INTRODUCTION
Metanephric adenoma (MA) is a rare benign tumor that originates in renal epithelial cells and is usually detected as an incidental finding 1. This neoplasm is difficult to differentiate from malignant tumors affecting the upper urinary tract in the pre-surgical stage.
The definitive diagnosis of MA is established based on immunohistochemistry and histopathological findings. However, it has been proposed that the presence of solid, single, well-defined, oval-shaped, unilateral lesions predominantly in the renal medulla and without extrarenal involvement in imaging studies is a useful finding to differentiate it from other tumors. This finding also allows considering MA as a differential diagnosis in patients with upper urinary tract masses and thus avoid ablative treatment in the affected kidney and ureter 2.
The following is the case of a patient who underwent right radical nephroureterectomy due to clinical suspicion of high-risk upper urinary tract urothelial carcinoma, whose final diagnosis was MA according to the histopathological study.
CASE PRESENTATION
A 51-year-old mixed-race woman from Bogotá D.C. (Colombia), housewife, from a lower-middle income household and without a relevant medical history, presented to the outpatient clinic of the urology service of a public tertiary healthcare center in Bogotá with mild to moderate bilateral lumbar pain of non-specific characteristics that lasted three months. The patient was referred by the general medicine service, which had previously requested a renal/urinary tract ultrasound that showed a right renal mass with ipsilateral grade I hydronephrosis.
During the history-taking process, the patient was asymptomatic, and no relevant findings were found on the physical examination. Consequently, a computed tomography (CT) urography was ordered, and the results were analyzed at the follow-up appointment 2 weeks after the initial assessment. CT showed a 44x44x28mm lesion in the right renal pelvis with invasion of the renal parenchyma at the lower pole and adenopathies in the para-aortic nodes that measured up to 17mm (Figures 1 and 2). Given this finding, a CT scan of the chest (assessed in a second follow-up appointment) was requested, finding no metastatic involvement, as well as a serial urinary cytology with findings consistent with a type II category lesion according to the Paris classification (negative for high-grade urothelial carcinoma) 3. In addition, the glomerular filtration rate (GFR) estimated with the CKD-EPI equation at that time was 101 mL/min/1.73 m2.

Source: Document obtained during the study.
Figure 1 Computed tomography (CT) urography. Nephrogenic phase; axial plane.

Source: Document obtained during the study.
Figure 2 Computed tomography (CT) urography. Excretory phase; coronal plane.
Due to the high suspicion of high-risk upper tract urothelial carcinoma (staging: cT3N1M0 according to the 2017 TNM classification), the presence of hydronephrosis, and the detection of adenopathies on the CT scan, during the second follow-up appointment, the patient was considered a candidate for a transurethral cystoscopy and then right radical nephroureterectomy with bladder cuff and right retroperitoneal lymph node dissection with curative intent. The surgery was scheduled to be conducted in an outpatient clinic with the urology department one month following the initial assessment.
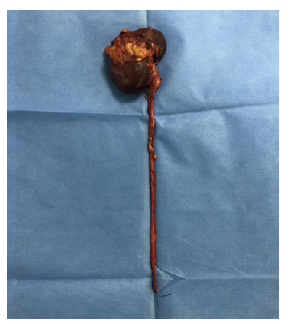
Transurethral cystoscopy revealed no endoluminal bladder lesions, however during surgery, a tumor of approximately 5cm in diameter was discovered in the right renal pelvis with no macroscopic involvement of adjacent tissues; no retroperitoneal adenopathies were observed on macroscopic examination (Figures 3 and 4). It should be noted that nephroureterectomy with bladder cuff was performed using the intravesical approach.

