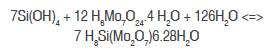In analytical laboratories, where the nutritional elements are quantified, the determination of available silicon is affected by the P interference, making this the main problem in the determination of silicon by colorimetry. Atkins and Wilson (1926) reported that the P does not interfere with colorimetric determination of silicon, but Jimenez-Prieto and Silva (1998) explained that the main drawback in the determination of phosphorus and silicon are mutual interference of both species, which strongly influences the performance of analytical methods.
In the quantification of silicon and phosphorus concentrations in soil samples by colorimetric methods is used the reaction that occurs between phosphates and silicates with molybdate in an acidic medium, this forms the corresponding yellow heteropolymolybdates and occurs the subsequent reduction to molybdenum blue. But in this process there is interference between both species, which strongly influences the performance of analytical methods. This interference is because the ammonium molybdate is capable of forming phosphomolybdates complexes, which absorb the same wavelength in that the silicomolybdate.
Schwahtz (1942), Bunting (1944) and Case (1944) carried out studies to control the interference by P in quantifying silicon with organic acids oxalic, tartaric and citric by eliminating phosphomolybdate, to determine the silicon content in copper alloys and silicate rocks (Carlson and Banks (1952), Milton (1951) and Shell (1962) found that the silicomolybdate was stable with the addition of organic acids, only if the acid is added after the complex formation, also they found that it is possible to eliminate it after the complete formation of silicon-molybdate.
Jolles and Neurath (1988) developed the method of molybdate in the determination of inorganic silicon in water and soil; they additionally indicated that for the determination of silicon is necessary to employ the colorimetric method because it is very sensitive. This is based on the formation of silicomolybdate (SiO2.12MoO3), as shown in the following equation:
In recent years studies about silicon chemistry have been intensified and the probable because this is an beneficial element for plants, several studies have showed benefits in crops, especially when plants are subject to different kind of stress, resistance to adverse factors (biotic and abiotic), which contribute to increase crop productivity specially grasses (Pulz et al., 2008); (Crusciol et al., 2009; Datnoff et al., 2001).
In South America and Colombia there are few studies that have been conducted to determine the real value of organic acids in phosphate removal during Si determination. Although many scientists have carried out research from the 1940's to the 1980's and determined that tartaric acid efficiently eliminated the interference caused by P in the analysis of silicon. Currently Korndorfer et al. (2004) eliminates this interference with tartaric acid.
Moreover, when it is need to determine and quantify the content of available silicon in soil and water other organic acids have showed similar or even greater efficiency in eliminating the P interference in soils containing high concentrations of phosphorus and silicon. Rocha et al. (2005), indicated that tropical soils are characterized by a high degree of weathering and low content of available phosphorus but due to the fact that currently the available silicon is quantified in soils with high content of P, different acids fulfill the same function of the tartaric acid. The aim of this study was to evaluate the effects of different organic acids to eliminate the interference caused by phosphorus in the chemical analysis of silicon.
MATERIALS AND METHODS
Four different organic acids (tartaric, citric, oxalic and malic acids) were used at two concentrations (0.8 and 1.33 mol L-1 as suggested by Korndorfer et al. (2004) and Freitas and Da Gloria (1976).
Six solutions containing silicon at 1 mg L-1 were prepared separately with six concentrations of phosphorus (0.0, 0.2, 0.4, 0.6, 0.8 and 1.0 mg L-1). The objective of that each experimental unit was to keep the same concentration of silicon with different concentration of P. This procedure was performed with four repetitions.
From these solutions were taken aliquots of 10 mL, which were added in 50 mL plastic cups. According to Korndörfer et al. (2004) to each aliquot was added 1 mL of sulfomolibdica solution for the complex formation of α-molibdosilicato (yellow color), than occurrs after 5 min at pH 1.4-2.0.
After 10 min elapsed of adding the sulfomolibdica solution, 2 mL of organic acids at 1.33 mol L-1 were added to eliminate the P interference as proposed by Korndörfer et al. (2004). Also the concentration of organic acids 0.8 mol L-1 was added to an aliquot of 6 mL for each acid as suggested by Freitas and Da Gloria (1976).
During this process, it was necessary to wait 5 min for the corresponding reactions; that eliminate the fosfomolibdate complex, which are the causative agents of this interference. After this reaction 10 mL of ascorbic acid (3 g L-1) were added to reduce molibdosilicate yellow into the molybdenum blue complex. One hour later readings were made at a spectrophotometer Perkin Elmer lambda XLS + at 660 nm.
