Introduction
Chicken eggs are the least expensive source of high quality animal protein and all essential nutrients. However, their consumption may increase blood cholesterol and the risk of developing coronary heart diseases (Burke et al., 2007). Therefore, research efforts have been directed to provide a low-cholesterol egg (Weiss et al., 1967; Elkin and Rogler, 1990; Pesti and Bakalli, 1998). Because genetic selection of chickens for this goal has been of little success, efforts have been centered on dietary manipulation (Hargis, 1988; Rahardja et al., 2010; Abdouli et al., 2014a). Fenugreek seeds have been among the several ingredients attempted because they have many therapeutic effects like hypoglycemic, hypocholesterolemic, anti-inflammatory, antipyretic, antimicrobial and anthelmintic properties (Bash et al., 2003). On this subject, almost all reviewed studies (Moustafa, 2006; Safaa, 2007; Nasra et al., 2010) used fenugreek seeds at dietary inclusion levels not exceeding 2%. In these reports, egg yolk cholesterol content was decreased by 7% at most (Nasra et al., 2010). When fenugreek seed was given at 6 g/hen/d (which corresponds to an incorporation level in feed of 5.7%), there was, on one hand, a decrease in hens’ serum total cholesterol level but not in that of egg yolk and, on the other hand, a trend for an increase of feed conversion ratio (Abdouli et al., 2014a). This finding suggested that fenugreek seed contains anti-nutritional factors, which may have an adverse effect when ingested by non-ruminant animals.
Indeed, as for others leguminous plants, dormant fenugreek seed has been reported to contain anti- nutritional factors such as trypsin and chymotrypsin inhibitors and tanins (Billaud and Adrian, 2001). Several studies have shown that during seed germination anti- nutritional factors such as protease inhibitors, phytic acid and tannins decreased significantly (Malleshi and Desikachar, 1986; Udayasekhara Rao and Deosthale, 1988; Osuntogun et al., 1989). Moreover, during a previous in vitro study in our laboratory, Mabrouki et al. (2015) demonstrated that fenugreek seed pre- germination or autoclaving significantly increased fat, crude protein and acid detergent fiber contents and reduced phenols, tannins and flavonoids levels. The authors also found that these processes enhanced protein digestibility index to 45.39% and digestible energy by 3.8% and metabolizable energy by 4.3 to 6.7%. Therefore, autoclaved and pre-germinated fenugreek seeds need to be further evaluated in in vivo studies.
The main objective of the present study was to evaluate the effect of dietary addition of unprocessed (UPFS), autoclaved (AFS), or pre-germinated (PGFS) fenugreek seeds on laying hens performance and egg physical characteristics and chemical composition, particularly its triglycerides and cholesterol contents.
Material and methods
Ethical considerations
The experimental protocol was approved by the Official Animal Care and Use Committee of the Higher School of Agriculture of Mateur (protocol N° 05/15) before the initiation of research and followed the Tunisian guidelines approved by the committee on care, handling, and sampling of the animals.
Pre-germinated and autoclaved fenugreek seeds preparation
Fenugreek seeds of bitter, non-identified genotype were cleaned manually to remove foreign matter and damaged seeds. Autoclaving and pre-germination were carried out as reported by Mabrouki et al. (2015). For pre-germinated fenugreek seeds, 8 Kg of seeds were initially mixed with 16 L of distilled water and divided into 3 plastic containers. In each container, fenugreek seeds were spread in a layer of about 10 mm between two layers of sheet paper and covered with a black plastic sheet to ensure darkness. During this process, which lasted for 21 h, distilled water was used to wet (sprinkle) the upper paper sheet so that the overall seeds:water ratio was 1:2.5. When approximately 95% of the seeds germinated (at the emergence of the radical), the pre- germination was stopped first by cold air ventilation with fans for 48 h and then by drying at 40 °C in an air forced oven for 20 h. For autoclaved fenugreek seeds, 8 Kg of fenugreek seeds were autoclaved at 121 °C for 15 min, then dried by cold air ventilation with fans for 26.5 h.
