1. INTRODUCTION
The coordination bond that boron atoms form with nitrogen atoms, especially cyclic ethers formed by alkanolamines, increase the water and temperature resistance of the boron containing organic compounds.
Boron atoms adsorbed on the surface of the metal form an isolated layer with it, while also overcoming the catalytic effects of metal in the oxidation process. With an increase in temperature B2O3, Fe3B2 and BN layers are formed. It improved the tribiological properties of the additives and improved its oxidation and corrosion properties.
Existence of  fragments in nitrogen-containing com-pounds, for example: in borated succinimides and amines, increasing the temperature resistance and at the same time detergent-dispersive properties. In order to increase the dissolving of boron compounds in oil and effectiveness of a complex of boron atoms with nitrogen-containing organic compounds, boron ethers are used at the same time. [1]-[6].
fragments in nitrogen-containing com-pounds, for example: in borated succinimides and amines, increasing the temperature resistance and at the same time detergent-dispersive properties. In order to increase the dissolving of boron compounds in oil and effectiveness of a complex of boron atoms with nitrogen-containing organic compounds, boron ethers are used at the same time. [1]-[6].
Due to this,it is possible that their exploitation features would be increased when embedding the boron atom in the nitrogen molecule containing alkylphenolate additives.
2. EXPERIMENTAL DEVELOPMENT
Obtaining the synthesized alkylphenolate typed boron-containing additives was carried out in the following steps:
1. Condensation of alkylphenol or methylene-bis-alkylphenol with formaldehyde or para-form and amine compounds (ammonia, octadecylamines, mono-and diethanolamines, alkenyl-succinimids). Synthesized on Mannich base to be controlled for ester variant by refraction ratio of mixture.
2. Processing of the condensation product with boric acid; (6.6 8.0% due to alkylphenol upon provided additives)
3. Neutralization of the obtained product with calcium hydroxide. (18-20% due to alkylphenol (weight part) upon provided additives).Diluent oil is diluent I-12A. Each additive is released from the water, getting dried, centrifuged for cleaning from impurities and thinners (benzole, heptane). Within the framework of the aforementioned steps, we have obtained boron-nitrogen-containing alkylphenolate additives (not known to literature) with the formulas indicated below [7]-[9].
AKI-114B
Calcium salt from the product of condensation of alkylphenolate with formaldehyde and ammonia and processing with boric acid;
AKI-212B
Calcium salt from the product of condensation of alkylphenolate with formaldehyde and octadecyl amines and processing with boric acid;
AKI-115B
Calcium salt of the product of condensation of methylene-bis-alkylphenol with formaldehyde and mono ethanolamine and processing with boric acid;
AKI-210B
Calcium salt from the product of condensation of methylene-bis-alkylphenol with formaldehyde and diethanolamine and processing with boric acid;
AKI-214B
Calcium salt from the product of condensation of methylene-bis-alkylphenol with alkenyl- succinimide and paraform and processing with boric acid.
AKI-219B
Carbonated calcium salt from the product of condensation of methylene-bis-alkylphenol with diethanolamine and formaldehyde and processing with boric acid.
This additive is a carbonated variant of AKI-210B additive. The feed rate of CO2 gas during carbonatation is 15-16 ml/min at the temperature 70-800C.
The characterization of physicochemicaL and functional properties of boron nitrogen containing additives is provided in Table 1. R-spectrums were recorded using a Fourier-Nicolet ¡S-10 IR Spectrometer, made in the United States of America
Table 1 Physicochemical and functional properties of boron-nitrogen-containing alkylphenolate additives.

