Introduction
Atrial fibrillation (AF) is a common disorder with a growing prevalence and incidence 1,2, and its occurrence has been associated with prior cardiovascular diseases 3. In Colombia, this disease occurs more often in patients over the age of 60 and increases its incidence and prevalence as the patients' age increases. Within its categories, nonvalvular atrial fibrillation (NVAF) has been found to be more frequent than valvular atrial fibrillation (VAF) 4.
In AF, the patients' heart rhythm and rate are altered, facilitating thrombus formation. In addition, patients with AF may have associated comorbidities such as arterial hypertension (HTN), heart failure (CHF) or diabetes mellitus (DM), among others 4, producing a greater risk of complications like cerebrovascular accidents (CVAs) or transient ischemic attacks (TIAs). Therefore, oral anticoagulants (OACs) are the recommended cornerstone of AF treatment, to prevent these types of complications 1,5. This treatment may also cause hemorrhagic complications; thus, scales like CHADS2 and CHADS2-VASc have been developed to determine the type of patients who need this treatment, and scales like HAS-BLED are used to evaluate the patients' risk of bleeding 1.
The OACs used to treat AF include vitamin K antagonists (VKAs) like warfarin, and non-vitamin K antagonists (NOACs), including the direct thrombin inhibitor dabigatran as well as factor Xa inhibitors like apixaban, edoxaban and rivaroxaban 3,5. It is important to note that both VKAs as well as NOACs have proven to be effective in preventing CVAs in patients with AF. However, the European (ECS) and American (AHA/ACC/HRS) guidelines recommend NOACs as first-line treatment, and do not recommend using antiplatelet (AP) medications. The latter are not recommended due to a lack of scientific evidence in AF treatment and the comparable risk of patients developing hemorrhages while receiving this treatment 3,6,7. Nevertheless, in some countries, NOACs can only be prescribed if the international normalized ratio (INR) on VKAs is suboptimal; that is, if VKA treatment has failed 6. In addition, and despite international recommendations, the most prescribed treatment for AF at diagnosis in Latin America is VKAs ± APs, followed by NOACs ± APs and APs alone 8.
Since there is little available literature on the characteristics and treatment of people with AF in Latin America, and given the prevalence of this disease in Colombia, this study seeks to analyze the epidemiological and clinical characteristics of a population with NVAF on OAC treatment at Instituto Cardiovascular del Tolima (Ibagué, Colombia). This will be achieved through the following specific objectives: 1) To describe the sociodemographic and clinical characteristics of the patients participating in the study; 2) To identify the type of anticoagulation used by the patients participating in the study; 3) To identify the risk (CHADS2 and CHADS2-VASc) and frequency of CVAs/TIAs in the study participants; 4) To identify the risk (HAS-BLED) and frequency of hemorrhages in the study participants; and 5) To analyze the relationship between the clinical characteristics and the presence of atrial fibrillation.
Materials and methods
This was a cross-sectional observational study of the use of anticoagulants in treating NVAF. It included patients over the age of 18 with NVAF who were seen by cardiology at a tertiary care center and who signed informed consent in which they agreed to participate in the study. Patients with VAF and those with any other indication for anticoagulation were excluded.
The participants for this study were selected through non-probability, convenience sampling, obtaining a sample of 146 participants. The patients were invited to participate at their cardiology appointments, during which their medications were reviewed, since some of the participants were found to be using non-recommended doses. Subsequently, the study data was taken from the participants' medical charts between 2018 and 2019, with a one-year follow up of each chart at Instituto Cardiovascular del Tolima (Ibagué, Colombia).
Data collection was intended to gather sociodemographic data, including age, sex, weight, height and BMI, as well as clinical data including glomerular filtration rate (GFR), comorbidities (coronary artery disease [CAD], congestive heart failure [CHF], HTN, DM, cerebrovascular disease [CVD], peripheral vascular disease [PVD]), OAC and/or AP treatment with its dosing, and complications like CVAs/TIAs, systemic embolisms, major or minor hemorrhages and death.