Experimental diets preparation
A conventional mash diet (control) for laying hens based on corn and soybean-meal was prepared. Thereafter, 3 diets were prepared: unprocessed fenugreek seeds (UPFS) supplemented diet, autoclaved fenugreek seeds (AFS) supplemented diet and pre- germinated fenugreek seeds (PGFS) supplemented diet. These were individually prepared by mixing the conventional feed thoroughly with the corresponding ground fenugreek seed type at the incorporation level of 5.7% as shown in Table 1.
Animals and experimental design
Forty-eight 31 week-old Novogen white laying hens (initial live weight 1,420 g ± 91 g) were divided into 4 homogeneous groups of 12 hens each. Each group was randomly allocated to one of the 4 following dietary treatments: (1) control diet, (2) unprocessed fenugreek seeds (UPFS) supplemented diet, (3) autoclaved fenugreek seeds (AFS) supplemented diet, and (4) pre-germinated fenugreek seeds (PGFS) supplemented diet.
In all groups, hens were fed their assigned diet (100 g/hen/d) from the beginning (d 1) to the end of the experiment (d 63). Diets were restricted to 100 g/hen/d to ensure that hens consumed almost all of the distributed feed and to reduce the feed-selection behavior typically observed in laying hens. Hens were housed in individual cages with individual feed-trough and common water-trough in a room with ambient temperature of about 20 °C and a photoperiod of 16 h light: 8 h darkness cycle. Water was provided ad libitum throughout all the trial period.
Measurements and sampling
All birds were weighed individually at the beginning and at the end of the experiment to determine the live weight changes. Feed was offered once daily at 7:30 a.m. and refusal was measured weekly. Egg production and weight were recorded daily. Daily feed consumption, laying rate (number of laid eggs x 100/number of feeding days), and feed conversion ratio (feed consumption/number of eggs x egg weight) were calculated per week. Eggs laid during 5 days of the 1st, 5th, and 9th weeks were used for egg physical characteristics measurements (egg albumen, yolk, and shell weights), as well as triglycerides and cholesterol determination. Eggs laid during days 59, 62, and 63 of the experimental period were pooled per hen and used for analyses of yolk: total lipids and color.
Table 1 Ingredients and chemical composition of experimental diets.

£Cell content = 100 - NDF.
¥Metabolizable energy = 2707.71 + 58.63 EE − 16.06 NDF (Nascimento et al., 2007).
Eggs laid on day 60 and 61 of the experiment were hard-boiled for 20 min and used for egg crude protein content determination.
Chemical analyses
Dry matter of diets (DM) was determined at 105 °C for 24 h, while all other analyses were done on samples dried at 65 °C and ground in a mill to pass through a 0.5 mm screen.
Ash content was determined by igniting the ground sample at 550 °C in a muffle furnace for 10 h. The Association of Official Analytical Chemists method (AOAC, 1984) was used for crude protein (CP) determination. Neutral detergent fiber (NDF) was determined as described by (Van Soest et al., 1991), but without using sodium sulphite and alpha amylase. Cell content was calculated using the following formula:
Cell content = 100 - NDF (% DM).
Egg yolk samples pooled per hen were solubilized in 2% (w/v) NaCl solution (Pasin et al., 1998) and used for cholesterol and triglycerides determination using standard enzymatic-colorimetric methods (cholesterol enzymatic colorimetric test, CHOD-PAP and triglycerides enzymatic colorimetric (GPO-PAP, Biomaghreb, Ibn Khaldoun, Ariana,Tunisia). Total lipids of homogenized egg yolk were extracted with isopropanol: hexane (30:70, v/v) mix and then gravimetrically estimated. Eggs collected on days 60 and 61 were boiled for 20 min, blended and used for total egg crude protein determination by the Kjeldhal method (AOAC, 1984). The egg yolk color was evaluated visually by means of the Yolk Color Fan® Scale DSM Yolk Color Fan (DSM Nutritional Products Europe, Wurmisweg 576, CH-4303 Kaiseraugst, Switzerland) where color intensity is gradually scored (1 for light yellow and 15 for orange).
