and scientific importance, it ranks fifth among fungal pathogens (Dean et al. 2012); additionally, the range of action is very wide and includes plants, animals, arthropods and humans (Koyyappurath, 2015). It is characterized by being a filamentous, hyaline fungus, with asexual reproduction, and has a great agricultural impact due to the formae speciales (f. sp.) reported (Gordon, 2017), which have the ability to attack different types of plants with high economic value (Agrios, 2013; Dean et al. 2012), located in 73 botanical families (Edel-Hermann & Lecomte, 2019).
The F. oxysporum strains show different forms of growth in the host tissues, classified as biotrophic and necrotrophic, which is determined by the type of colonization, which can be inter or intracellular, resulting in the death or not of cells adjacent to the infection (Gordon, 2017). However, most can be classified as hemibiotrophic since the initial infection resembles that of a pathogen that depends on a living host (Ma et al. 2013), but it has also been shown that entry to xylem, as in the particular case of F. oxysporum f. sp. ciceris does not necessarily result in expression of symptoms, the latter depending on the virulence of the pathogen and the level of resistance of the host (Jiménez-Fernández et al. 2013).
Taking these two types of strategies into account, it is possible to classify Fol as hemibiotrophic, since it is reported that this type of microorganism initially, in the biotrophic phase, evades the recognition of the host; later, in the necrotrophic stage, they release the toxins and it is at that moment where the symptoms are noticeable, hence pathogens of this type are difficult to manage since the plants are initially asymptomatic (Ávila & Romero, 2017; McGovern, 2015). This transition is conditioned by environmental and metabolic signals (Rampersad, 2020).
The fungus is a morphospecies that is divided into specialized groups, pathotypes, or formae speciales due to the degree of pathogenicity that they exhibit between varieties of the same plant species (Sutherland et al. 2013). Currently, around 150 highly specific are referenced (Koyyappurath et al. 2015), morphologically identical and that together constitute a complex (Botero et al. 2018); however, Edel-Hermann & Lecomte (2019) found 106 formae speciales well documented and characterized, 53 of them are only associated with specific plants species.
The selectivity of the formae speciales is due to that only the host plants and their radical exudates supply the necessary nutrients for the growth and development of the fungus. Likewise, the fungus is subdivided into races according to its ability to infect cultivars with different levels of resistance, a fact that is associated with mutations in avirulence genes and horizontal gene transfer (Sutherland et al. 2013). Additionally, the evolution of the pathogenicity has been acquired through the horizontal chromosome transfer of non-pathogenic strains (Li et al. 2020a; Inami et al. 2014). Genetically, the physiological races are determined by the possession of different combinations of SIX proteins (Secreted In Xylem). Specially during the plant colonization by Fol 14 of SIX proteins are working (Li et al. 2020b).
Of the formae speciales of the fungus that attack tomato crops, Fol is the most limiting because it causes vascular wilt. Three physiological races are known: 1, 2 and 3; differentiating each other by their pathogenicity towards various tomato cultivars. Races 1 and 2 have global dispersion in the producing areas and most cultivars are resistant to them, while race 3 has been detected in California, Australia, Georgia, Brazil and Mexico, and few materials are known to have resistance to it (Biju et al. 2017).
The disease. Called vascular wilt, it is the most limiting pathology in tomato production (Akrami & Yousefi, 2015). Some authors consider it a monocyclic disease; however, the appearance of symptoms and the progress of the disease varies in the field, so the appearance of a secondary infection is common (Okungbowa & Shittu, 2014). Other authors mention that tillage practices, agricultural equipment, floods or rains and the dissemination at short distances of some macroconidia formed on plant residues can promote the spread of the fungus, hence it is considered a polycyclic disease in this case (Okungbowa & Shittu, 2014). It affects the quality of the fruits and decreases the yield between 50% and 60% (Báez-Valdez et al. 2010) and, when the plant withers, the loss can reach 100% for susceptible varieties when the soil conditions and temperatures are high during most of the crop cycle (Agrios, 2005).
Plants are susceptible at all ages and the characteristic symptoms are wilting, partial yellowing of leaves, folding and dwarfing of shoots, decrease of the general growth of the plants, accompanied by a coloration that can vary from whitish, yellowish or brown of the vascular bundles (Botero et al. 2018). Also, especially in conditions of high humidity, a white, pink or orange growth of the fungus can appear on the outside of the affected stems (Okungbowa & Shittu, 2014). Foliar chlorosis begins in one sector of the plant, progressively increasing throughout it until it causes wilting and subsequent death; fruit production becomes scarce or not inexistent. At the base and internal part of the stem, without covering the medulla, a dark brown coloration is observed (Báez-Valdez et al. 2010; Okungbowa & Shittu, 2014), and occasionally adventitious roots are formed (Agrios, 2005).