The patients were classified using the CHADS2 scale (a validated scale used in phase III randomized clinical trials of NOACs), the CHADS2-VASc scale (a scale subsequently validated for risk stratification for CVAs or systemic embolisms in patients with AF) and the HAS-BLED scale (a risk stratification scale for bleeding, which can occur in patients as a side effect of this type of medications). Each scale evaluated the following: CHADS2, age ≥ 75 years, a history of CHF, HTN, DM, and prior CVA/TIA symptoms; CHA2DS2-VASc, C: congestive heart failure; H: hypertension; A: age ≥ 75; D: DM; S: CVA/TIA, prior embolism; V: vascular disease; A: age 65 to 74 years; and Sc: sex category; and HAS-BLED H: uncontrolled HTN; A: altered kidney or liver function; S: prior CVA; B: a history of or predisposition to hemorrhaging; L: INRb, E: age >65 years; and D: medications (APs, NSAIDs) and/or excessive alcohol consumption. Based on each scale's score, patients were classified as having a low, moderate or high risk for developing CVAs/ TIAs or hemorrhages, as applicable.
We consider this study's main bias to be information bias, since the data recorded in the medical chart depends on the evaluators' criteria and therefore the data collected may be affected by different factors such as the type of interview, patients' recall, and attendance at medical appointments.
A descriptive univariate analysis of continuous and categorical variables was conducted using R version 4.0, (copyright (C) 2020 The R Foundation for Statistical Computing). Descriptive statistics, measures of central tendency, tests of data dispersion, normality analyses and graphs were developed for continuous variables. Frequencies, proportions, lower and upper limits and illustrative graphs were used for each of the categorical variables. In addition, a bivariate analysis was run using a Pearson correlation matrix between continuous variables, and contingency tables between categorical variables, in which association coefficients were calculated using the Chi2 test.
Results
Of the 146 participants with a diagnosis of NVAF at Instituto Cardiovascular del Tolima (Ibagué, Colombia), 53% were women and 47% were men, with a median age of 77 years (SD: 8.9 years). The most commonly diagnosed type of atrial fibrillation was the permanent type, in 56.85% (n=83), paroxysmal (n=59) in 40.41% and persistent (n=4) in 2.7% of all the analyzed patients. A total of 16.43% (24/146) were found to be taking concurrent acetylsalicylic acid (ASA) and direct OACs; only 2% (n=3) of the patients in the database were receiving dual AP therapy with ASA and clopidogrel (Table 1).
Table 1 Baseline characteristics of the population.
| Characteristic | Category | Frequency by category (%) | Lower limit of the frequencies (95%) | Upper limit of the frequencies (95%) |
|---|---|---|---|---|
| Sex | Female | 77 (53) | 45 | 61 |
| Male | 69 (47) | 39 | 55 | |
| Age range | 46 - 50 | 2 (1) | 0 | 3 |
| 51 - 55 | 3 (2) | 0 | 4 | |
| 56 - 60 | 3 (2) | 0 | 4 | |
| 61 - 65 | 4 (3) | 0 | 5 | |
| 66 - 70 | 25 (17) | 11 | 23 | |
| 71 - 75 | 22 (15) | 9 | 21 | |
| 76 - 80 | 37 (25) | 18 | 32 | |
| 81 - 85 | 32 (22) | 15 | 29 | |
| 86 - 90 | 12 (8) | 4 | 13 | |
| 91 - 95 | 6 (4) | 1 | 7 | |
| Type of NVAF | Paroxysmal | 59 (40) | 32 | 48 |
| Permanent | 83 (57) | 49 | 65 | |
| Persistent | 4 (3) | 0 | 5 | |
| Treatment with ASA | No | 123 (84) | 78 | 90 |