Statistical analysis
Data of repeated measurements (feed refusal and intake, laying rate, egg mass, feed conversion ratio, egg physical characteristics, yolk triglycerides, and total cholesterol) were tested for diet, week on diet effects and their interaction using mixed models with compound symmetry covariance structures. Differences between means for diets averaged over weeks were tested using the ESTIMATE statement in SAS (Littell et al., 1998). When week and/or week-diet interaction effects were significant at α = 0.05, differences between means for each week and the average of means for subsequent weeks within each diet were tested using the ESTIMATE statement (Littell et al., 1998). Data of non-repeated measurements (hens’ live weight change, whole egg crude proteins, yolk lipids and color) were tested for diet effect using the General Linear Model (GLM) procedure according to the following model:
Y ij = µ + T i + e ij
Where:
Y ij = dependent variable measured in animal j that was subjected to i treatment.
µ = general mean.
T i = fixed effect of i treatment.
e ij = random error.
All statistical procedures were performed using the SAS package (Statistical Analysis System, version 6, SAS Institute, Cary, NC, USA; 1989).
Laying performance
Table 2 summarizes hens’ live weight changes, daily refusal, feed consumption, laying rate, egg mass and feed conversion ratio (FCR, g feed/g egg mass). Although feed was restricted to 100 g/hen/day, feed refusal and consequently feed consumption were both affected (p<0.05) by diet, week on diet and their interaction.
Only feed refusal and consumption of UPFS fed group changed (p<0.05) over week on diet (Table 3). The UPFS-diet was associated with the lowest feed consumption during week 1 averaging 83.52 g DM and increased gradually thereafter so that the means of estimate differences between week 3 and the subsequent weeks became insignificant (p>0.05).
Table 2 Hens’ live weight changes and laying performances according to fed diets during the whole experimental period.

Control = diet with 0% fenugreek seeds. UPFS = control diet supplemented with unprocessed fenugreek seeds at 5.7%. AFS = control diet supplemented with autoclaved fenugreek seeds at 5.7%. PGFS = control diet supplemented with pre-germinated fenugreek seeds at 5.7%. SEM = standard error of the mean. D = diet. W = week. D-W = diet - week interaction. *** = p<0.0001; ** = p<0.001; * = p<0.05; NS = p>0.05.
Means within the same row with different superscript letters ( a, b ) are significantly different (p<0.05).
All hens showed slight loss of body weight at the end of the experimental period, but this weight loss was not affected (p>0.05) by dietary treatment.
In parallel, laying rate was not affected (p>0.05) by week on diet and was reduced (p<0.05) only in hens receiving the UPFS-diet from 92.52 to 84.52%. Consequently, daily egg mass production was the lowest for the UPFS-diet (p<0.05). Week on diet had a significant effect (p<0.05) on egg mass production.
For each dietary treatment, comparison of differences of daily egg mass production between each of week 1, 2 and 3 from their subsequent weeks (Table 3) showed that there was a week effect (p<0.05) only for UPFS-diet and that this effect became insignificant (p>0.05) beginning from week 3. Feed conversion ratio was not affected (p>0.05) by the dietary treatment and by week on diet. However, UPFS-diet was only associated with a FCR slightly higher (p>0.05) than that of the control.
Egg physical characteristics
The physical characteristics of eggs (egg, yolk, albumen and shell weights) are shown in Table 4.
Table 3 Duration effect of the diet´s distribution on feed intake and egg’s weight.

Control = diet with 0% fenugreek seeds. UPFS = control diet supplemented with unprocessed fenugreek seeds at 5.7%. AFS = control diet supplemented with autoclaved fenugreek seeds at 5.7%. PGFS = control diet supplemented with pre-germinated fenugreek seeds at 5.7%. p-values within the same row:
*** = p<0.0001; * = p<0.05; NS = p>0.05. Positive values indicate an increase over time.
Table 4 Physical characteristics of eggs according to fed diets.

Control = diet with 0% fenugreek seeds. UPFS = control diet supplemented with unprocessed fenugreek seeds at 5.7%. AFS = control diet supplemented with autoclaved fenugreek seeds at 5.7%. PGFS = control diet supplemented with pre-germinated fenugreek seeds at 5.7%. SEM = standard error of the mean. D = diet. W = week. D-W = diet-week interaction. *** = p<0.0001; * = p<0.05; NS= p>0.05. £Yolk weight = yolk without vitelline membrane.
