INTRODUCTION
Salinization due to the use of irrigation water with a high salt content affects more than 20 % of irrigated areas and reduces crop yield (Sako et al. 2018). Increasing demand and freshwater scarcity intensify the use of saline water to irrigate agricultural crops in many regions of the world, especially in arid and semi-arid regions (Batista et al. 2019; Dastranj and Sepaskhah, 2019).
Salinity is one of the main factors responsible for reducing agricultural production (El-Nasharty et al., 2019; Li et al., 2019; Astaneh et al., 2019). The adverse effect of salinity is mainly due to the accumulation of toxic ions (Na+ and Cl-) causing oxidative stress and decreasing osmotic potential in plants (Shams et al., 2019).
Initially, plants exposed to salinity undergo osmotic stress, with inhibition of cell division and limiting gas exchange and, if continued, may undergo ionic stress, causing nutritional imbalance, oxidative stress and inhibition of protein synthesis and enzymatic activity, reducing plant growth, development and survival (Bekhradi et al., 2015; Cirillo et al., 2019). The photosynthetic capacity of plants grown under saline conditions is lower depending on the severity and/or duration of stress, genotype and plant age (Sarabi et al., 2019).
Saline stress can disrupt the electron transport chain, reducing photosystem II efficiency and increasing fluorescence emission (Melo etal., 2017). Processes in and around PSII reaction centers change receptor redox balance and quantum energy yield and affect chlorophyll fluorescence (Bordenave et al., 2019).
Salicylic acid (SA) influences various physiological and biochemical functions in plants and has diverse effects on the tolerance to biotic and abiotic stress (Li et al., 2014; Silva et al., 2022). Exogenous SA can improve plant adaptation to salinity by various mechanisms, such as ameliorating photosynthetic capacity, increasing antioxidative protection, inhibiting Na+ and Cl- accumulation, accumulating soluble carbohydrates, stimulating ABA accumulation, improving mineral nutrient, enhancing N and S assimilation etc. (Nazar et al., 2011; Poór et al., 2011; Hao et al., 2012). SA alleviated the adverse effects of saline stress in mungbean (Vigna radiata - Fabaceae) through the improvement of plant photosynthesis, and growth and enhancing the antioxidant system (Khan et al., 2014).
Basil (Ocimumbasilicum L. - Lamiaceae), native to India, Africa, and Southern Asia (Tavallali et al., 2019), an annual or perennial herbaceous plant depending on where it is grown (Blank et al., 2015), has medicinal, seasoning, aromatic or ornamental use in pots and gardens (Blank et al., 2012). This plant is used as a condiment, and its essential oil, composed of a complex mixture of volatile and semi-volatile compounds that determine its specific aroma and the flavor of the condiment (Lung et al., 2016), can be used in the chemical, pharmaceutical and cosmetics industries (Jakovljevic et al., 2017). The aim of the present study was to evaluate growth, gas exchange, chlorophyll a fluorescence and chlorophyll indices of basil (cv. Cinnamon) plants under saline stress and salicylic acid.
MATERIAL AND METHODS
Experimental conditions and plant material
This experiment was carried out in a protected environment at the Center for Agricultural Sciences of the Universidade Federal da Paraíba in the municipality of Areia, Paraíba state, Brazil. The soil used was classified as Planosols, of a sandy loam texture, whose physical characteristics were sand: 756.9 g kg"1; silt: 59.1 g kg-1; clay: 184.0 g kg-1; bulk density: 1.38 kg dm-3; particle density: 2.67 kg dm-3; total porosity: 48 %; field capacity: 78 g kg-1; and permanent withering point: 43 g kg-1. The salinity of the water used was analyzed (Table 1).
