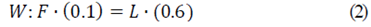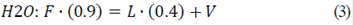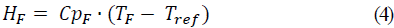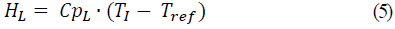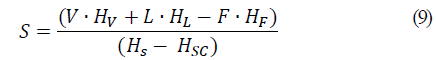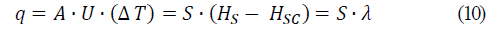1. Introduction
In pproduction of fresh beverages, such aguamiel, or concentrated products from these drinks (syrup), the use of evaporator system is the main step of the process, which consists of the use of evaporators for the concentration of foods similar to those used to produce apple jam from fruit concentration [1]. Studies such as standardization of the process to obtain agave syrup [2] have opened the doors for the production of this syrup with good commercial and industrial potential.
The hypothesis, through a predictive model using the RSM methodology, is possible to establish the optimal conditions of the process, and that use of an evaporator of simple effect presents advantages for increasing the production of agave syrup and will allow us to reach the required concentration of solids. An evaporator is a piece of equipment in which, through the action of heat, there are phase changes and phenomena associated with the concentration of a solution, such as the formation of solids and decomposition of certain substances [3].
A strategy for the design of the simple-effect evaporator is the use of response surface methodology (RSM), in which the design variables are evaluated to determine the optimal operating values. RSM is widely used in chemical engineering, biology, and other fields of science and technology [4,5] and employs the design of experiments and the development of predictive models for simulation and optimization of processes by characterizing system responses. Based on this background, the objective of the present study was to determine, by a predictive model using RSM methodology, the optimal operating conditions of a simple effect evaporator with long vertical tubes to produce agave syrup.
2. Materials and methods
2.1 Simple effect evaporator
The simple effect evaporator for the agave syrup production process used in this study is shown in Fig. 1, where F is the flow feed of fresh aguamiel, TF is the temperature at which fresh aguamiel is feed to the simple effect evaporator, W is the solid composition of aguamiel, H2O is the moisture composition, V is the mass flow of water vapor leaving the simple effect evaporator, L is the mass flow of the concentrate, S is the mass flow of the saturated water vapor fed to the evaporator, TL is the temperature at which the evaporator concentrate comes out, TS is the temperature of saturated water vapor that is fed to the evaporator, HS is the enthalpy of saturated water vapor that enters the evaporator of simple effect to the temperature of TS, HSC is the enthalpy of the steam that comes out condensed at the temperature of TS. The evaporator shall operate at a pressure of 101,325 kPa on the outside and 75 kPa on the space of the evaporator, after being considered a filtration step to remove impurities from fresh aguamiel, as well as traces of maguey leaves (pencas) or insects.
2.2. Response Surface Methodology (RSM)
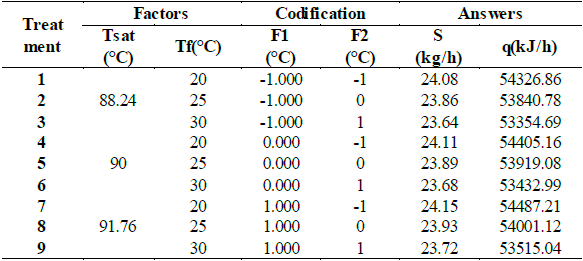
The optimization of evaporation process parameters was carried out using a prediction control model, which allows predicting the effect of the action of the control variables on the response of the process. RSM was used for establishing a series of experiments for predictions of a response y, fitting the model to determine the optimal conditions in the input variables. The factors that were considered in the RSM analysis are saturation temperature inside the evaporator (F1) and feed temperature (F2). Parametric optimization is done through RSM and Excel solver (Table 1).
Table 1 Saturation and feeding temperatures for agave syrup production.