Table 5 Duration effect of the diet´s distribution on eggs’ physical characteristics.
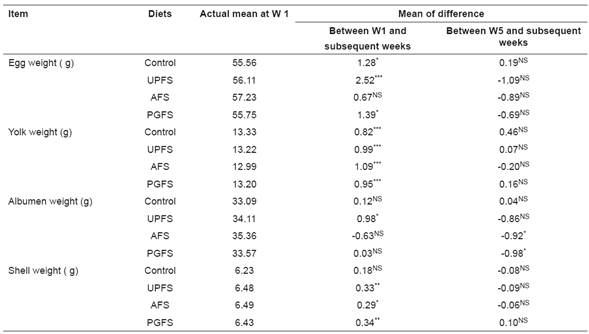
Control = diet with 0% fenugreek seeds. UPFS = control diet supplemented with unprocessed fenugreek seeds at 5.7%. AFS = control diet supplemented with autoclaved fenugreek seeds at 5.7%. PGFS = control diet supplemented with pre-germinated fenugreek seeds at 5.7%. *** = p<0.0001; ** = p<0.001; *
= p<0.05; NS = p>0.05.
All these characteristics were affected (p<0.05) only by week on diet. Therefore, for each diet, differences between weights at week 1 and their average weights at weeks 5 and 9, as well as differences between weights at weeks 5 and 9 were compared (Table 5). Tested differences of egg weight were not significant (p>0.005) for AFS- diet indicating no week effect. The other dietary treatments (control diet, UPFS-diet and PGFS-diet) were associated with a lower egg weight at week1 (p<0.05). For all diets, yolk weights were lower (p<0.05) at week 1 than at subsequent weeks. Albumen weight did not change (p<0.05) over time for the control, and increased (p<0.05) at week 5 for UPFS-diet and decreased (p<0.05) for AFS-diet and PEGF-diet at week 9.
Egg chemical composition and yolk color
Table 6 presents egg’s chemical composition and yolk color data. Crude protein measured on whole hard-boiled eggs and yolk lipids and color measured on fresh eggs collected on the last week of the experiment were not affected by dietary treatments (p>0.05). Yolk triglycerides and cholesterol contents were measured on eggs laid during 5 days of the 1st, 5th, and 9th week of the trial and pooled by hen at each week. Triglycerides concentrations (mg/g yolk) were not affected (p>0.05) by diet and week, yet they were affected (p<0.05) by diet- week interaction. Therefore, differences between each of week 1 and week 5 from average of their subsequent weeks were tested within each diet (Table 7).
It was found that while triglycerides concentrations did not change over time for diets containing AFS and PGFS, they increased in the control and decreased in UPFS-diet at the last week of the trial (week 9). When yolk weights were taken in account, yolk triglycerides contents (g/egg) did not change over time for AFS- diet and PGFS-diet and increased in the control and in UPFS-diet at week 9 and week 5, respectively. Yolk cholesterol concentrations (mg/g yolk) and contents (mg/egg) were not affected (p>0.05) by diet, week, and diet-week interaction.
Table 6 Egg crude proteins, lipid and cholesterol contents, yolk triglycerides and color.

Control = diet with 0% fenugreek seeds. UPFS = control diet supplemented with unprocessed fenugreek seeds at 5.7%. AFS = control diet supplemented with autoclaved fenugreek seeds at 5.7%. PGFS = control diet supplemented with pre-germinated fenugreek seeds at 5.7%. SEM = standard error of the mean. D = diet. W = week; D-W = diet-week interaction. *** = p<0.0001; * = p<0.05; NS = p>0.05.
δColor score range from 1(i.e. yellow) to 15 (i.e. orange).
Means within the same row with different superscript letters ( a, b ) are significantly different (p<0.05).
Table 7 Duration effect of the diet´s distribution on egg yolk triglycerides content.

