Introduction
Sarcoidosis is a multisystemic granulomatous disease of uncertain etiology. It is a diagnostic and treatment challenge since, even today, none of the tests generally used for its clinical management can be considered tests of choice and its course is, in many cases, unpredictable 1-4.
Its incidence and prevalence are not precisely known. In our country, the mean annual incidence rate is 1.37 per 100,000 inhabitants, although recent studies seem to raise that figure 5,6.
The mean age at diagnosis is 40 years, with a slight predominance of the female sex. It is a complex disease, highly suspected to have a genetic predisposition 1,5,7.
At a clinical level, it has miscellaneous symptoms, with pulmonary manifestations being the most common, although extrapulmonary involvement may also be found 7,8.
The cells that make up the sarcoid granuloma capture 18F-fluorodeoxyglucose (18F-FDG) and may be quantified. The algorithm most often used is the Standardized Uptake Value (SUV). These SUV variations allow hidden disease or disease located in uncommon locations to be detected, aiding diagnosis and the taking of biopsies 9. Furthermore, they make it possible to monitor the disease's activity and the response to treatment more accurately than clinical signs and symptoms, improving the performance of the usual monitoring tests 10. Thus, despite its lower availability and higher cost, the high sensitivity (90-100%) of 18F-FDG together with computed tomography (18F-FDG PET/CT) is making it an essential tool for managing patients with sarcoidosis 11-13.
The objective of this study was to evaluate the benefit of using 18F-FDG PET/CT (due to its ability to identify inflam matory activity in the sarcoid granuloma) for restratification and treatment adjustment compared with the signs and symptoms and tests commonly used for following patients with sarcoidosis.
Materials and method
An observational, analytical study was conducted of a retrospective cohort of patients treated for sarcoidosis in the internal medicine department of Hospital General Universitario de Ciudad Real (HGUCR), a referral hospital for a health catchment area of 191,468 patients.
The source of information was the patients' electronic medical records.
All patients diagnosed with sarcoidosis who were over the age of 18, had at least one 18F-FDG PET/CT during their clinical follow up and signed informed consent were included. The analyzed study period went from the first available electronic medical records at this center (in 2010) through May 2021. Patients who were not followed by internal medicine and those whose medical records did not include the minimum data needed for analysis were excluded.
The diagnosis of sarcoidosis was based on compatible anatomic pathology results (sarcoid granuloma) and/or compatible signs and symptoms. All cases were tested for tuberculosis and cancer. All patients were followed by the same internal medicine specialist (BD).
Sociodemographic data (age, sex, race, usual residence, occupation and work exposure, smoking and family history of autoimmune disease) and clinical signs and symptoms (asthenia, dyspnea and pain) were analyzed.
Analytical data were also evaluated (with angiotensin converting enzyme [ACE] levels), along with pulmonary function tests (PFTs), considering the percentages of forced vital capacity (FVC) and the diffusing capacity of the lungs for carbon monoxide (DLCO), as well as radiological study results (the Scadding Staging System [SSS] stages 14 using simple x-rays and CT) and nuclear medicine (18F-FDG PET/CT). Dermatological, ophthalmological and/or echocardiographic assessments were not routinely performed except in cases with symptoms suggestive of skin, eye or heart involvement.
All clinic visits in which an 18F-FDG PET/CT was per formed, and the reason for ordering it, were reviewed. The positive 18F-FDG PET/CT findings (with a pulmonary SUV range from 1.7 to 15.8) were analyzed, along with their location. Furthermore, the results were compared with the rest of the tests (described above) ordered simultaneously or most recently, whether during diagnosis or follow up. For staging, equivalence was established with the pulmo nary radiology studies.
Additionally, the treatments used for managing the disease were collected, as well as their modifications and the reasons for the modifications. The changes compared with the beginning of treatment (increases or additions) were motivated by 18F-FDG PET/CT uptake or in light of symptoms and increased SUV levels compared with prior 18F-FDG PET/CTs (when possible). On the other hand, the lack of uptake or decreased SUV levels justified dose reduction or discontinuation.
The variables were shown using the descriptive statistics appropriate for the nature of each variable: measures of cen tral tendency (mean) and dispersion (standard deviation) for quantitative variables and absolute and relative frequencies (percentages) for qualitative variables.
Results
Thirty-one patients (18 men and 13 women) aged 54.6±14.71 years, who had had sarcoidosis for 11±5.75 years, were evaluated. The rest of the main sociodemographic characteristics collected are shown in Table 1.