Source: Photographs obtained during the study.
Figure 4 Approach to the area of interest (in red). Projection of the mass contained in the right renal pelvis.
The patient presented no postoperative complications, her course was satisfactory, and she was discharged three days after the procedure. The pathology report showed a mixed epithelial and stromal tumor of the kidney with free borders, and immunohistochemistry confirmed the presence of a MA (positive for CD57, vimentin, and WT1; negative for CK7 and CD10).
During the first postoperative follow-up, 3 weeks after the procedure, the patient was asymptomatic and had a GFR according to the CKD-EPI formula of 85 mL/min/1.73m2. In the second follow-up, 6 months after surgery, the patient was still asymptomatic, and no tumor relapse was observed in the CT of the abdomen and pelvis.
DISCUSSION
MA is a rare renal neoplasm that accounts for 0.2% of adult renal epithelial tumors and has less than 200 cases reported in the literature 1,4. This type of tumor, according to Jiang et al.1, was classified by the World Health Organization in 2016 as a subtype of metanephric neoplasm, which also includes adenofibroma and stromal tumors.
MAs can occur at any age, but they are most common in patients aged 50 to 60 years, with a female-to-male ratio of 2-2.6:1 1,4. Genetic analyzes have revealed, as reported by Ding et al.5, that the missense mutation of BRAF V600E could be detected in approximately 90% of these types of neoplasms.
Patients with MA usually have no symptoms, although lower back pain, gross hematuria, and fever occur in 10-50% of cases 2,6. In addition, polycythemia has been reported in about 12% of MA patients with hematuria 1.
MAs have a low rate of diagnostic certainty using imaging and the diagnosis is usually incidental, which may be explained by the low suspicion and prevalence 2. Similarly, these tumors are difficult to differentiate in the pre-surgical stage from other malignancies such as Wilms' tumor, metastatic papillary thyroid carcinoma, and papillary renal cell carcinoma 7.
The imaging characteristics described for MAs in small case series, such as Zhu et al.2 in 8 patients with MA who were hospitalized at the Subei People's Hospital (China) between 2006 and 2011, refer to solid, single, well-defined, oval-shaped, unilateral lesions predominantly in the renal medulla and without extrarenal involvement. Ultrasound can detect hypo- or hyperechoic lesions with lower Doppler flow compared to other tumors 1. On nuclear magnetic resonance imaging, these neoplasms are hypointense with delayed enhancement of contrast material in T1W1 sequence and hyperintense in DWI sequence 1. Finally, the predominant findings on CT scan are lesions with well-defined boundaries with enhancement, calcifications (20% of cases), and homogeneous enhancement that is lower compared to that of renal parenchyma in all enhanced phases 1,8.
The origin of MAs in epithelial cells of the renal collecting duct explains the imaging finding of invasion or displacement of the collecting system and suggests a differential diagnosis with urothelial carcinoma of the upper urinary tract 1,2. However, Li and collaborators, cited by Yan et al.9, found that 83.3% of tumors of this type were located in the periphery of the renal cortex without involvement of the collecting system.
The characteristics of MAs have not been widely studied, but Jiménez-Hef-fernan et al.10, in their letter to the editor, established that cytology, either as fine-needle aspirates or during intraoperative procedures, can be quite useful in identifying these tumors. Thus, the characteristic cytology findings are generally small, clustered cells, oval or round, with uniform basophilic nuclei and without nucleoli and scant cytoplasm; the absence of nuclear atypia, necrosis, and mitosis are also common findings in patients with MA 7,11. Taking into account these histopathological findings in cytological studies, it is necessary to consider Wilms' tumor and papillary renal cell carcinoma as differential diagnoses 7,10,11.
On the other hand, as reported by Le Nué et al.12, MA diagnosis may be supported by core-needle renal biopsy. If biopsy is performed hemostasis must be double checked, as there is a possible association with acquired von Willebrand disease, as described in two pediatric cases.
MAs usually have a good prognosis, and the treatment of choice is complete surgical resection by partial or radical nephrectomy, depending on size; definitive diagnosis is established based on histopathological studies and immunohisto-chemistry 13-15.
Macroscopically, MA is described as a well-circumscribed brownish-yellow mass and with the presence of pseudocapsule 4,7,8. Microscopically, these neoplasms are comprised of tubules of different sizes and shapes, divided into lobes with fine fibrovascular septa; in addition, their cells are characterized by small cell nuclei and very low mitotic activity 2,5. These tumors have the following immunohistochemical pattern: positive for vimentin, pancytokeratin, WT1 and CD57; negative for CK7, AMACR, and CD10 2,5.
Li et al.16 conducted a cohort study of 18 patients with MA in which, through pathological examination, they found that 6 (33.3%) tumors had other carcinoma components concomitantly and 2 (11.1%) were malignant MA, a surprisingly high proportion of malignant cases. In the same study, the authors suggested a subclassification of these neoplasms into classic MA, malignant MA, and MA with concomitant malignant component, concluding that their biological behavior is not yet well defined 16.
As reported in the literature and with the experience of the case described here, it can be said that MA is a rare pathology that is within the differential diagnosis of upper urinary tract masses. Pre-surgical diagnosis is difficult and treatment in general does not differ from the standard for renal carcinoma or urothelial carcinoma of the upper urinary tract, depending on the case. The diagnostic suspicion of this type of tumor may increase in patients in whom endoscopic assessment with biopsy is possible along with magnetic resonance imaging to complement imaging studies. The surgical treatment implemented in the case reported here is based on the recommendations of the 2020 European Association of Urology Guidelines on Upper Urinary Tract Urothelial Carcinoma (staging: cT3N1M0) 17,18.
CONCLUSION
MA is a very rare benign tumor that affects the upper urinary tract and is difficult to differentiate from other tumors with imaging studies. Its diagnosis is usually made by histopathological study and immunohistochemistry, although some research suggests the usefulness of pre-surgical fine-needle aspiration cytology. Given the low prevalence of this disease, reports and case series are of great importance as they allow to broaden the knowledge on the subject and to propose possible pre-surgical diagnostic strategies that, in turn, allow to implement treatment strategies that preserve the renal unit involved.