Introduction
Water is used in every action and reaction within the cell, such as transportation, cell integrity, and the regulation of body temperature. Low quality water has been shown to reduce animal performance (Marai et al., 2005).
Underground water is used as drinking water in areas with limited water supply. In these areas, water quality is poor, presenting a major challenge to poultry farming (Attia et al., 2013; 2015). Total dissolved salts, salinity, and nitrates are among the major factors determining the suitability of a particular water source for poultry (Morsy et al., 2012). In addition, the presence of excessive concentrations of inorganic ions such as Ca++, Mg++, Na+, Cl-, SO4 -, and HCO3 in water could induce poor performance, illness, or even death (Kellems and Church, 2002). The magnetic field of the earth naturally charges the water in lakes, wells, and running streams. However, water loses its magnetic charge as it passed through treatment plants and pipes. The exposure of water to magnetic fields restores and balances its natural energy (Ovchinnikova and Pollack, 2009).
The influence of magnetic treatment for improving agriculture has been studied with major emphasis on plants, while research on animal productivity has received less attention (Hozayn and Abdul Qados, 2010; Attia et al., 2015). Magnetized water improves health by inhibiting bacterial growth, while reducing pain, swelling, and weakness (Verma, 2011). Bergsrud and Linn (1990) reported an increase in growth of calves and sheep and a decrease in carcass fat of sheep that received magnetized water. In addition, Lin and Yotvat (1989) showed that milk yield increased when dairy cattle drank magnetized water. El-Hanoun et al. (2013) observed an improvement in productive and reproductive performance of doe rabbits, and Attia et al. (2015) observed an improvement in buck rabbit performance after receiving magnetized drinking water. Nonetheless, Sargolzehi et al. (2009) found that exposing water to a magnetic field produced no significant effect in performance, carcass composition, blood constituents or milk nutrient profile of lambs and goats.
Hence, this study sought to evaluate the effects of water quality and magnetized water on productivity and blood parameters of Egyptian geese.
Materials and methods
Ethical considerations
The scientific and ethics committee of the Animal Production Research Institute approved this experiment (protocol number 1-1-3-429; 29-5-2015).
Animals
A total of 108 one-year-old Egyptian female geese and 12 male geese were used in the experiment. The geese were randomly divided into four treatment groups comprised of 27 geese each, with three replicates of nine female geese plus 1 gander per replicate. Each geese group was subjected to one of the following treatments: tap water (TW), well water (WW), magnetized tap water (MTW), or magnetized well water (MWW). The WW was obtained from a 120 m depth desert well at Borg El-Arab city, and TP was obtained from the same city. The MWW consisted on exposing well water to a magnetic field of 4,000 gauss, while MTW was tap water exposed to the same magnetic field. The experiment lasted 28 weeks, from November 2012 to May 2013.
The geese were housed in twelve windowless pen houses (2 x 3 m2) furnished with wood shavings as litter. Each pen was equipped with six nests. Geese were exposed to natural light during daytime. Natural mating was allowed during the breeding season. Geese were housed in an intensive system with confinement in a house during the experiment and fed a commercial mash feed offered ad libitum, containing 16% CP, 2750 Kcal ME/Kg, 3% Ca, 0.35% non-phytate phosphorus, 0.60% total sulphur amino acids, and 0.70% lysine (AOAC, 2007). At the start of the experiment, all birds were vaccinated against avian influenza.
The physical characteristics of both WW and TW were determined with a Jenway 3505 pH meter (pH) and a Jenway 4150 conductivity meter (conductivity; Jenway, Stone, Staffordshire, UK). Elemental analysis were carried out using a DR3900 spectrophotometer (Hach, Loveland, CO, USA), a FIAstar 5000 analyser, model FO 55 (Foss A/S, Hillerod, DK, Denmark), and a flame photometer (Spectronic 20d, Thane, Maharashtra, India). Results are shown in Table 1.
Water was exposed to a magnetic field using a compact magnet (Aqua Correct, Blue Goose Sales, Post Falls, ID, USA; Figure 1). This is a coaxial permanent system that produces a magnetic field strength up to 4,000 gauss. The unit works as a physical magnet changing the crystal structure of lime. The strength of the magnet was measured using a Teslameter (Magnetic Sciences Inc., Acton, MA, USA) at the Applicate Laboratory in the City for Scientific Research and Biotechnology of the Egypt- Japan University of Science and Technology, Egypt.
Table 1 Physicochemical characteristics of tap and well water with (+) and without (-) magnetization.