Table 1 Analysis of saline water with conductivities, from 0.50 to 6.00, used for irrigation of basil cv. Cinnamon
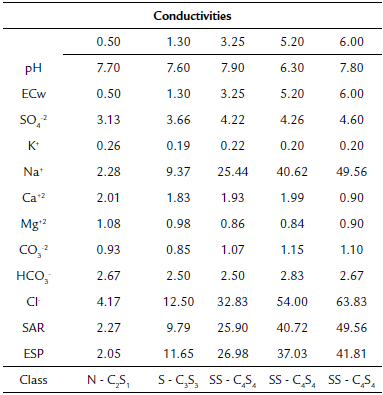
Class= classification; N= normal; S= saline; SS= sodium saline; SAR= sodium adsorption ratio; ESP= exchangeable sodium percentage.
The basil seedlings were produced in 162-cell polyethylene trays and, after 25 days of planting, transplanted into 5.0 dm3 pots with 100 g poultry manure. The chemical analysis of this mixture was: pH: 6.9; P: 11.71 mg dm-3; K+: 873.43 mg dm-3; Na+: 0.24 cmol dm-3; H++Al+3: 1.6 cmol dm-3; Al+3: 0.00 cmol dm-3; Ca+2: 4.65 cmol dm-3; Mg+2: 0.39 cmol dm-3; sum of bases: 7.52 cmol dm-3; cation exchange capacity: 9.12 cmol dm-3; base saturation: 82.45 %; and organic matter: 22.73 g dm-3.
The basil plants were irrigated with saline water after transplantation. The irrigation water with the desired electrical conductivity (ECw) was a mixture of NaCl, CaCl2.2H2O, and MgCl2.6H2O (7:2:1) salts in non-chlorinated water (0.5 dS m-1) obtained at the experiment site. The total water used for irrigation was defined as four plants in the soil with ECw of 0.5 dS m-1.
EXPERIMENTAL DESIGN
The experimental design was a randomized block design in a 5x5 incomplete factorial scheme generated through the central composite design. The factors were five electrical conductivities of irrigation water (ECw- 0.5, 1.3, 3.25, 5.2, and 6.0 dS m-1) and five doses of salicylic acid (SA- 0.0, 0.29, 1.0, 1.71 and 2.0 mM), with five replications and two plants per replicate.
GROWTH MEASUREMENT
Plants were evaluated 60 days after the application of salt stress. Plant height (cm), number of branches, number of leaves, stem diameter (mm), inflorescence dry mass (g), leaf dry mass (g), stem dry mass (g), leaf area (cm2), leaf area ratio (cm2 g-1), specific leaf area (cm2 g-1), stem mass ratio (g g-1), root mass ratio (g g-1), robustness quotient, sclerophilia index and root/shoot ratio were evaluated. The plants were divided into roots, stem, and leaves and dried in an oven with forced air circulation at 65 °C for 72 hours. Then, the plant parts were weighed on a precision scale (0.001 g).
The leaf area ratio was obtained using the formula: leaf area (LA)/total dry mass (TDM). The specific leaf area was obtained using the formula: LA/leaf dry mass (LDM). The stem mass ratio was obtained using the formula: stem dry mass/TDM. The root mass ratio was obtained using the formula: root dry mass/TDM. The robustness quotient was obtained using the formula: plant height/stem diameter. The sclerophilia index was obtained using the formula: LDM/LA.
GAS EXCHANGES MEASUREMENT
Gas exchange was determined at 60 days from the beginning of saline irrigation (DAI) in an infrared gas analyzer (LI-COR® - model LI-6400XT, Nebraska, USA), with measurements made between 9 and 10 a.m. Stomatic conductance (gs- mol H2O m-2 s-1), transpiration (E- mmol H2O m-2 s-1), net photosynthesis (A- µmol CO2 m2 s-1), internal carbon concentration (Ci- (µmol CO2 mol air1), vapor pressure deficit (VPD), instantaneous water use efficiency (WUE- A/E), intrinsic water use efficiency (iWUE-A/gs) and intrinsic carboxylation efficiency (iCE- A/Ci) were then measured gas exchanges.