The toxins produced by the fungus are fusaric acid, lycomarasmin and dehydrofusaric acid (Srinivas et al. 2019); in tomato, the effects of fusaric acid are associated with reduced photosynthesis, lipid peroxidation, wilt, leaf necrosis, cell death (Singh et al. 2017), decrease in cell viability and improvement in fungal capacity (Srinivas et al. 2019). Also, the tomatinase, produced by Fusarium oxysporum f. sp. lycopersici is an enzyme recently identified as a virulence factor involved in the degradation of tomato defense compound tomatine (Murugan et al. 2020). Severely infected plants die and release the chlamydospores to the soil, where they remain for long periods of time; the cycle repeats when they have the optimal conditions for their germination and invade tissue from a susceptible host again (Agrios, 2005).
This type of symptomatic response is not always noticeable, especially in field conditions where inoculated plants respond very differently, being influenced by climatic, edaphic and physiological factors that cannot be controlled as strictly as in the laboratory or greenhouse, even being inoculated with high concentrations of the pathogen, a fact that can be related to what Van der Does et al. (2019) report, who found that Fol, even under an immune response, can grow as an endophyte and colonize the tissues of tomato plants without showing any symptoms.
Another fact that confirms the importance of making a correct and thorough identification of the isolates typical of an area or country are the results obtained by Carmona et al. (2020), when evaluating 32 producing farms in the Andean zone of Colombia where plantations presented symptoms of wilt and vascular necrosis initially attributed to Fol. However, the results showed that out of 120 isolates, only two were positive and virulent. These findings show that the presence of Fol in the tomato farms is possibly being considered true, and even more so, cataloged as the causative agent of the disease due to the macroscopic symptoms only and even the morphology of the colonies and reproductive structures, ignoring that other pathogens that induce similar symptoms are common in producing areas.
Many farmers use plant material that, according to seeds producers, is tolerant to a wide range of pathogens; however, many of these materials are not of national origin and when exposed to local conditions, do not behave as expected. Therefore, it is necessary to carry out research work with infection methodologies in field conditions to evaluate the interaction between Fol and different tomato varieties and identify determining factors such as plant genotype, the amount of inoculum of the pathogen, damage to roots, and plant age.
Interaction tomato-Fol. Understanding the defense and resistance mechanisms of plants against pathogens is mediated by the interaction of both organisms, for which resistance (R) and avirulence genes (Avr) have been extensively studied (Meng & Zhang, 2013; Guan et al. 2012; Gururani et al. 2012). To better comprehend this concept, we must remember the initially undervalued gene-by-gene theory of Harold Flor, which states that for every R gene in the host there is an Avr gene in the pathogen, which means that there is a close relationship and a specific recognition between these components of the two organisms and, depending on the evolution and specificity of each, the result can be disease or immunity.
Thus, for the infection to be successful and the disease to occur, the pathogen must go through different processes, from the location of the host and evasion of the defense responses of the plant, to the germination of spores and systemic infection (García-Enciso et al. 2017). This recognition originates through highly conserved molecules called elicitors, which are responsible for activating the host's defense responses (Muthamilarasan & Prasad, 2013; Meng & Zhang, 2013).
Elicitors involved in pathogenesis processes are regulated in the plant by the perception of microbe or pathogen associated molecular patterns (MAMPs and PAMPS), allowing the basal response to be triggered (García-Enciso et al. 2017) by activating specific transcriptional factors of the response through a cascade of kinases (Jones & Dangl, 2006; Boller & He, 2009). This recognition induces a mechanism in plants called PAMP-triggered immunity or PTI, allowing to enable different response pathways against microorganisms in general (Jones & Dangl, 2006), among these the activation of proteins such as MAPKs (mitogen-activated protein kinase) and CDPKs (calcium-dependent protein kinases) that cause early defense reactions and include stomatal closure, ethylene and salicylic acid production, Ca+2 mobilization and phytoalexin production (Couto & Zipfel, 2016; Li et al. 2016), (Figure 1).