Final body weight, egg number, egg weight, egg mass, feed intake, feed conversion ratio, water consumption, and mortality rates were recorded. Eggs were collected daily and stored in a storage room for seven days at 24 °C and 60% relative humidity. The geese were allowed to incubate and hatch their eggs naturally to determine fertility and hatchability for each treatment. A total of 12 hatches per group were used. Fertility was defined macroscopically on the 7th day of incubation. Hatchability of fertile eggs (HFE) was defined as the number of hatched goslings divided by the number of fertilized eggs per goose x 100. The number of hatched goslings per goose was calculated as = HFE x EN per bird/year, where EN is the total egg number.
During December, February, and April, 10 eggs per replicate (for a total of 30 eggs per water type) were collected to measure egg quality traits according to Attia et al. (1994). Eggs were weighed and broken open, and the eggshell, yolk, and albumen were weighed individually and expressed as a percentage of total egg weight. In addition, shell thickness (μm) was measured using a micrometre (B.C. Ames, Framingham, MA, USA) and egg shape index [(egg width/egg length) x 100] was estimated.
At 15 months of age, a blood sample (6 mL) was collected in the morning, before access to feed, from four hens chosen randomly per replicate for a total of 12 samples per treatment. An aliquot of blood from each bird was placed in a heparinised tube, and another in a non-heparinised tube. Plasma and serum were obtained by sample centrifugation at 1,500 x g for 20 min, and plasma and sera were stored at -20 °C until analyses. Serum urea, creatinine, alanine aminotransferase (ALT), and aspartate aminotransferase (AST) were determined using a commercial kit (Diamond Diagnostics, Egypt), as reported by Attia et al. (2009). Total antioxidant capacity (TAC) was determined according to Koracevic et al. (2001), while thiobarbituric acid-reactive substances (TBARS) were measured according to Tappel and Zalkin (1959).
Plasma 17β-estradiol (E2) and progesterone (P4) were assayed by radioimmunoassay using the commercial kits DSL-43100 and DSL-3900, respectively (Diagnostic systems Laboratories Inc., Webster, TX, USA), according to Abraham (1977). The progesterone/estrogen ratios were also calculated.
All hatched goslings from each water treatment group (with average ranging from 600 to 1,200 goslings/ treatment) were raised in four floor pens furnished with wood shavings using common management practices until 20 weeks of age. Growth performance, such as BW, body weight gain, feed intake, and feed conversion was recorded from four to 20 weeks of age. At 20 weeks of age, five goslings were randomly chosen from each treatment and slaughtered for evaluation of carcass and organ traits. Meat produced per goose per year (MPG) was calculated using the following equation: MPG (Kg) = number of goslings produced per goose per year x body weight (Kg) of the goslings at 20 weeks of age.
Statistical analysis
Data were subjected to analysis of variance using the factorial experimental design (2 types of water × 2 magnetic treatments) of the general linear model procedure of the SAS software program, version
9.2 (SAS Institute Inc., Cary, NC, USA). Difference among means was determined using Duncan’s new multiple range test (SAS, 2004) at p<0.05.
Results
Exposure to the magnetic field increased mineral levels, salinity and conductivity of both types of water (Table 1). The water treatment had no effect (p>0.05) on initial body weight nor on feed intake. However, consumption of MTW improved (p<0.05) final body weight (+19.6%), number of eggs (+18.7%), egg weight (+4.27%), egg mass (+24.7%), feed conversion rate (-18.2%), and water consumption (+33%) compared to the non-treated tap water and well water groups, whether treated or not (Table 2). Fertility rate (+9%), hatchability of fertile eggs (+11.6%) and number of hatched goslings (+45.8%) were improved by consumption of MTW compared to the other types of water (Table 3).
There was no effect (p>0.05) of water treatment on the egg shape index. However, geese that received MTW exhibited greater (p<0.05) eggshell percentages (+13.5%) and thickness (+10.5%) -indicating better eggshell quality- plus greater yolk (+8.3%) and lower albumen (-6.6%; Table 4).
The interaction of water type × magnetic exposure affected all of blood parameters (p<0.05), with MTW geese presenting higher E2 and P4 and total antioxidant capacity values, and decreased blood urea, creatinine, AST, ALT, and TBARS compared to the other treatments (Table 5).
Goslings from geese that drank MTP had the best productive performance compared to the other treatments (Table 6). There was no effect (p>0.05) of treatment on liver, heart, or gizzard percentage. Dressing percentage was higher and abdominal fat and skin percentages were lower in goslings produced from geese that drank MTP (Table 7).
Discussion
As expected, well water exhibited lower quality than tap water. Well water resulted in poor performance, impaired renal and liver functions, and TBARS compared to TW. These impairments in different indices of geese performance and physiological functions can be explained by the 10% decrease in water consumption by geese receiving WW. The negative effect on MPG matched the lower number of hatched goslings due to the low fertility and hatchability of fertile eggs, and as previously mentioned, could be attributed to decreased water consumption. Poor water quality has been shown to decrease animal performance and induce health risks (Wahaab and Badawy, 2004; Attia et al., 2013; 2015). Moreover, De Blas (2013) reported that compositional alterations in the gut are a possible cause of gut disorders, retarded growth, and health problems in animals.