| Yes | 23 (16) | 10 | 22 | |
| Treatment with clopidogrel | Yes | 3 (2) | 0 | 4 |
| No | 143 (98) | 96 | 100 | |
| Treatment with amiodarone | No | 117 (80) | 74 | 87 |
| Yes | 29 (20) | 13 | 26 | |
| History of CAD | No | 103 (71) | 63 | 78 |
| Yes | 43 (29) | 22 | 37 | |
| History of HTN | No | 26 (18) | 12 | 24 |
| Yes | 120 (82) | 76 | 88 | |
| History of CHF | No | 131 (90) | 85 | 95 |
| Yes | 15 (10) | 5 | 15 | |
| History of DM2 | No | 109 (75) | 68 | 82 |
| Yes | 37 (25) | 18 | 32 | |
| History of CVA | No | 139 (95) | 92 | 99 |
| Yes | 7 (5) | 1 | 8 | |
| History of CKD | No | 139 (95) | 92 | 99 |
| Yes | 7 (5) | 1 | 8 | |
| History of PVD | No | 133 (91) | 86 | 96 |
| Yes | 13 (9) | 4 | 14 | |
| LVEF | 40-49 | 13 (9) | 4 | 14 |
| < 40 | 18 (12) | 7 | 18 | |
| > 50 | 114 (79) | 72 | 85 | |
| GFR by Cockroft Gault | Mild impairment 60-89 | 50 (34) | 27 | 42 |
| Mild-moderate impairment 45-59 | 52 (36) | 28 | 44 | |
| Moderate-high impairment 30-44 | 24 (17) | 11 | 23 | |
| Severe impairment 15-29 | 7 (5) | 1 | 8 | |
| Normal or high, greater than or equal to 90 | 12 (8) | 4 | 13 | |
| NOAC dose | Apixaban 2.5 mg x 2 | 11 (8) | 3 | 12 |
| Apixaban 5 mg x 2 | 19 (13) | 8 | 18 | |
| Dabigatran 110 mg x 2 | 1 (1) | 0 | 2 | |
| Dabigatran 150 mg x 2 | 3 (2) | 0 | 4 | |
| Rivaroxaban 15 mg | 35 (24) | 17 | 31 | |
| Rivaroxaban 20mg | 76 (52) | 44 | 60 | |
| VKA dose | Warfarin 5 mg | 1 (1) | 0 | 2 |
| EPS: Health instance company; NVAF: Nonvalvular atrial fibrillation; ASA: Acetylsalcylic acid; CAD: Coronary artery disease; HTN: Arteral hypertension; CHF: Congestive heart failure; DM2: Type 2 diabetes mellitus; CVA: Cerebrovascular accident; CKD: Chronic kidney disease; PVD: Peripheral vascular disease; LVEF: Left ventricular election fraction; GFR: Glomerular filtration rate; NOAC: Non-vitamin K antagonist oral anticoagulants; VKA: Vitamin K antagonists. | ||||
Description of sociodemographic and clinical characteristics
Regarding the participants' clinical characteristics, the median body mass index was 25.9 kg/m2 (SD: 4.76). The median creatinine was 0.99 mg/dL and the median glomerular filtration rate was 55.8 mg/dL.
Data was collected on the medical history of the 146 study patients. Seventy-one percent of the patients did not have coronary artery disease and 82% had arterial hypertension, in line with what is reported in the literature, where this is a frequent association. Ten percent of the patients had heart failure (79% LVEF > 50%), 25% had DM, 5% had had a CVA, 5% had chronic kidney disease (87% mild, mild-moderate or moderate-high reduction) and 9% had PVD. Table 1 differentiates the patients' baseline characteristics.
Regarding treatment, 84% of the patients (123 of them) were not receiving ASA and 98% were not being treated with clopidogrel, in line with the clinical practice guidelines which establish that APs are not indicated in this type of patients. Twenty percent of the patients (29 of them) were being treated with amiodarone. For NOACs, 76% of the patients were treated with rivaroxaban at a dose of 15 mg/day (24%) and 20 mg/day (52%); 7% were treated with apixaban at a dose of 2.5 mg twice a day and 13% with apixaban at 5 mg twice a day; 2% were treated with dabigatran at 150 mg twice a day, and 1% were treated with dabigatran at 110 mg twice a day. It is important to note that only 1% of the 146 patients were being treated with warfarin.