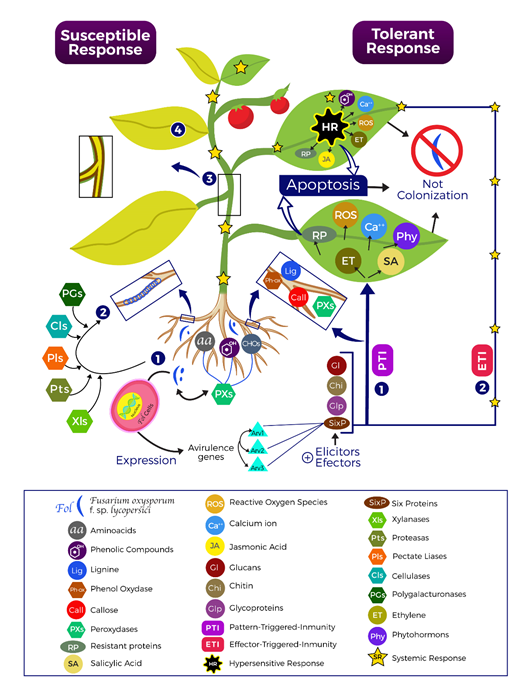
Figure 1 Tomato and Fol interaction. Under field conditions it is normal to have tomato plants in soil contaminated with Fol . As part of the growing process of the plants, they release molecules such as phenolic compounds, amino acids (aa), carbohydrates (CHOs) and reactive oxygen species (ROS), those are recognized by Fol through peroxidases (PXs) and activate their virulence mechanisms (1), which lead to the germination of spores on the plant to induce hyphal formation. Then, (2) through the production of cellulases (Cls), polygalacturonases (PGs), xylanases (Xls) and proteases (Pts) the cell wall of the plant is deraded, achieving its penetration and colonization in the vascular bundles (3), causing the infection system and the development of the disease through symptoms like yellowing and wilt (4). The plant, for its part, when it detects the elicitors and effectors released by Fol activates its PTI and ETI defense responses, to secret molecules like hormones, ions, and proteins. In addition, is induces stomatal closure, cell wall hardening, hypersensitive response (HR); all those strategies conducting cell death to avoid the Fol colonization.
In tomato, phytoalexin α-tomatin is known to induce programmed cell death by activating signaling pathways through tyrosine kinase and protein G, causing intracellular accumulation of Ca++ and reactive oxygen species (ROS) such as hydrogen peroxide (H2O2) (González et al. 2012). Among other defense mechanisms are the accumulation of NB-LRR proteins, compounds in the cell wall such as callosum, lignification, the production of polyphenol oxidases and peroxidases that help block the action of the pathogen (Andersen et al. 2018; Eitas & Dangl, 2010) (Figure 1).
A second defense pathway called Effector-Triggered Immunity (ETI) occurs in response to virulence and pathogenesis factors (García-Enciso et al. 2017). This is a specific mechanism for a type of pathogen stronger than PTI and culminating in a hypersensitive response (HR), which leads to the death of the infected cell thus delaying the pathogenic process (Jones & Dangl, 2006); this response is presented when the penetration of the cell wall by the pathogen occurs causing the movement of the nucleus in the plant cell, the increase in cyclosis, the rearrangement of the microtubules of the cytoskeleton, the breakdown of DNA, the production of phenolic compounds and reactive oxygen species (ROS), deterioration of organelles and subsequent cell death (Figure 1).
Simultaneously, chemical signals such as jasmonic acid, ethylene, and phytohormones are released, which activate other defense processes, a mechanism that allows the plant to block the passage of nutrients to the affected area, encapsulate the pathogen and trigger cell death, thus allowing the limitation of the area of action of the attacker. This response is used by the plant to stop hemibiotrophic organisms but it is not effective with necrotrophic organisms since they can grow on dead tissue and thus spread the infection (Camagna & Takemoto, 2018) (Figure 1).
For this reason, the hypersensitive response in the tomato-Fol pathosystem is used differently and, in fact, the gene that prevents the signaling of cellular apoptosis is related to resistance, since this mechanism would favor infectious processes due to the necrotrophic nature of the fungus (Essarioui et al. 2016), preventing it from killing the cells to obtain food (Andolfo et al. 2014). Therefore, recognition through PTI in pathogens such as F. oxysporum, which have the ability to completely destroy tissue during infection, is not effective (Jones & Dangl, 2006), because the fungus produces effector molecules that facilitate cell death and prevent recognition by specific pathogen recognition proteins (PRRs), thus blocking PTI and initiating colonization in the plant (Muthamilarasan & Prasad, 2013; Meng & Zhang, 2013) (Figure 1).