Control diet supplemented with autoclaved fenugreek seeds at 5.7%. PGFS = control diet supplemented with pre-germinated fenugreek seeds at 5.7%.
*** = p<0.0001; ** = p<0.001; * = p<0.05; NS = p>0.05. Positive values indicate an increase over time. *** = p<0.0001; * = p<0.05; NS = p>0.05.
Discussion
Although feed was restricted to 100 g/hen/day, UPFS diet was associated with the highest feed refusal during week 1 averaging 4.83 g DM and decreased gradually thereafter so that the means of estimate differences between week 3 and the subsequent weeks became insignificant. This low feed consumption and gradual adaptation during the first two weeks observed only for UPFS-diet could be attributed to any unknown secondary metabolite sensitive to autoclaving and pre-germination.
A previous research (Mabrouki et al., 2015) on the same fenugreek seeds batches used in the present study showed that compared with UPFS, AFS had lower flavonoids content and PGFS had lower total phenols, tannins and flavonoids contents. Accordingly, it was suggested that flavonoids might have produced flavors and odors that were not acceptable by the birds until they gradually got adapted.
Such suggestion was not, however, in agreement with the usual use of flavonoids as feed additives in poultry for their antioxidative properties (Hager- Theodorides et al., 2014).
Furthermore, Robert et al. (2013) reported that two plant extracts rich in flavonoids increased feed intake of laying hens subjected to heat stress (35 °C for 4 days). If the first 2 weeks were considered as an adaptation period and, therefore, not taken in account, feed refusals would become very low and feed consumptions became similar for the 4 treatments throughout the trial period. In such case, our finding would agree with the reported lack of effect on feed intake of fenugreek seeds at an incorporation level of 2 to 6 g/day/hen (Abdouli et al., 2014a).
Regardless of their dietary treatment, all hens showed slight loss of body weight at the end of the experimental period, which was most likely due to the restriction on feed distribution. In parallel, laying rate was reduced only for the UPFS diet from 92.52 to 84.52%. Consequently, daily egg mass production was the lowest for the UPFS-diet (p<0.05). The results herein did not agree with reported increased egg production rate and egg mass related to fenugreek seeds dietary supplementation at 0.01 to 0.5% level (Moustafa, 2006; Awadein et al., 2010). Furthermore, FCR of hens fed the UPFS diet tended to increase. Thus, UPFS could have, if any, only a slight adverse effect on dietary nutrients digestion and metabolism even though, a previous in vitro evaluation of the same batches of fenugreek seeds showed the presence of some anti-nutritional factors (total phenols and tannins) at equal levels in UPFS and AFS and at much lower levels in PGFS (Mabrouki et al., 2015). In addition, calculated metabolizable energy content, using the general equation for energy and protein feedstuffs of Nascimento et al. (2009), was equal to 2,650 Kcal/Kg DM of UPFS and was higher in AFS (2,760 Kcal/Kg DM) and in PGFS (2,831 Kcal/Kg DM). To the best knowledge of the authors, adverse effect of UPFS on hens’ laying performance had not been reported in the literature. However, almost all reviewed studies used fenugreek seeds at an incorporation level not exceeding 2.5% of the diet. When ground fenugreek seeds were used at the rate of 6 g/hen/d, there was a trend of reduction in laying rate (83.67 vs. 79.39%) and egg mass production (51.85 vs. 48.63 g/hen/day) and a trend of increase in FCR from 1.93 to 2.17 (Abdouli et al., 2014a).
Our data showed that egg characteristics were not affected by dietary treatment and that observed changes over time were not significant and, therefore, of little physiological significance.
To the best of our knowledge, no information related to the effect of processed fenugreek seeds on egg physical characteristics were found in the literature.
As far as UPFS-diet was concerned, results reported herein are consistent with those reported by Abdalla et al. (2011) and Abdouli et al. (2014a). However, they are not in agreement with those of other studies (El-Kaiaty et al., 2002; Abaza, 2007; Nasra et al., 2010), wherein authors found a decrease of egg yolk percentage, an increase of yolk and albumen weights, and an increase of shell thickness and albumen weight, respectively.
