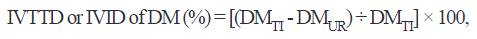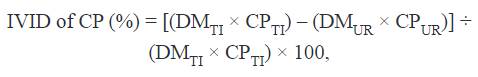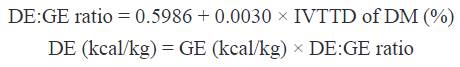Introduction
Soybean milk has become popular as a protein supplement for humans. After squeezing the soybean slurry, raw soybean milk is produced. The residue is called soybean milk by-product (SMBP; Toda et al., 2007; Kim et al., 2016). Soybean milk by-products are used in human and animal diets, but the majority is disposed (Woo et al., 2001). As SMBP often contains more than 30% of crude protein (CP), this ingredient can be used as an alternative protein source for animals (Woo et al., 2001; Li et al., 2012).
An accurate nutritional evaluation of feed ingredients is required for a precise feed formulation (Son et al., 2017). However, information on available energy and nutrients in SMBP as a swine feed ingredient is very limited (Kortelainen et al., 2014). In vitro methods have been widely used to estimate nutritional values because these assays are highly correlated with in vivo data (Boisen and Fernández, 1997; Son et al., 2017). Therefore, the objective of this study was to determine energy concentrations and protein utilization of SMBP using in vitro assays simulating digestion and absorption by pigs.
Materials and methods
Sample preparation
Four soybean milk by-product samples were obtained from three soybean milk-producing facilities in the Republic of Korea. Samples 2 and 3 were from the same facility, and sample 3 was dried at the soybean milk-producing facility. Samples 1, 2, and 4 were dried to a constant weight at 55 °C using a forced-air oven (FC- PO-150, Dongseo Science LTD., Seongnam, Republic of Korea). Dried test ingredients were finely ground to pass a 1-mm screen (Cyclotech 1093; Foss Tecator AB, Höganäs, Sweden).
In vitro assays
In vitro total tract disappearance (IVTTD) of dry matter (DM) in ground SMBP was determined using a three-step procedure (Boisen and Fernández, 1997). In the first step, 0.5 g of sample was weighed into a 125 mL conical flask, and 25 mL of phosphate buffer solution (0.1 M and pH 6.0), and 10 mL of 0.2 M HCl were added to the flask. The pH of the solution was adjusted to 2.0 using 1 M HCl and NaOH solution, and 1 mL of freshly prepared pepsin (25 mg/mL; ≥ 250 U/mg solid, P7000, Pepsin from porcine gastric mucosa, Sigma- Aldrich, St. Louis, MO, USA) was added. Thereafter, 0.5 mL of chloramphenicol (C0378, Chloramphenicol, Sigma-Aldrich, St. Louis, MO, USA; 0.5 g/100 mL of ethanol) was added to prevent bacterial fermentation. The flasks were incubated in a shaking incubator for 2 h at 39 °C.
In the second step, the flasks were added with 10 mL of phosphate buffer (0.2 M and pH 6.8), and 5 mL of 0.6 M NaOH solution. Then the pH was adjusted to 6.8 using 1 M HCl and NaOH solution, and 1 mL of freshly prepared pancreatin solution (100 mg/mL; 4 × USP, P1750, pancreatin from porcine pancreas, Sigma-Aldrich, St. Louis, MO, USA) was added. The flasks were incubated in a shaking incubator for 4 h at 39 °C.
After the second incubation, 10 mL of 0.2 M EDTA solution was added, and the pH was adjusted to 4.8 using 30% of acetic acid and 1 M NaOH solution. The flasks were added with 0.5 mL of Viscozyme (V2010, Viscozyme® L, Sigma-Aldrich, St. Louis, MO, USA), and incubated for 18 h at 39 °C.
After the incubation, undigested residues were filtered through pre-dried and weighed glass filter crucibles containing 0.4 g of celite using the Fibertec System (Fibertec System 1021 Cold Extractor, Tecator, Hӧganӓs, Sweden). The test flasks were rinsed twice by distilled water, followed by rinsing twice with 10 mL of 95% ethanol and 99.5% acetone. Then, glass filter crucibles with undigested residues were dried at 130 °C for 6 h. Glass filter crucibles were weighed after cooling for 1 h.