For its part, Fol has the ability to perceive the location of tomato plants due to the release of amino acids and the peroxidase activity of the roots, which is regulated by proteins TMP1, TMP2 and CEVI1, activities perceived by the hyphae of the fungus. In this chemiotropic response, the importance of mitogen-activated protein kinases (MAPKs) has been highlighted for their crucial role in activating the pathway that results in gene expression and transcription to regulate the process of infection and development of the disease, through the expression of pathogenicity, infectious growth and fixation to the roots of the host (Hamel et al. 2012).
Other studies in Fol have shown that physiological and developmental processes are regulated by three signaling pathways, each of which has a specific function involving virulence and hyphal fusion, cell wall characteristics, osmoregulation responses and response to stress (Segorbe et al. 2017). Indeed, it has been documented that the tomato infection process begins with the germination of Fol spores due to the accumulation of exudates rich in amino acids, sugars, phenols, proteins and carbohydrates produced by the roots, and the branching of the germ tube leading to the formation of hyphae (García-Enciso et al. 2017). Next, the fungus, using an enzymatic complex that involves the secretion of enzymes such as cellulases, polygaracturonidases (PGs), pectatoliases (PLs), xylanases and proteases, degrades the cell wall allowing penetration and subsequent colonization (Srinivas et al. 2019) such that, as soon as the root hypodermis has been invaded by the pathogen, it locates in the cortex.
During this process, mycotoxins are also released in the host, activating defense mechanisms in the plant that include the production of physical barriers, the proliferation of parenchymal cells; activation of the jasmonic acid pathway and the release of signaling hormones (González et al. 2012; Ignjatov et al. 2012; Ortiz et al. 2014), Due to the interaction between resistant tomato plants and Fol, the overexpression of more than 2000 genes has been reported, which are linked to the maintenance of cellular structures and cellular homeostasis and 14 auxin-binding genes, important as phytoregulators (Andolfo et al. 2014). Likewise, the plant induces chemical changes that are focused on strengthening the cell wall, increasing the concentration of phenolic compounds and releasing antimicrobial aggregates, as well as the deposition of calluses, gels and the formation of tyloses (Srinivas et al. 2019) (Figure 1).
Furthermore, various combinations of R and Avr genes have been identified in tomato, but only three of those immunity genes, called I, I2, I3, have been introduced from wild tomato relatives to commercial cultivars and confer resistance against Fol (Van der Does et al. 2019). These genes encode proteins that recognize the effector proteins produced by the different races of the fungus in the plant xylem, which are called SIX, as in “Secreted in the xylem” (Inami et al. 2014) and are encoded by an avirulence gene; SIX-1 (Secreted in Xylema-1) encoded by the Avr3 gene, the SIX-3 protein encoded by the Avr2 gene, and the SIX-4 effector protein, encoded from the Avr1 gene. The SIX-1 and SIX-3 proteins are necessary for the complete virulence of the pathogen; in this regard, Selim et al. (2015), mention that the SIX1 protein is rich in cysteine and has been shown to be indispensable for the virulence of Fol. In Fol, the avirulence genes are carried according to the race of the pathogen: Avr 1 is exclusive to race 1, Avr 2 is found in races 1 and 2 and Avr 3 is present in all races (Andolfo et al. 2014).
In addition, effectors Avr2 and Avr3 are involved in the ETI response, which are expressed during colonization of the pathogen from the root to the xylem of tomato plants where they are recognized by resistance genes I2 (Immunity 2) and I3, respectively. Effector gene Avr1 is not recognized by the R genes in tomato plants with I2 and I3 favoring colonization in the xylem, until causing vascular wilt, for which it has been indicated that Avr1 suppresses the resistance mediated by I2 and I3 (Van der Does et al. 2019).
CONCLUSION
It is important to isolate and identify races of endemic F. oxysporum f. sp. lycopersici present in commercial tomato crops in Colombia, of which there are no reports, performing morphological studies and relying on the use of molecular and biotechnological tools that generate precise and forceful information. This should be done in such a way that this information allows us to understand the interaction between Fol and tomato, in order to demonstrate local loss percentages and thus focus on effective integrated management programs that include plant genetic improvement supported by biotechnological tools such as micrografting, transgenesis and genomic editing, which contribute to providing improved materials that can be incorporated in sustainable production systems while minimizing economic losses. Finally, it is vitally important to intelligently take advantage of the reserves of wild genetic materials, which are mostly unexplored and without any agronomic support in genebanks, which constitute a tool essential for the integrated management of vascular wilt.