Source: Prepared by the authors.
The responses to be optimized were the flow required for the saturated steam entering the evaporator, S, (kg/h), and the heat required for the operation of the evaporator, q, (kJ/h). These data are analyzed by means of RSM and using a polynomial model associated with the experimental design:
The Excel Solver was used as a calculation tool to obtain the required optimal values and variables of the predictive model B0, B1, B2, B11, B22, B12, SSR, R^2 (Table 2).
2.3. Mass balance for production of agave syrup
The chemical composition and physicochemical characteristics of the fresh aguamiel were taken from the mean values reported by Castro et al [6] considering 10% solids (W) and 90% humidity (H2O) content, and for agave syrup, 60% of total soluble solids, data generated from aguamiel, and agave syrup obtained in the ejido "Las Mangas" Municipality of Saltillo Coahuila Mexico.
The general equation for the realization of global mass balances was:
2.3. 1Global Mass Balanc
Where F is fresh aguamiel feed flow (25 kg/h), V is the mass flow of saturated steam leaving the evaporator for a simple effect, and L is the mass flow of the concentrate.
2.3.2 Balance sheets by component
Knowing the percentage of solids in the composition of fresh aguamiel (10%) and the final percentage of solids in agave syrup (60%), the mass flow value of agave syrup (L) was determined according to Eqs. 1, 2, and 3, to determine the massive flow of the input and output currents. The value of the mass flow of saturated steam (V) was calculated later, since in that the current has a percentage of 100% humidity that is to say that it only has water vapor.
2.4. Energy balance for production of agave syrup
The energy balance was used to determine the heat that is required within the simple effect evaporator, and the value of the mass flow of saturated water vapor that is needed, as well as the value of condensed water vapor this is due to the enthalpy of the saturated water vapor entering the equipment and enthalpy of the condensed steam coming out, according to input and output definitions in Fig. 1.
For calculation of enthalpy, HF and HL, enthalpy of fresh aguamiel feed and concentrate, respectively, a temperature of 0 °C was used for water, and temperature of fresh aguamiel was 20 °C. The equations for calculation of aguamiel enthalpy at the entrance, and concentrate at the process end were as follows:
Where: Tref is the temperature at 0°C with respect to water, CpF is the heat capacity of the feed and CpL the heat capacity of the concentrate.
The saturated water vapor inputs required by the evaporator (S) were calculated in the energy balance, as well as the condensed steam output (SC). It was required to calculate enthalpy values of the water vapor at the operating pressure and enthalpy of fresh aguamiel feed and the concentrate, HF, and HL, respectively, using the equation of the heat capacity that is a function of the percentage of humidity (Eqs. 4 and 5).
Table 3 shows properties of water vapor at a pressure of 101,325 kPa on the outside of the evaporator and 75 kPa on the space of the evaporator [7], these data allow to determine the enthalpy of saturated water vapor at the entrance to the equipment (HS), enthalpy of saturated steam leaving the equipment (HV) and enthalpy of the condensed steam coming out (HSC).
Eqs. 6, 7, and 8 were applied for the calculation of the saturated water vapor required by the evaporator and the transfer heat in the equipment.
Isolating the water vapor required by the evaporator, S, the following equation is obtained.
Where A is the heat transfer area of the equipment, U is the heat transfer coefficient and λ corresponds to the latent heat condensation of the water vapor.
3. Results
3.1 Response Surface Methodology RSM
The analysis by RSM allowed for the observation of the interaction of the evaluated parameters, where all the factors were varied simultaneously over a set of experimental executions to establish the optimal operating conditions of the simple effect evaporator. The responses to optimize, either maximize or minimize the value, was the flow required for the saturated steam entering the evaporator (S, kg/h) and the heat required for the operation of the evaporator (q, kJ/h). It was determined the predictive model to obtain the optimal values for the model of saturated steam entering the evaporator (S, kg/h) shown in Table 4, Where B0, B1, B2, B11, B22, and B12 are the model variables according to their linear or quadratic contribution, respectively SSR is the sum of squares due to regression.
Table 4 Predictive model parameters for saturated water vapor required by the evaporator.

Source: Prepared by the authors.
Table 5 shows the behavior of the predictive model using , R1= B0 + B1*F1+B2*F2+B11*F12+B22*F22+B12*F1*F2 such as the model equation for saturated steam entering the evaporator (S, kg/h) used for nonlinear regression of R1. F1 is temperature of the saturated water vapor inside the evaporator, F2 is feed temperature of the raw material, R1 is mass flow of saturated water vapor required by the evaporator and S(Rmodel)is the R1 calculated using the predictive model with which the adjustment of the data is 1, so that the parameters are reliable.
Table 5 Predictive model for the flow required for saturated water vapor entering the evaporator.
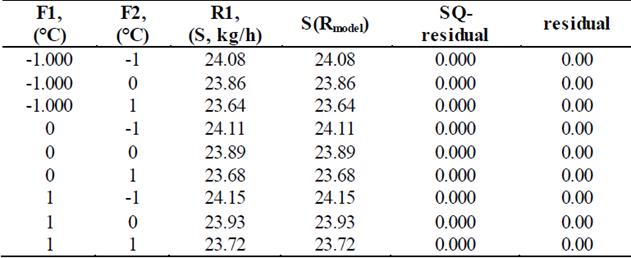
Source: Prepared by the authors.
Table 6 shows the optimal values for maximum and minimum operating conditions in the saturated water vapor response entering the evaporator according to the selected factors. F1 is the temperature of the saturated water vapor inside the evaporator, F2 is the feed temperature of the raw material, R1 is the mass flow of saturated water vapor required by the evaporator.
Table 6 Maximum and minimum operating conditions for saturated water vapour entering the evaporator.