According to the classification (Table 2), 24% of the patients included in the study were classified as CHADS2 1, 47% as CHADS2 2, and 18% as CHADS2 3, adding up to 89% of all the study data. These data are related to 90% of the patients classified as CHADS2-VASc 2-5. Six percent of the patients with classifications of 0 and 1 received anticoagula tion therapy despite this not being the current clinical practice guideline recommendation. Of the 146 patients analyzed, 12% had a HAS-BLED 1 classification, 45% had a HAS-BLED 2 classification, and 30% had a HAS-BLED 3 classification.
Table 2 CHADS2, CHA2DS2-VASc, and HAS-BLED classification scales.
| Characteristic | Category | Frequency by category (%) | Lower limit of the frequencies (95%) | Upper limit of the frequencies (95%) |
|---|---|---|---|---|
| CHADS2 scale | 0 | 9 (6) | 2 | 10 |
| 1 | 35 (24) | 17 | 31 | |
| 2 | 68 (47) | 38 | 55 | |
| 3 | 26 (18) | 12 | 24 | |
| 4 | 7 (5) | 1 | 8 | |
| 5 | 1 (1) | 0 | 2 | |
| CHA2DS2-Vasc scale | 0 | 3 (2) | 0 | 4 |
| 1 | 6 (4) | 1 | 7 | |
| 2 | 24 (16) | 10 | 22 | |
| 3 | 44 (30) | 23 | 38 | |
| 4 | 47 (32) | 25 | 40 | |
| 5 | 17 (12) | 6 | 17 | |
| 6 | 4 (3) | 0 | 5 | |
| 8 | 1 (1) | 0 | 2 | |
| HAS-BLED scale | 0 | 4 (3) | 0 | 5 |
| 1 | 18 (12) | 7 | 18 | |
| 2 | 65 (45) | 36 | 53 | |
| 3 | 44 (30) | 23 | 38 | |
| 4 | 11 (8) | 3 | 12 | |
| 5 | 4 (3) | 0 | 5 | |
| EPS: Health insurance company; NVAF: Nonvalvular atrial fibrillation; ASA: Acetylsalicylic acid; CAD: Coronary artery disease; HTN: Arterial hypertension; CHF: Congestive heart failure; DM2: Type 2 diabetes mellitus; CVA: Cerebrovascular accident; CKD: Chronic kidney disease; PVD: Peripheral vascular disease; LVEF: Left ventricular ejection fraction; GFR: Glomerular filtration rate; NOAC: Non-vitamin K antagonist oral anticoagulants; VKA: Vitamin K antagonists. | ||||
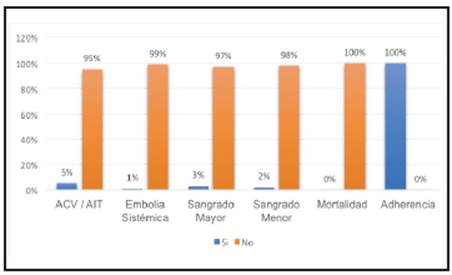
Finally, regarding clinical outcomes, 95% (n=139) of the patients did not have a CVA or TIA, 99% (n=144) of the patients did not have a systemic embolism, and 98 (n=143) and 97% (n=141) of the patients did not have minor or major bleeds, respectively. Within the major hemorrhages, a fatal bleed, a gastrointestinal bleed and an intracranial hemorrhage were documented in 3% (n=5) of the participants. One hundred percent of the patients adhered to the prescribed treatment (Figure 1).
Relationship between sociodemographic and clinical characteristics
Applying the Pearson correlation matrix for continuous variables, significant correlations (p < 0.05) were found between age and weight loss in 27.4%, BMI in 23.5% and GFR in 44.3%. Regarding the variable of weight, significant correlations were found with age as previously described: BMI with a 75.1% increase, a 28.7% increase in serum creatinine and a 48.8% increase in the glomerular filtration rate. The glomerular filtration rate is correlated, increasing 42.8%. Serum creatinine is significantly correlated with a 54.9% reduction in GFR and a 28.7% increase in weight. Finally, the glomerular filtration rate is correlated with most of the variables in significant percentages; it decreases with age, increases with weight and BMI, and is correlated with a 54.9% reduction in serum creatinine.