Improved water quality occurred with exposure to a magnetic field. Exposed water showed considerable changes in its physicochemical characteristics, such as pH (8.69 vs 7.37 %), conductivity (9.83 vs 5.24 %), salinity (2.47 vs 9.69 %), Na (6.78 vs 15.78 %), Ca (15.02 vs 20.58 %), Mg (15.81 vs 20.08 %), Cl (27.05 vs 18.60 %), and dissolved oxygen (44.09 vs 37.07 %) of WW and TW, respectively, as published by Attia et al. (2015). According to the literature, exposing water to magnetic fields changes its properties, with a slightly alkaline pH (>7), higher mineral (higher salinity) and oxygen contents, and lowers germs, which fulfils animal requirements for water quality (Mg-Therapy, 2000). Magnetized water reduces the hydrogen-oxygen bond angle within the water molecule from 104 to 103 degrees, leading to better water absorption across cell membranes (Verma, 2011). The increasing salinity of well water, as shown in Table 1, may be due to an increase in soluble salts associated with conductivity. Water passing through a magnetic field acquires a finer and more homogeneous structure (Tkachenko and Semyonova, 1995), which increases its fluidity and ability to dissolve various constituents such as minerals and vitamins (Kronenberg, 1985). Consequently, it improves the biological activity of solutions, positively affecting performance of animals and plants (Al-Mufarrej et al., 2005). Another aspect of using magnetic fields to improve water quality is the decrease in environmental pollution (Attia et al., 2013; 2015).
Table 2 Effect of tap (TW) or well water (WW) with (+) or without (-) magnetization on initial (IBW, g) and final body weight (FBW, g), egg number (EN, egg/hen/year), egg weight (EW, g), egg mass (EM, g), feed intake (FI, g/bird/day), feed conversion ratio (FCR, g/g), and water consumption (WC, cm3/hen/day) of Egyptian geese (mean ± SE).

a, b, c, d Means within a row of each criteria of the interaction effect not sharing common superscripts are significantly different (p<0.05). A, B Means within a row of each criteria of the water type effect not sharing common superscripts are significantly different (p<0.05). x, y Means within a row of each criteria of the magnetic treatment effect not sharing common superscripts are significantly different (p<0.05).
Table 3 Effect of tap (TW) and well water (WW) with (+) or without (-) magnetization on fertility percentage (FP, %), fertile eggs` hatchability (FEH, %), and number of hatched goslings (NHG) per geese (mean ± SE).
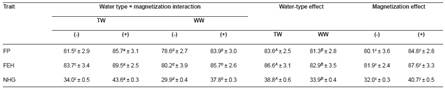
a, b, c, d Means within a row of each criteria of the interaction effect not sharing common superscripts are significantly different (p<0.05). A, B Means within a row of each criteria of the water type effect not sharing common superscripts are significantly different (p<0.05). x, y Means within a row of each criteria of the magnetic treatment effect not sharing common superscripts are significantly different (p<0.05)
Table 4 Effect of tap (TW) and well water (WW) with (+) or without (-) magnetization on eggshell percentage (ESP, %), shell thickness (ST, μm), egg shape index (ESI), yolk percentage (YP, %), and albumen percentage (AP, %) of Egyptian geese (mean ± SE).
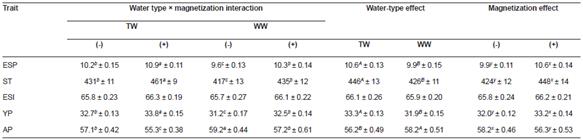
a, b, c, d Means within a row of each criteria of the interaction effect not sharing common superscripts are significantly different (p<0.05). A, B Means within a row of each criteria of the water type effect not sharing common superscripts are significantly different (p<0.05). x, y Means within a row of each criteria of the magnetic treatment effect not sharing common superscripts are significantly different (p<0.05)
Table 5 Effect of tap (TW) and well water (WW) with (+) or without (-) magnetization on plasma estrogen (E2, pg/mL), progesterone (P4, ng/mL), urea (U, mg/dL), creatinine (C, mg/dL), aspartate aminotransferase (AST, IU), alanine aminotransferase (ALT, IU), total antioxidant capacity (TAC, mmol/L), and thiobarbituric acid reactive substances (TBARS, nmol/mL) of Egyptian geese (mean ± SE).