The data were analyzed using analysis of variance, mean test, and contrast analysis and pairwise tests using the statistical software R (Development Core Team, version 3.2.2)
RESULTS AND DISCUSSION
It was found that organic acids did not eliminate the interference caused by P, especially as its concentration increased. The acids used followed the same trend of the reference acid (tartaric acid) proposed by Korndorfer et al. (2004) (Figure 1).
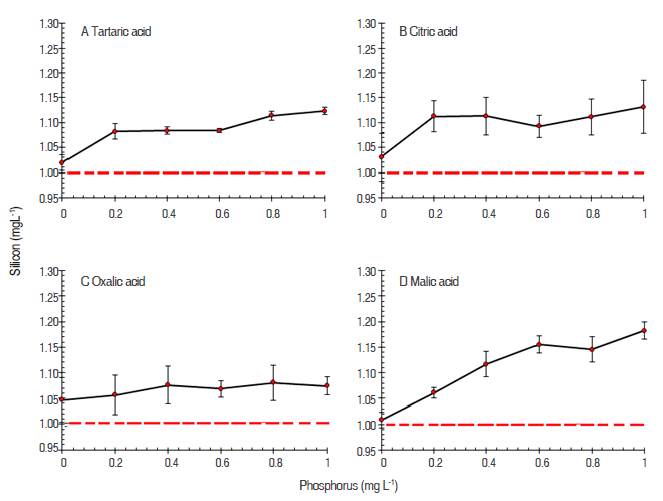
Figure 1 Silicon concentration (mg L-1) determinated in the presence of increasing concentrations of phosphorus and four organic acids at 1.33 mol L-1: A Tartaric acid, B citric acid, C oxalic acid and D malic acid.
The organic acids used at 1.33 mol L-1, in average overestimated the silicon concentration of 1 mg L-1 by tartaric acid0.08 mg L-1, oxalic acid by 0.07 mg L-1, citric acid 0.1 mg L-1 and the malic acid 0.11 mg L-1.
It should be noted that as the P concentration increased in the Si solution, the organic acids used at concentration of 1.33 mol L-1 overestimate the amount of Si initially applied. It is possible to consider that at this concentration the acids did not destroy the α- phosphomolybdate complex; in this way, quantification the α- phosphomolybdate complex is quantified as α-molibdosilicato. Since these complexes have additive effects there are overestimations on the concentration of silicon.
According to Galhardo et al. (2000) the elimination of the P interference by organic acids can be explained by a ligand exchange reaction with phosphomolybdate complex. This produces phosphate and molibdo - acid (acid used for removing interference) while the silicomolybdate complex undergoes not change. This complex is relatively inert and does not allow that to occur a ligand exchange with the acid used.
It is clear the oxalic acid at 1.33 mol L-1 was the acid that presented the most effective to stabilize the silicon quantization, in despite of the increasing concentration. Dabin et al. (1968) recommended oxalic acid to remove phosphorus interference.
It was also found that at the concentration of 0.8 mg L-1 the tartaric acid overestimates the silicon concentration of 1 mg L-1 in 0.1 mg L-1 and the citric acid in 0.15 mg L-1.
According to Carpenter et al. (1997) the rate of complex formation of molybdosilicate and molybdophosphate depends on pH and temperature, also the formation of molybdophosphate is completed in less than 1 min and the rate increases in the more acidic solutions, while the formation of molybdosilicate would be slower (10 min), but it decreases in more acidic solutions.
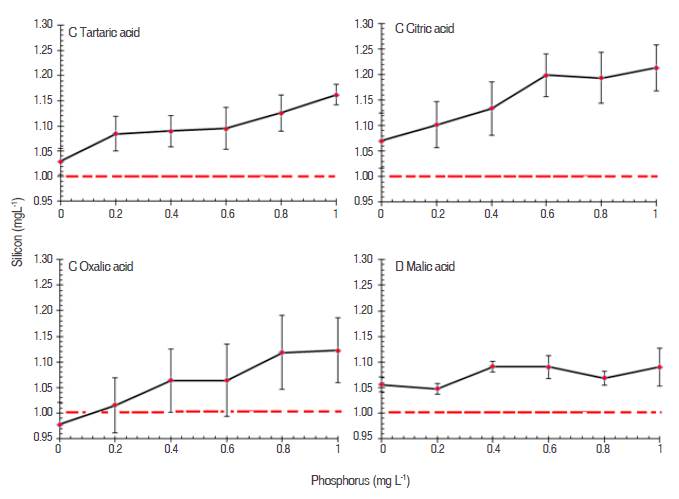
Figure 2 Silicon concentrations (mg L-1) determined in the presence of increasing concentration of phosphorus and four organic acids (at 0.8 mg L-1): A Tartaric acid, B citric acid, C oxalic acid, and D malic acid.
