1. Introduction
This article is an extension of the paper originally presented in ExpoIngeniería (2022) [1]. Iron is considered an essential micronutrient for plants, but many soils lack sufficient iron in a form that is readily available to crops to ensure satisfactory growth, especially in tropical and sub-tropical regions.
Iron is one of the most important micronutrients for crop plants due to its use in important physiological processes such as photosynthesis, mitochondrial respiration, metal homeostasis, and chlorophyll synthesis [2]. Although iron is one of the most abundant elements in the earth's crust, its availability to plant roots is very low [3].
Iron deficiency affects major crops worldwide, including some of the major economic importance, such as maize and lettuce. On the one hand, maize is a cereal of great importance as it is a basic food staple for humans and animals and a source of a large number of industrial products [4]. In Colombia maize is one of the most important crops in the agri-food sector, its production increased by 76% between 1961 and 2016, while its demand grew at a faster rate [5]. Lettuce, on the other hand, is the most cultivated leafy salad vegetable in Colombia [6]. The importance of this horticultural species is determined by some of its biological characteristics and by its content of vitamins and nutritional substances [7].
Substituting nanofertilizers for conventional methods of fertilizer application is a method to deliver various macro and micro nutrients into the soil gradually and in a controlled way, thus preventing accumulation and pollution of various natural resources [8]. Particularly, iron oxide nanoparticles have emerged as an innovative and promising method of iron application in agricultural systems [9,10]. Iron oxide nanoparticles are usually stabilized in an aqueous suspension to be subsequently applied to crops. Therefore, the main aim of this study was to evaluate the effect of the application of different concentrations of suspensions of iron oxide magnetic nanoparticles of magnetite-maghemite on the germination and growth of maize and lettuce seeds.
2. Materials and methods
2.1 Iron oxide nanoparticles
The aqueous suspensions of magnetite-maghemite nanoparticles stabilized with chitosan used in this research were synthesized and characterized at the Instrumentation and Spectroscopy Laboratory of the Universidad EAFIT. The structural and magnetic characterization of nanoparticles obtained was carried out by Electron Transmission Microscopy (TEM), Infrared spectroscopy (FTIR), Transmission Mössbauer spectroscopy (TMS) and Vibrating sample magnetometry (VSM) at room temperature, indicating that the sample is composed by particles of magnetite (Fe3O4) and its highly oxidized state of maghemite (Y-Fe2O3), with a magnetic-polymer core-shell structure and a mean diameter of 11 nm estimated by TEM. The percentages of magnetite and maghemite in the samples, determined by Mössbauer spectroscopy, were 42(2) % and 58(2) % respectively. The iron content in the suspension of magnetic NPs stabilized with quaternized chitosan was obtained by visible spectrophotometry, obtaining a value of 454 ppm. The stock suspension of iron oxide NPs was diluted in deionized water to obtain the desired concentrations for the different tests.
2.2 Plant material
Seeds of maize (Zea mays L.) variety FNC31AC, used in this project, were supplied by “Federación Nacional de Cultivadores de Cereales, Leguminosas y Soya (FENALCE)”. Lettuce seeds (Lactuca sativa L.) were supplied by “Tierragro”. Both types of seeds were stored in sealed bags at 10 °C. Preliminary studies showed that the average germination rate of these seeds was 90 % for maize and 70 % for lettuce.
2.3 Seed germination trial (phytotoxicity evaluation)
The seed germination trial consisted of incubation of the seeds in a humid chamber, which consisted of placing 10 and 20 seeds for maize and lettuce, respectively, per Petri dish (100 mm x 15 mm). Each Petri dish contained a cellulose filter paper at the bottom, impregnated with 3 mL of deionized water (control) or with aqueous suspension of nanoparticles at different iron contents (25 ppm, 35 ppm and 45 ppm). Petri dishes were stored in a dark place at 23 °C for 8 days. Each treatment was carried out by triplicate. After this time, the fresh biomass and root length of germinated seeds were recorded.
2.4 In vitro assay
The Murashige and Skoog (MS) basal medium was used [11]. This medium was supplemented with 30 g·L−1 sucrose and a suspension of iron oxide NPs at different iron contents (25, 35 and 45 ppm). MS basal medium without addition of NPs suspensions was used as a control. The culture media were solidified with 0.4% (w/v) bacteriological agar (Oxoid) and the pH was set to 5.7. Next, the media were autoclaved at 121 °C for 20 minutes and stored in a room at 30 °C for one week to rule out any presence of microorganisms.
Before starting the in vitro culture, the seed disinfection protocol was performed. First, the seeds were immersed in water, soap with 7 % Povidone iodine for 30 minutes and then washed with abundant water. Next, the seeds were incubated in 2 % NaOCl for 5 minutes for Lettuce and 15 % NaOCl for 15 minutes for maize, under constant agitation. Seeds were triple rinsed with distilled water, dried on Kraft paper and subsequently inoculated on MS medium, with 4 seeds in each flask for maize, and 5 seeds for lettuce. For each treatment, 15 repetitions were used. Flasks were kept in the growth room at 16 °C with a photoperiod of 3 weeks for maize and 7 weeks for lettuce.