Table 1 Percentages of the study patients' main sociodemographic variables.
| Variables | N (%), Except prevalence (%c) | |
|---|---|---|
| Individuals | 31 | |
| Sex | ||
| Male | 18 (58.1) | |
| Female | 13 (41.9) | |
| Mean age/years since onset | 54.6±14.71/11±5.75 | |
| Autoimmune history* | ||
| Yes | 2 (6.45) | |
| No | 25 (80.65) | |
| NA/DK** | 4 (12.9) | |
| Race | ||
| Caucasian | 29 (93.5) | |
| Other | 2 (6.5) | |
| Chemical exposure | ||
| Yes | 19 (61.29) | |
| No | 9 (29.03) | |
| NA/DK | 3 (9.68) | |
| Prior infection | ||
| Yes | 5 (16.13) | |
| No | 21 (67.74) | |
| NA/DK | 5 (16.13) | |
| Smoking | ||
| Yes | 12 (38.71) | |
| No | 16 (51.61) | |
| NA/DK | 3 (9.68) | |
| Sarcoidosis prevalence | 16.19 | |
| *AI: autoimmune.** NA/DK (no answer/does not know). |
Twenty-eight diagnostic biopsies were found, 17 taken from enlarged mediastinal lymph nodes. In 20 patients (64.5%), the histological study was confirmatory. In the remaining patients, the diagnosis was based on clinical cri teria. None of the patients were diagnosed with cancer and those with a positive tuberculosis screening had received treatment.
The 52 clinical reviews in which an 18F-FDG PET/CT was performed were analyzed. The reasons for ordering the test were to help in diagnosis and to evaluate inflammatory activ ity in patients with reiterated symptomatic complaints and standard tests which were inconclusive for active disease. An average of 1.67 18F-FDG PET/CTs were ordered per patient. The three most common symptoms were asthenia (78.8% of the cases), pain (46.2%) and dyspnea (38.5%). Besides the 18F-FDG PET/CTs, 41 PFTs were performed. Altogether, 17.3% had decreased DLCO and 23% had reduced FVC. The concentration of ACE was measured on 48 occasions and was elevated in 53%. Five cases had a high-resolution CT (HRCT).
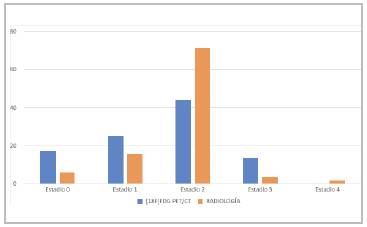
A total of 84.6% of the reviews in which an 18F-FDG PET/CT was performed showed positive uptake. These results were compared with those found in previous radio logical tests. The stages (by equivalence with radiological tests) were: Stage II (SII) in 44%, SI in 25%, S0 in 17.3% and SIII in 13.5%. All patients staged as SIV by radiol ogy had 18F-FDG PET/CT activity. 42.3% of the time, the objectified finding allowed patient restaging compared to the result obtained by other radiological studies. In 15 of these follow ups, the 18F-FDG PET/CT staging was lower than what was established by the other radiological tests reported (Figure 1).
In 44 of the 52 clinical follow ups, the patients were on some type of pharmacological treatment for the disease. The most commonly used drugs for monotherapy were corticosteroids (44.2%) and the most frequent combined therapy joined corticosteroids with methotrexate (13.5%). The 18F-FDG PET/CT result justified the choice of treatment in 37 of the 52 reviews. The treatment modifications are presented in Figure 2.
Discussion
Recent papers have objectivized the high sensitivity of 18F-FDG PET/CT for identifying inflammation, with positive uptakes in most of the analyzed reviews and mostly coincid ing with radiological Stages II and III 11,15. The results in our series of patients (greater than 80% uptake) appear to confirm this data. In this vein, and similar to Mostard et al. 's results, in our sample, all patients classified as SIV by radi ology had 18F-FDG PET/CT activity and, therefore, would benefit from a treatment adjustment 15. This supports the fact that 18F-FDG PET/CT can identify active disease in patients with sarcoidosis who have persistent symptoms but little variation in the usual laboratory, imaging and respira tory tests used 16.
The most notable results in this study regarding the role of 18F-FDG PET/CT were related to disease monitoring and treatment adjustments. 18F-FDG PET/CT provided greater accuracy and objectivity in determining activity, changes in activity and disease extension, compared with other tests routinely ordered in our hospital for periodic follow up of these patients 15,17. This had significant therapeutic implications, which have already been described by other authors, like Sobic-Saranovic et al., whose positive 18F-FDG PET/CT results justified treatment changes in more than 70% of the reviews, with treatment onset in a significant number of cases (highlighting patients with radiological SIV and a positive PET) 17.
The main weakness of this study is that the data was ob tained retrospectively from medical records in which some results and tests (like HRCT) were not available in all cases. On the other hand, the positive points to be noted are the adequate sample size, compared with other similar studies 11 and that the ordering of diagnostic tests and treatment decisions in all patients were done by a single professional (BD), which provides homogeneity in the results.
We coincide with Treglia et al. in the need to delve deeper in cost-benefit studies 18. Therefore, it would be relevant to evaluate the performance of 18F-FDG PET/ CT in monitoring sarcoidosis, trying to determine the most cost-efficient test or adapted algorithm, not only to improve the quality of care, but also to optimize the available resources. In this regard, 18F-FDG PET/CT could play a key role, as it is able to determine the degree of disease activity more accurately.
Conclusions
18F-FDG PET/CT provides additional benefits in sarcoidosis staging and therapeutic management, compared to signs and symptoms and other clinical tests commonly employed in follow-up, due to its greater accuracy in determining the disease's activity and extension.
Further study is needed of the markers which could pro vide greater specificity to this test and/or treatment regimens that would optimize the available resources and provide an excellent approach to patients with sarcoidosis. This opens the door to continue new studies.











 text in
text in