Whole egg crude protein varied from 11.50 to 12.10% corresponding to 5.18 and 5.83 g/egg, respectively. These values were slightly lower than those (14.7 to 15%) reported for blue, brown-yellowish and white eggs from hens (Isa Brown, Lohman, Araucana ) on free range or on conventional diet (Gultermirian et al., 2009). Yolk total lipids content varied from 30.96 to 31.68% corresponding to a range of 14.60-15.18 g/egg. These values were higher than those found in Isa Brown eggs (19.8%) and Lohman eggs (27.8%) and lower than that found in Araucana eggs (35%; Gultermirian et al., 2009). For yolk triglycerides and cholesterol contents, changes over time and differences between dietary treatments were not significant and could be without physiological significance. Our results are in accordance with reported lack of effect of ground fenugreek seeds, and of water and hexane insoluble fraction of whole fenugreek seeds supplementation at 2 to 6 and 1.75 g/hen/day, respectively (Abdouli et al., 2014a,c). In contrast, other studies reported reduced egg yolk cholesterol due to fenugreek seeds supplementation at 2% (Safaa, 2007), 0.1 or 0.5% (Nasra et al., 2010) and 0.05, 0.1 or 0.15% (Moustafa, 2006).
In the studies cited above, egg yolk cholesterol reduction attributed to fenugreek seeds supplementation was of small magnitude, not exceeding 7%, and the mechanisms underlying such effect has not been reported to date.
Reduction of cholesterol absorption and synthesis may both be mediated by 4-hydroxylisoleucine, saponins and other bioactive phytonutrients as these compounds have been found to reduce serum cholesterol level of dyslipidemic hamster (Narender et al., 2006), streptozotocin diabetic rats (Petit et al., 1995), Wistar rats (Olfa et al., 2010) and also that of laying hens (Abdallal et al., 2011; Abdouli et al., 2014a).
It is worth noting, however, that reduction of serum cholesterol concentration may not be always associated with a reduction of egg cholesterol (Abdouli et al., 2014a). The effect of fenugreek seeds supplementation on egg yolk cholesterol may be influenced by many factors like the genotypes of fenugreek seeds, the hens’ strain and age, the ingredients composition of the basal diet, and the methods of cholesterol extraction and determination.
In the present study, egg yolk color score estimated by the DSM Yolk Color Fan (DSM Nutritional Products Europe, Wurmisweg 576, CH-4303 Kaiseraugst, Switzerland) was negatively affected by dietary addition of UPFS. This method, although subjective, is the most common assay used to evaluate egg yolk color. The observed lower score associated with UPFS-diet administration could not be explained. Previous evaluation of the same batches of fenugreek seeds showed that β-carotene and lycopene contents were the highest in AFS, intermediate in UPFS and the lowest in PGFS (Mabrouki et al., 2015), suggesting that the observed low score might not be related to these carotenoids in UPFS-diet. On this subject, it was reported that unprocessed fenugreek seeds supplementation did not affect egg yolk color score (Abdouli et al., 2014b).
It was concluded that the incorporation of unprocessed fenugreek seeds in laying hens’ diet at a rate of 5.7% reduced feed consumption until hens got adapted within 2 weeks and decreased over all laying rate. However, it did not affect feed conversion ratio. Furthermore, this dietary incorporation had no effect on egg physical characteristics nor chemical composition, namely on triglycerides and total cholesterol contents. Both autoclaved and pre- germinated fenugreek seeds were without adverse effects on feed consumption but were inefficient for egg cholesterol reduction.
Further investigations are needed to evaluate the impact of fenugreek seeds on organoleptic characteristics, nutritional value, fatty acids profile and lipid peroxidation status of polyunsaturated fatty acids enriched egg.
Acknowledgments
The authors wish to thank Dr. Bruce Seal and Dr. Brain Oakley for reviewing the English language of the whole manuscript and Dr. Caroline VANDERGHEM for reviewing the Spanish and Portuguese language of the “resumen” and the “resumo”, respectively. We are also greatful with Mr. Lassad TAYACHI, for his precious technical assistance.