2.5 Observations of in vitro cultures and biometric evaluation
The biometric data of the developed seedlings, such as the length (cm) of the primary root, plant height (cm), and dry biomass (g), were performed after 3 weeks for maize and 7 weeks for lettuce.
2.6 Statistical analysis
The measurements obtained from the in vitro and phytotoxic assays were processed using the analysis of variance (ANOVA) with 95 % of confidence, previously verifying the assumptions, Levent test for homoscedasticity, Shapiro-Wilks test for normality and Durbin-Watson test for independence. Non-parametric analyses were used when the data analyzed did not meet any of the assumptions. The statistical analysis was made by using RStudio software (version 4.2.2).
3. Results and discussion
3.1 Phytotoxic evaluation of iron oxide magnetic nanoparticles in Maize seeds
In previous trials, the effect of iron oxide nanoparticle suspension on maize seeds was evaluated by defining a set of aqueous suspensions of nanoparticles with iron contents of 3.12, 6.25, 12.5, 25, 50, 75 and 100 ppm. These suspensions were applied to yellow maize seeds (FNC31AC) in a wet chamber. As a result, it was obtained that the biomass and root length of germinated seeds decreased from the iron content higher than 50 ppm. On the contrary, for iron contents lower than this value, root length and biomass variables increased [1]. It was concluded that iron contents lower than 50 ppm did not present phytotoxic effects on maize seeds in the morphological aspect, since it did not inhibit their germination process and there was no evidence of seed necrosis.
Based on these results, the range of iron contents supplied by magnetite-maghemite nanoparticles to be evaluated was further delimited, so three of them were chosen, 25, 35 and 45 ppm. With these iron contents, the wet chamber test was performed again with yellow maize seeds (FNC31AC), as detailed in the methodology section 2.2. As shown in Fig. 1, the maize seeds exposed to the different treatments presented a germination percentage higher than 80 %, suggesting that the iron content evaluated did not cause an inhibition of the seed germination process, suggesting that there was no phytotoxic effect at the morphological level. Furthermore, according to the Kruskal-Wallis analysis, the treatments evaluated did not show statistically significant differences between them.
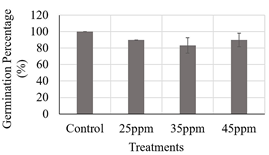
Source: own elaboration.
Figure 1 Evaluation of three iron contents supplied by suspension of nanoparticles (25, 35 and 45 ppm) on maize seeds. Kruskal-Wallis analysis with 95% confidence, germination percentage p-value: 0,0922.
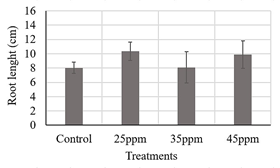
Source: own elaboration.
Figure 2 Evaluation of three iron contents supplied by suspensions of nanoparticles (25, 35 and 45 ppm) on maize seeds. ANOVA analysis with 95 % confidence, root length p-value: 0.291.
In the root length results, presented in Fig.2, none of the treatments showed statistically significant differences from the control treatment and between them, again suggesting that these iron contents did not have a phytotoxic effect in maize seeds.
3.2 In vitro trial with Maize and Lettuce seeds
Previous results suggested that the iron contents evaluated did not have a negative effect on maize seeds. To determine if this result is consistent with another type of crop, aqueous suspensions of nanoparticles with iron contents of 25, 35 and 45 ppm were evaluated on Lettuce seeds. In addition, to demonstrate not only the effect of this suspension on seed germination, but also on the growth and development of its seedlings, an in vitro assay was performed, as explained in the methodology, section 2.4.
Lettuce seeds were impregnated with the different nanoparticle suspensions in a wet chamber test to evaluate the effect of these nanocomposites on the seeds. As a result, the germination percentage of the seeds was higher than 67 % (75 % for the control treatment, 72 % for the 25 ppm and 45 ppm treatments, 67 % for the 35 ppm treatment, p-value ANOVA test 0,802). As for the root length variable, no statistically significant differences were observed from the control treatment and between the treatments, according to the ANOVA test (Fig. 3), suggesting that there is no phytotoxic effect of the suspension of magnetic nanoparticles on these seeds.
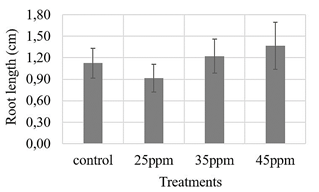
Source: own elaboration.
Figure 3 Evaluation of three iron contents supplied by nanoparticles (25, 35 and 45 ppm) on Lettuce seeds. ANOVA analysis with 95 % confidence, root length p-value: 0.227.
On the other hand, and having verified that the suspension does not present phytotoxic effects on maize and lettuce seeds, the in vitro test was carried out, where the seeds were sown in MS culture medium mixed with the determined iron content of the nanoparticle suspension.