At this concentration (0.8 mg L-1), the acids with higher capacity of breaking the phosphomolybdate complex were oxalic and malic; in the presence of both acids the Si overestimated was 0.06 and 0.07 mg L-1, being the malic acid the most effective in the elimination of interference along the increasing gradient of P.
These results indicate the limited capacity of these acids to eliminate the interference by phosphorus at a concentration 1.33 mol L-1 in the quantification the available silicon (Table 1). According to Dabin (1968) and Mysumi and Tarutani (1961) the organic acids for eliminate interference phosphomolybdate are the oxalic and tartaric acids.
Table 1 Test DMS for the silicon content with different methods, using different doses of phosphorus.
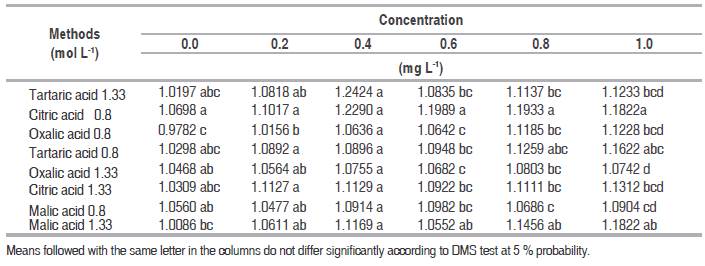
Similarly by comparing the results of the initial concentration of silicon (1 mg L-1) with the silicon concentration obtained with organic acids at a concentration of 0.8 mol L-1 was determined than the values obtained the available silicon were equals statistically between the same dose of each acid, when compared to the data obtained with tartaric acid at this concentration. These results indicate that the organic acids used have the same ability to neutralize interference by phosphorus, that tartaric acid at concentrations of 0.8 and 1.33 mol L-1.
These results may be explained by the ability of these organic acids have of ligand exchange with the phosphomolybdate complex, which is the causative agent of interference in the analysis of silicon. Galhardo et al. (2000) demonstrated than the quantification of phosphates and silicates by sequential injection, through spectrophotometric determination using the molybdenum blue chemical method, was possible eliminate interference caused by phosphates using oxalic acid.
To verify the test results obtained with the least significant difference (LSD), orthogonal against (tartaric acid) were performed (Table 2).
Table 2 Contrasts averages for the silicon content of eight organic acids and six doses phosphorus in removing phosphorus chemical interference.
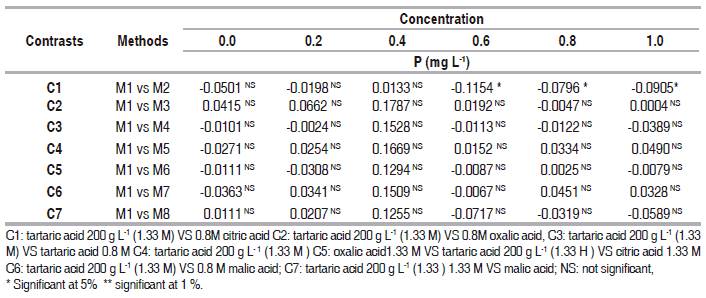
It was determined that there were not significant differences among the effect of tartaric acid and C2, C3 , C4 , C5 , C6 and C7 at lower doses of 0.4 mg L-1 of phosphorus added. However, at the P concentrations of 0.6, 0.8, and 1.0 mg L-1 there were significance differences among tartaric acid and other organic acids used. This showed that the organic acids have greater ability to eliminate interference by phosphorus than the tartaric acid at a concentration the 1.33mol L-1 in the analysis of available silicon.
The malic acid can be used as an alternative to eliminate interferences caused by phosphorus. Chalmers and Sinclair (1966), indicate that the use of tartaric acid, to mask phosphate interference in determining silicate reduces the sensitivity of the method. This is likely due to a marked change in the extinction coefficient of phosphomolybdate formed and the electrochemestry's factors that determine its response should be very different.
This results should be corroborated to check the use of malic acid in quantifying silicon by molecular adsorption. In addition, the malic acid is more economical compared to tartaric acid.
CONCLUSIONS
The malic acid at a concentration of 0.8 and oxalic acid at 1.33 mol L-1 can be used to eliminate the interference of phosphorus in the quantification available silicon.
The organic acids had less overestimation of available silicon when there were concentration of P larger than 0.6, 0.8, and 1.0 mg L-1, than the tartaric acid used as reference acid.