Chlorophyll a fluorescence measurement
Chlorophyll a fluorescence was evaluated at 60 DAI on the third leaf, using a non-destructive method, with a fluorometer (Opti-Sciences Inc.-Model OS-30p, Hudson, USA) with leaf clamps placed for 30 minutes before readings for adaptation of the leaves in the dark. The initial fluorescence (F0), maximum fluorescence (Fm), and quantum yield of photosystem II (PSII= Fv/Fm) was the chlorophyll fluorescence variable evaluated.
RESULTS
The harmful effects of saline stress up to 1.3 dS m-1 on the inflorescence dry mass (idm), plant height (ph), stem diameter (sd), leaves dry mass (ldm), root dry mass (rdm), stem dry mass (sdm) and a number of leaves (nl) were attenuated with the application of 0.29- and 1.71-mM salicylic acid (Fig. 1a). The harmful effects of irrigation water salinity above 1.3 dS m-1 on basil growth were not mitigated by the application of salicylic acid.
The harmful effects of saline stress up to 1.3 dS m-1 on the stem mass ratio (smr), leaf area (la), Dickson's quality index (dqi), sclerophilia index (si), and root mass ratio (rmr) was attenuated with the application of 0.29 and 1.71 mM of salicylic acid (Fig. 1b). The exogenous application of 1.0 and 1.71 mM of salicylic acid attenuated the harmful effects of salinity on the leaf area ratio (lar), specific leaf area (sla) and robustness quotient (rq).
The harmful effects of saline stress up to 5.2 dS m-1 on Ci, gs, E, and A were mitigated by the application of salicylic acid above 0.29 mM (Fig. 2a). The exogenous application of 1.0 mM of salicylic acid attenuated the harmful effects of saline stress on WUE, VPD, and iWUE.
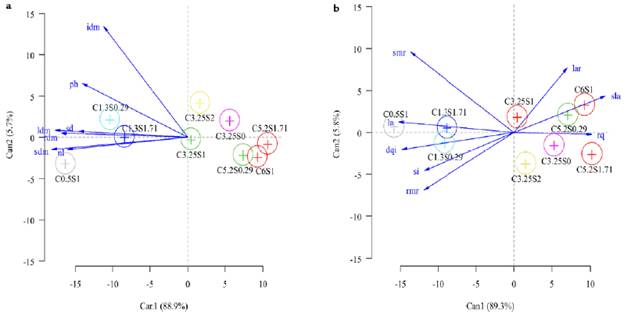
Figure 1 Canonical variables analysis and confidence ellipses between growth variables (a and b) of Ocimum basilicum under saline stress and salicylic acid. C= electrical conductivities of irrigation water; S= salicylic acid; idm= inflorescence dry mass; ph= plant height; tdm= total dry mass; sd= stem diameter; ldm= leaf dry mass; sdm= stem dry mass; nl= number of leaves; smr= stem mass ratio; la= leaf area; dqi = Dickson's quality index; si= sclerophilia index; rmr= root mass ratio; lar= leaf area ratio; sla= specific leaf area; rq= robustness quotient.
The application of salicylic acid up to 1.71 mM attenuated the harmful effects of saline stress on maximum fluorescence (Fm) and quantum yield of photosystem II (Fv/ Fm), chlorophylls a and b of basil plant under ECw of 5.2 and 6.0 dS m-1 (Fig. 2b). Saline stress up to 3.25 dS m-1 favored the highest chlorophyll indices (a and b).
DISCUSSION
Salicylic acid (SA) attenuated the deleterious effects of moderate saline stress (ECw 1.3 dS m-1) on the growth of basil plants because this phytohormone increases stimulation in physiological and biochemical processes (Jini and Joseph, 2017), in addition to increasing the K+ content and decreasing Na+ accumulation in the shoot of plants (Jayakannan et al., 2013). SA decreased the harmful effects of saline stress in rice plants (Oriza sativa - Poaceae) (Jini and Joseph, 2017) and periwinkle (Catharanthus roseus -Apocynaceae) (Idress et al., 2011).