For maize seeds used in this test, the germination percentage was higher than 60%, with the 45 ppm treatment having the highest value (84 %) and the 25 ppm iron concentration treatment having the lowest value (60%). However, according to the non-parametric Kruskal-Wallis analysis, there is no statistically significant difference between the control treatment (media MS) and the treatments with aqueous suspensions of nanoparticles (p-value: 0.1618).
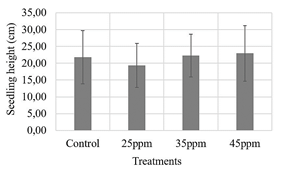
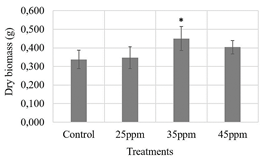
Regarding root length and seedling height measurements, 35 ppm treatment was the one that presented a higher value than the other treatments. However, neither showed statistically significant differences (Figs. 4, 5). The opposite was the case for the dry biomass variable, in which the control treatment had the lowest value and showed a statistically significant difference with the 35 ppm treatment (Fig. 6), which could support the above results, suggesting that the nanoparticle suspensions do not have a phytotoxic effect on maize seeds.
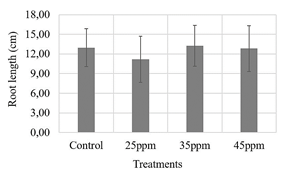
Source: own elaboration.
Figure 4 Evaluation of three iron contents supplied by nanoparticles (25, 35 and 45 ppm) on maize seeds. ANOVA test with 95% confidence, Root length p-value: 0.748
Source: own elaboration.
Figure 5 Evaluation of three iron contents supplied by nanoparticles (25, 35 and 45 ppm) on maize seeds. ANOVA test with 95% confidence, Seedling length p-value: 0.877
Source: own elaboration.
Figure 6 Evaluation of three iron contents supplied by nanoparticles (25, 35 and 45 ppm) on maize seeds. ANOVA test with 95 % confidence, Dry biomass p-value: 0.0365. *statistically significant difference respect to control treatment (Dunnet test, p-value: 0,0309).
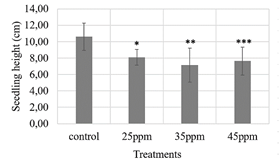
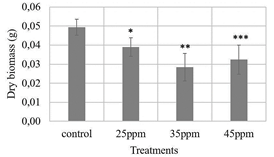
The lettuce seeds had a germination percentage above 80 %, with the control treatment having the lowest value (83 %) and the 25 ppm iron content treatment having the highest value (96 %). However, according to the ANOVA analysis with 95 % confidence, there were no statistically significant differences between the control and the other treatments (p-value: 0.118). This is not the same case for the seedling length variable (Fig. 7) and the dry biomass variable (Fig. 8), which showed significant differences between the control and the treatments evaluated.
Source: own elaboration.
Figure 7 Evaluation of three iron contents supplied by nanoparticles (25, 35 and 45 ppm) on lettuce seeds. Kruskal-Wallis test with 95% confidence, p-value: 0.0025. The asterisk represents a statistically significant difference from the control treatment, Pairwise-Wilcox test. *25ppm treatment-control treatment (p-value: 0,007), **35ppm treatment-control treatment (p-value: 0,007), ***45ppm treatment-control treatment (p-value: 0,011).
Source: own elaboration.
Figure 8 Evaluation of three iron contents supplied by nanoparticles (25, 35 and 45 ppm) on lettuce seeds. Kruskal-Wallis test with 95% confidence, p-value: 0.0001. The asterisk represents a statistically significant difference from the control treatment, Pairwise-Wilcox test. *25ppm treatment-control treatment (p-value: 0,0210), **35ppm treatment-control treatment (p-value: 0,0035), ***45ppm treatment-control treatment (p-value: 0,0035).
According to Figs. 7 and 8, it could be thought that the application of suspensions of magnetite-maghemite nanoparticles in the lettuce seeds might be slowing down the growth of the seedlings compared to the control treatment. However, with adequate seed germination and seedling growth, the results would suggest that no phytotoxic effect is occurring in the plants. It is advisable to carry out a greenhouse trial where the plants complete their full growth cycle and are under more similar conditions of temperature and humidity to those in the field to verify these initial results.
4. Conclusion
The results obtained from the tests carried out in this project to evaluate the effect of applying aqueous suspensions of magnetite-maghemite nanoparticles stabilized with chitosan, synthesized at EAFIT University, on maize and lettuce seeds, suggest that these suspensions did not have a phytotoxic effect on the seeds, as their germination and seedling growth took place normally. However, to verify more precisely and to evaluate whether the suspension could have a positive effect on the growth and development of the plants, it is recommended to carry out a greenhouse test where the plants are exposed to the suspension for a longer period of time.