The contingency tables and Chi2 test showed statistically significant (p< 0.05) associations between the cerebrovascular accident or transient ischemic attack variable (CVA/TIA, primary efficacy outcome variable of the phase III randomized clinical trials) and the CHADS2, CHA2DS2-VASc, and HAS-BLED scores, which supports the idea that patients with atrial fibrillation and comorbidities have a greater risk of both thrombotic and bleeding events. The type of AF is significantly associated as is the history of CVA/TIA (p<0.05). It is important to note that associations were found between this variable and the type of major bleed and minor bleeds in the analyzed cohort.
The major and minor bleed variables (primary safety outcome variables in phase III clinical trials) were significantly correlated with the HAS-BLED score (p<0.05), in line with the current scientific evidence which supports the fact that patients with high HAS-BLED scores have a greater probability of bleeding. Finally, there were no fatalities in the cohort and all the patients analyzed adhered to treatment.
Discussion
Atrial fibrillation is associated with high morbidity and mortality, as it increases the risk of cerebrovascular diseases like CVA/TIA. In this study of 146 medical charts, the mean age of 77 years and more prevalent female sex, permanent-type NVAF and high blood pressure as the most frequent comorbidity were characteristics similar to those reported in the scientific literature 9,10. For instance, the AFINVA study had a mean age of 77.4 years, more than half of the participants were female, there was a 60.8% frequency of permanent-type AF, and 78% of the participants had high blood pressure as a comorbidity 9. Also, L.Y. Mantilla-Villabona et al.'s study had an average participant age of 76.47 years, 60.80% were female, 82.41% had NVAF and, of these, 84.7% had associated high blood pressure 10.
Regarding treatment, 99% of the patients were treated with NOACs (76% of whom received rivaroxaban), 84% were not taking ASA and 98% were not being treated with clopidogrel, in line with the international clinical practice guidelines 3,6,7. However, according to the CHADS2 and CHA2DS2-VASc classification, 6% of the patients classified as 0 and 1 received anticoagulant treatment, despite this not being currently recommended. Likewise, of the 146 patients analyzed, most (45%) had a HAS-BLED 2 classification. Of the information provided, it should be noted that the treatment selected for the study participants was aligned with the patient treatment guidelines which establish that antiplatelet medications are not indicated for this type of patients. This shows progress compared to the treatment in Latin America reported in the insights from the GARFIELD-AF registry which documented monotherapy with AP medications in 24% of the patients and AP medications together with VKAs or NOACs in 61% of the cases 8.
Regarding clinical outcomes, more than 90% of the patients did not have a CVA/TIA, systemic embolism, or major or minor bleed, respectively. However, a statistically significant association (p< 0.05) was found between the CVA/TIA variable and the CHADS2, CHA2DS2-VASc, and HASBLED scores, which supports the idea that patients with AF and comorbidities have a higher risk of both thrombotic and bleeding events.
We recognize that the design selected for this study and the data obtained from the medical charts may have introduced information biases. However, these are not determinant.
The study was carried out in a population from the department of Tolima who were seen at a tertiary care institution in the city of Ibagué, and therefore the results cannot be extrapolated to the entire Colombian population. However, a hypothesis can be made regarding the clinical behavior, comorbidities and treatment of Colombian patients with NVAF.
Based on the results, it can be concluded that the documented choice of NOACs for treatment aligns with the international guidelines and, just like those reported in the literature, patients with NVAF at Instituto Cardiovascular del Tolima (Ibagué, Colombia) are older and have multiple comorbidities. Likewise, there is a growing use of NOACs and decreased use of AP medications, in line with the international recommendations.











 text in
text in