SA relieved the damaging effects of salt stress (up to 5.2 dS m-1) on Ci, gs, A, and E due to its effect on increasing photosynthetic rate, carbon fixation, transpiration, stomatal conductance, and antioxidant activity (Jayakannan et al., 2015). In addition, the positive effect of SA on A may be due to its stimulatory effects on the pigment contents and Rubisco enzyme activity (Li et al., 2014). The reduction of gas exchange by saline water irrigation with high ECws is due to the high concentration of Na+ and/or Cl- accumulated in chloroplasts (Shahbaz et al., 2016). These ions, when accumulated in chloroplasts, reduce stomatal opening and, consequently, photosynthesis, with lower CO2 diffusion in the substomatic chamber (Bybordi, 2012; Silva et al., 2015), as reported for two basil cultivars (Attia et al., 2011). In addition, excess salts in the transpiratory stream (Bekhradi et al., 2015) accumulate reactive oxygen species and cause severe oxidative damage (El-Esawi et al., 2018). The lack of effect of ECw on net photosynthesis (A) may be associated with mechanisms of restriction of Na+ uptake and recycling in the xylem stream to the root system and sequestration of this ion in vacuoles and exportation outside cells (El-Esawi et al., 2018).
The increase in the Fv/Fm under high ECw is related to the tolerance of this Cinnamon cultivar to water salinity stress with adjustment mechanisms in electron transport and entrapment in the PSII reaction center (Kalaji et al., 2016). Stress response may vary according to the species' adaptive capacity (Rocha et al., 2016). These may also be related to net photosynthesis (Li et al., 2019) and plant physiological responses to salt stress that depend on the phenotypic plasticity and genotypic characteristics of each plant (Batista et al., 2019). The average Fv/Fm of 0.78 indicates the lack of damage to the PSII, as the plant Fv/Fm under stress is lower than 0.75 (Silva et al., 2015). Plants, under saline conditions, adapt by osmotic adjustment, osmoprotectant accumulation, and modifications in ion transport to prevent ionic toxicity and enzyme activation (Javed et al., 2019).

Figure 2 Canonical variables analysis and confidence ellipses between gas exchange variables (a) and chlorophyll variables (b) of Ocimum basilicum under saline stress and salicylic acid. C= electrical conductivities of irrigation water; S= salicylic acid; gs= stomatal conductance; A= net photosynthesis; E= transpiration; Ci= internal carbon concentration; WUE= instantaneous water use efficiency; iWUE= intrinsic water use efficiency; iCE= intrinsic carboxylation efficiency; F0= initial fluorescence; Fm= maximum fluorescence; Fv/Fm= quantum yield of photosystem II.
The increase in chloroplast pigments under the SA treatment might be due to the ability of SA to increase the activity of certain enzymes, thereby stimulating chlorophyll biosynthesis or reducing chlorophyll degradation, leading to increased net photosynthesis under saline stress tolerance (Li et al., 2014). In addition, the positive effect of SA on photosynthetic pigments could be attributed to its stimulatory effects on RuBisCO activity and the rate of photosynthesis (Idrees etal., 2012). The increase in chlorophyll a, b, and total levels with ECw are due to the intensity/duration of salt stress on photosynthetic pigment biosynthesis, chlorophyllase activity, and protein complex instability, and the maintenance of chlorophyll accompanied by the conservation of photochemical reactions, like those of the PSII (Huang et al., 2015; Elhindi et al., 2016). The decrease in dry mass by ECw is due to Na+ accumulation and reduction in the K+/Na+ ratio (Kaushal and Wani, 2016) and reduced water absorption capacity due to osmotic stress in basil plants (Morales et al., 2012).