Source: Prepared by the authors.
These results allow us to establish what conditions must be considered to reducing the consumption of saturated water vapor required by the evaporator.
Solving the equation with RSM methodology yields the parameters for the heat equation required for the operation of the evaporator (q, kJ/h) (Table 7). Where B0, B1, B2, B11, B22, and B12 are the model variables according to their linear or quadratic contribution, respectively, SSR is the sum of squares due to regression.
Table 7 Predictive model parameters for the heat required for evaporator operation.

Source: Prepared by the authors.
Table 8 shows the behavior of the predictive model using, R2= B0 + B1*F1+B2*F2+B11*F12+B22*F22+B12*F1*F2 as a model equation for the heat required for evaporator operation (q, kJ/h) used for nonlinear regression of R2. Where: F1 is temperature of the saturated water vapor inside the evaporator, F2 is feed temperature of the raw material, R2 is transfer heat in the evaporator and q(Rmodel)is R2 calculated using the predictive model with which we can see that the adjustment of the data is 1, so that the parameters are reliable.
Table 8 Predictive model for the flow required for saturated water vapor entering the evaporator.

Source: Prepared by the authors.
Table 9 presents the optimal values for maximum and minimum operating conditions in the heat response required for the operation of the evaporator according to the selected factors. According to this, it is possible to estimate the conditions that should be considered decreasing heat supply to the evaporator. Where: F1 is temperature of saturated water vapor inside the evaporator, F2 is feed temperature of the raw material, R2 is heat required by the evaporator.
3.2 Mass and energy balances
At this step, the maximum operating conditions for R1 and R2, presented in Tables 6 and 9, respectively, were used. Global mass balance was calculated by Eq. 1 where F = 25 kg/h is the fresh aguamiel feed.
From the individual balances, obtained with the equations 2 and 3, it was possible to determine values of concentrate flow, L = 4.17 kg/h and water vapor, V = 20.83 kg/h, having a ratio of 1:6 with respect to the feed and the concentrated product.
The equations used for the calculation of aguamiel enthalpies at the beginning and concentrate at the end of the process equations 4 and 5 were developed using the equation of Cpi = 0.0273*(%H ) + 1.4317 to obtain CpF = 3.89 kJ/kg °C and HF = 77.77 kJ/kg at the input and, CpL = 2.52 kJ/kg °C and HL = 231.57 kJ/kg at the output, respectively, according to studies of the heat capacity (Cp) (kJ kg-1 °C-1) for Colombian honeys from Boyacá, defining the function Cp = 0.0273*(%H ) + 1.4317 for a humidity range between 20-45%, for honey crystals [7].
3.3 Energy balance (Heat):
The equations necessary for the calculation of the water vapor required by the evaporator and transferred heat in the equipment were equations 6, 7, 8, 9 and 10. Considering S as the massive flow of water vapor required by the evaporator, S = 24.15 kg/h, and the transfer heat required by the process was q = 54487.21 kJ/h (15135.34 Watts).
4. Discussion
According to predictive models, the model equation used for the nonlinear regression of R1 and R2 is adequate with the parameters obtained, therefore using the RSM methodology of response surface the optimal values of the conditions for F1 and F2 with which the maximum and minimum results can be achieved were obtained. The inlet and outlet flow of the evaporator of simple effect at conditions of a pressure of 75 kPa inside and 101,325 kPa outside, with a temperature of 20 ° C in the feeding and a heat of 54487.21 kJ / h, are F = 25 kg / h, S = 24.15 kg / h and V = 20.83 kg / h, L = 4.17 kg / h, respectively.
The evaporation process is affected by different parameters such as saturation temperature at which the saturated vapor required by the evaporator is fed, the temperature inside the evaporator and the feed temperature.
In Tables 5 and 8, it is observed that to achieve a lower consumption of saturated water vapor and heat supplied to the evaporator, according to the results obtained, indicate that operating conditions to reduce consumption of saturated water vapor and heat supplied to the evaporator are 88.24 ° C inside the evaporator and 30 ° C the aguamiel, achieving an agave syrup production of 4.17 kg / h by feeding 25 kg / h of aguamiel. A evaluation of thee results with those reported by González Montemayor et al, (2020) [2], it can be seen the impact of temperature during the process of making agave honey also affects processing time, that using the maximum operating conditions would reduce the processing time but a greater consumption of saturated water vapor, supplied heat would be obtained.
5. Conclusion
The present study shows how the temperature inside the evaporator and the temperature at which the aguamiel is fed can affect the consumption of saturated water vapor and heat supplied to the evaporator. The response surface methodology (RSM) is one viable method for developing, improving, and optimizing the response variable. The methodology used allowed to identify the operational considerations to reduce consumption of saturated water vapor and heat supplied to the evaporator.
The mass and energy balances allow establishing that the theoretical expectations of founding, through a predictive model, the optimal operating conditions of a single-effect evaporator were met, since the predictive model proved to be an efficient tool in the systematic control of the operating parameters.