Thermooxidation properties of synthesized alkylphenolate additives have been studied by derivatographic analysis at Of 0Д-102T Paulik (Hungary) typed derivatograph with dynamic working mode at air conditions and a temperature of 10SC. As a sample, 0.1g of standard aluminum oxide is used.
3. RESULTS AND ANALYSIS
It is clear from Table 1 that boron-nitrogen-containing additives are significantly better (0.03-0.08% against 1.0-1.2%, 0.38-0.40 mm against 0.5-0.55 mm) than their single nitrogen-containing analogues regarding antioxidation and antiwear properties, as well as regarding detergent-dispersive and anticorrosion properties. For example: a succinimide fragment containing AKI-214"B" additive has better detergent properties than its analogue without boron (0 point)
The multifunctionality of boron-nitrogen-containing alkylphenolates makes it possible to create high-quality motor oils based on them. Creation of ether groups in modifications obtained on the basis of alkanolamine was determined by IR spectrum analysis of the products received from first condensation through to last neutralization.
Absence of a peak belonging to NH-group in the IR spectrum of aminomethylation product of alkhanolamines with methylen-bis-alkylphenol and presence of 1049.54 cm-1 zone characterizing -C-O- bond in alcohols, and diffuse line overlapping by OH groups of phenol at 3362,29 cm-1 confirms the structure of the aminomethylized product (Figure 1). In the IR spectrum of the intermediate product obtained after condensation of aminomethylized product with B(OH)3 we observe the -C-OH- group, the absence of 1049.54 cm4 zone, and instead of it formation of 1160.40 cm-1 zone, which can be said belongs to the -C-O- bond of ether groups (Figure 2)

Figure 2 IR spectrum of the product of aminomethylation of alkylphenol with diethanolamine and condensation with boric acid.
Appearance (for sliding) of simple ether groups at 1131.56 cm-1 or 1129.46 cm4 in the final product - phenolates proves the creation and maintenance of ether-covalent >B-0-C-group bonds until the end.
Coordination bonds can be easily created at low temperatures (80-85°C). Virtual absence of observed zone in phenolic OH after condensation of phenolate with boric acid in the IR spectrum of phenolates confirms that the phenolate was obtained (Figures 3 and 4).

Figure 3 IR spectrum of calcium salt from the product of aminomethylation of alkylphenol with diethanolamine and condensation with boric acid

Figure 4 IR spectrum of calcium salt from the condensation product of alkylphenol with monoethanolamine and boric acid
COMPARISON OF THERMOOXIDATION PROPERTIES OF BORON-NITROGEN-CONTAINING ALKYLPHENOLATE ADDITIVES WITH INDUSTRIAL AND SYNTHESIZED ANALOGUES
The thermostability of additives is determined by the ending temperature of the first stage and beginning temperature of the second stage of destruction, thermogravimetric (TQ) curves by weight loss (T10%, T20%, T50%, T70%), and the thermooxidation degrees of examples are defined by exothermic effects and this is shown in differential-thermal analysis curves (DTA).
The thermooxidation stability of AKi-114"B" is 3200C and difference between it and AKi-114 is just 100C. At the same time, as can be seen from the DTA of AKi-114"B", the exo-effect is considerably less than the same indicator for the additive without boron. In this part of the curve, borders have a spread shape. But in the condensation products of additives (CP) the beginning of the second stage of destruction for CP of AKi-114 and AKi-114"B" additives is 2850C and 3100C, respectively. The maximum level for thermodestruction without boron is 6100C, and with boron it is 7000C (Figure 5).