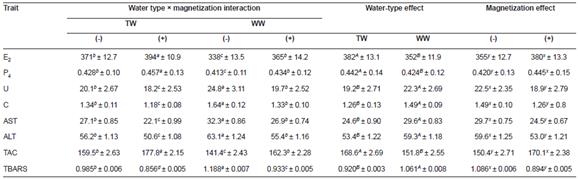
a, b, c, d Means within a row of each criteria of the interaction effect not sharing common superscripts are significantly different (p<0.05). A, B Means within a row of each criteria of the water type effect not sharing common superscripts are significantly different (p<0.05). x, y Means within a row of each criteria of the magnetic treatment effect not sharing common superscripts are significantly different (p<0.05)
Table 6 Effect of tap (TW) and well water (WW) with (+) or without (-) magnetization on initial (IBW, g) and final body weight (FBW), body weight gain (BWG, g), feed intake (FI, g/bird/day), feed conversion ratio (FCR, g/g), meat produced/geese/year (MPG, Kg), water consumption (WC, cm3/geese/day), and mortality rate (MR,%) of Egyptian gosling during 4-20 weeks of age (mean ± SE).

a, b, c, d Means within a row of each criteria of the interaction effect not sharing common superscripts are significantly different (p<0.05). A, B Means within a row of each criteria of the water type effect not sharing common superscripts are significantly different (p<0.05). x, y Means within a row of each criteria of the magnetic treatment effect not sharing common superscripts are significantly different (p<0.05)
Table 7 Effect of water (TW) and well water (WW) with (+) or without (-) magnetization on dressing percentage (DP), and percentage of skin (PS), abdominal fat (PAF), liver (PL), heart (PH), and gizzard (PG) of 20 week-old Egyptian gosling (mean ± SE).
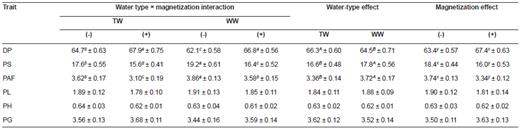
a, b, c, d Means within a row of each criteria of the interaction effect not sharing common superscripts are significantly different (p<0.05). A, B Means within a row of each criteria of the water type effect not sharing common superscripts are significantly different (p<0.05). x, y Means within a row of each criteria of the magnetic treatment effect not sharing common superscripts are significantly different (p<0.05).
Magnetic treatment of both types of water improved productive performance, such as number, weight and mass of eggs, feed conversion ratio, egg fertility, and egg hatchability, as well as performance of growing goslings. The amount of meat produced by geese receiving MTW increased by 63.7% compared to the geese receiving WW (154.4 vs 94.3 Kg, respectively). The enhanced geese and gosling performance was in agreement with increases in water consumption of geese and goslings receiving MTW when compared to those receiving the other water types. Increased water consumption improved biological performance and health status of geese, as indicated by lower blood levels of urea and creatinine, associated with an improvement in renal function, and lower activities of AST and ALT enzymes, which may be a result of better liver function. Additionally, there was an increase in reproductive hormone levels and total antioxidant capacity, and decrease of TBARS levels in geese ingesting MTW. All of these effects could be due to increased mineral solubility, facilitating nutrient transfer across cell membranes and, thus, uptake and utilization (El-Kholy et al., 2008; Attia et al., 2013; 2015). Moreover, magnetic treatment may improve health status of animals by improving water quality, reducing lime deposition in pipes and bacterial load (Sargolzehi et al., 2009).
Improved performance of goslings hatched from geese consuming magnetized water during growth showed a long-term carryover effect of water type and magnetization on progeny performance. This could be explained by improvements in health and immunity of geese and, thus, passive immunity of goslings, which is similar to the results reported by Attia et al. (2013; 2015). These improvements in progeny performance are in agreement with those reported by Lin (1995), who found that MTW improved performance of farm animals.
Interestingly, magnetized water showed a quite large long-term carryover effect in further experiments, as shown by the increased dressed carcass weights of goslings hatched from geese that drank MTW and MWW by 4.9 and 7.8%, respectively. A promising feature was a decrease in skin percentage (11.4 and 14.6%) and percentage of abdominal fat (14.4 and 7.3%) of goslings produced by geese that consumed MTW and MWW, respectively. These results indicate that magnetic exposure induced a greater effect on percentage of dressed carcass and skin of geese that drank WW, but the opposite was shown for their abdominal fat. The ability of magnetic treatment to affect lipid metabolism observed in this study is similar to that reported by Bergsrud and Linn (1990), who found an increase in growth of calves and sheep and a decrease in carcass fat of sheep receiving magnetized water.
In conclusion, the ingestion of magnetized tap water and, to a lesser extent, of magnetized well water, improved body weight and feed conversion rate of the birds, besides renal and hepatic functions; it also increased production, quality and hatchability of eggs, levels of reproductive hormones (progesterone and estrogen), and the blood antioxidant status.