Figure 5 Thermogravimetric curves for the condensation products of AKI-114 ( 1) and AKI-114B (2) additives 1.AKI-115; 2. AKİ-115B, 3. AKI-114B
The first stage for thermodestruction of the condensation products of AKi-115 and AKÍ-115B additives is 3000C and 3200C respectively, but in the additives it is 3450C and 3600C, respectively. But half-life period T50% makes 3600C for the AKi-115 additive, and 4000C for AKi-115"B". The thermooxidation efficiency (exo-effect) of AKi-115B in comparison with analogues is observed at temperatures of more than 200C (Table 2, Figure 6).
As can be also seen from Table 2, the difference between thermostability indicators of AKi-114B and AKi-115B additives is 400C (320-3600C). It shows moving thermo-chemical transformations (thermooxidation stability) of solvent oil contained in the additives, which usually takes place at 280-2900C to high-temperature zones [10].
At the same time according to Table 2, the half-life period for such industrial samples as ACK and MACK, which are part of the carboxylate group containing additives, makes 340-3450C. The thermooxidation stability of nitrogen-containing carboxylate can be compared with boron-nitrogen-containing ones. Both of them are a little higher than sulfur-containing ones.
AKI-114 - Calcium salt of condensation product of alkylphenol with ammonia and formaldehyde [11];
AKI-115 - Calcium salt of condensation product of alkylphenol with monoethanolamine and formaldehyde (7)
VNIINP-714-Carbonated calcium salt of thio-bis-alkylphenol [12];
OLOA-218A-Carbonated calcium salt of thio-bis-alkylphenol [13]; ASK-Calcium alkylsalicylate;
MASK-High-alkalinity alkylsalicylate [14],
It is clear from the table that the modified alkylphenolate additive AKI-115"B" has the highest thermostability indicators (360°C)
It has been established thatthermostability of:
Boron-containing additives is higher (10-20°C) than analogues without boron;
Carbonated high alkalinity additives Is much higher (10-15°C) than average alkalinity ones;
Additives obtained on the basis of alkanolamines is higher (30-40°C) than additives containing primary amine.
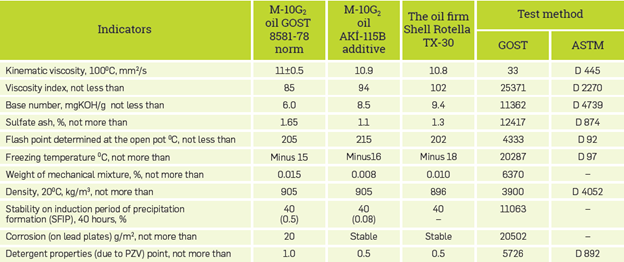
Through use of the above-mentioned heteroatom-containing additives, several motor oil compositions for gasoline and diesel engines have been developed. For example, M-10G2 oil is an industrial diesel, especially heavy-loaded auto-tractor diesel oil produced to GOST 8581. This M-10G2 oil has been prepared by use of VNIINP-360 additive with insignificant multifunctionalty tribological properties and average alkalinity.
Through use of the boron-nitrogen-containing AKI-115B, an additive composition meeting the requirements of the oil has been created.
On the basis of the oil and a small portion of AKI-210B and AKI-115B additive and other components, oil compositions have been developed and tested M-10G2k, M-10V2 [15], [16].
In Tables 3 and 4 there are ASTM numbers and GOST methods according to which physicochemical and functional properties of oils are determined. Both compositions of oils M-8B and M-10G2. meet modern requirements. [17]-[19]
So, it has been shown that the multi-functionality and high efficiency of boron nitrogen-containing alkylphenolate additives make possible their use as part of motor oil for gasoline and diesel engines that meet modern requirements.
CONCLUSIONS
For the first time, on the basis of the condensation of alkylphenoles with formaldehyde, amines, alkanol amines and boric acid step by step, boron-nitrogen-containing alkylphenolate additives have been synthesized, with several advantages having been determined for boron-containing additives (AKÍ-114B, AKÍ-115B, AKÍ-210B, AKi-212B, AKI-214B, AKI-219B) in comparison with their analogues without boron:
Detergent properties (0 points against 0.5-1.0 points);
antiwater stability: alkalinity reduction (13-15% against 18-20%);
prevailing thermooxidation properties: according to results of the thermogravimetric analysis, the beginning of the second phase of thermodestruction makes 320-3600C;
AKI-115"B"additive obtained from alkanolamine has higher thermal oxidation stability (3600C) than additives obtained from primary amines;
the antiwear properties of boron-containing additives is higher than the same properties for nitrogen-containing analogues (wear scar diameter 0.40 mm against 0,55 mm).
Taking into account everything mentioned above, it is possible to create modern motor oil compositions through the application of additives containing boron-nitrogen.