In vitro ileal disappearance (IVID) of DM in ground SMBP was determined using a two-step procedure (Boisen and Fernández, 1995). The first and second steps were similar to the procedures of IVTTD. For IVID, 1.0 g of sample was used, and the concentrations of pepsin and pancreatic solutions were reduced to 10 and 50 mg/mL, respectively, while the incubation times were increased to 6 and 18 h, respectively. After the incubation, the test flasks were added 5 mL of 20% sulfosalicylic acid solution and left at room temperature for 30 min to precipitate indigested protein. After 30 min of precipitation, undigested residues were filtered through pre-dried and weighed glass filter crucibles containing 0.5 g of celite using the Fibertec System (Fibertec System 1021 Cold Extractor, Tecator, Hӧganӓs, Sweden). The test flasks were rinsed twice by distilled water followed by rinsing twice with 10 mL of 95% ethanol and 99.5% acetone.
Then, glass filter crucibles with undigested residues were dried at 80 °C for 24 h. Glass filter crucibles were weighed after cooling for 1 h. After conducting the two-step procedure, undigested residues on filter crucibles were collected for analyzing CP concentration to calculate IVID of CP. The IVTTD and IVID for each ingredient were measured in triplicate.
Chemical analysis
Test ingredients were analyzed for DM (method 930.15; AOAC, 2005), CP (method 990.03; AOAC, 2005), ether extract (method 920.39; AOAC, 2005), calcium (method 978.02; AOAC, 2005), phosphorus (method 946.6; AOAC, 2005), and ash (method 942.05; AOAC, 2005). Test ingredients were also analyzed for neutral detergent fiber (NDF) and acid detergent fiber using an Ankom A2000 Fiber Analyzer (Ankom Technology, Macedon, NY, USA). Gross energy (GE) was analyzed in each ingredient using a bomb calorimeter (Parr 1261 bomb calorimeter; Parr Instruments Co., Moline, IL, USA). Amino acids in SMBP were analyzed using ion-exchange chromatography with postcolumn derivatization with ninhydrin (AOAC, 2005; method 994.12). Methionine and cysteine in test ingredients were determined by method 985.28 (AOAC, 2005).
Calculations and statistical analysis
The IVTTD or IVID of DM and IVID of CP were calculated with the following equations, respectively:
where: DMTI and DMUR are the weight of DM concentration in the test ingredient and undigested residues, respectively.
Where: CPTI and CPUR are CP concentration expressed as DM basis in the test ingredient and undigested residues, respectively. Based on determined IVTTD, digestible energy (DE):GE ratio and DE in SMBP were estimated with the following equations (Park et al., 2012):
Metabolizable energy (ME) was calculated using an equation as follows (Noblet and Perez, 1993):
Tryptophan concentration in SMBP was estimated using an average value of tryptophan to lysine ratio in soybean meal and full- fat soybean according to Goebel and Stein (2011) and NRC (2012). The average value of tryptophan to lysine ratio was 0.22.
The GLM procedure of SAS (SAS Inst. Inc., Cary, NC, USA) was used, and least squares means for response criteria were calculated for each ingredient. Each flask was considered as an experimental unit. Differences among least squares means were tested using the PDIFF option with Tukey’s adjustment (Seo et al., 2018).
Results
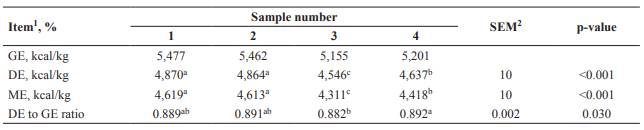
Crude protein and GE concentrations in SMBP ranged from 32.2 to 37.0% and 5,155 to 5,477 kcal/kg on an as-is basis (Table 1), respectively. The average ratio of methionine and threonine to lysine in SMBP was 0.22 and 0.73, respectively. The IVTTD of DM in sample 4 was greater than that of sample 3, whereas sample 4 had less IVID of DM compared with sample 1 (p<0.05; Table 2). Sample 2 showed greater IVID of CP than samples 1 or 3 (p<0.05). Metabolizable energy was not different between samples 1 and 2, and sample 3 had the least value (p<0.05) among SMBP samples (Table 3).
Table 1 Analyzed composition of soybean milk by-product, as-is basis.
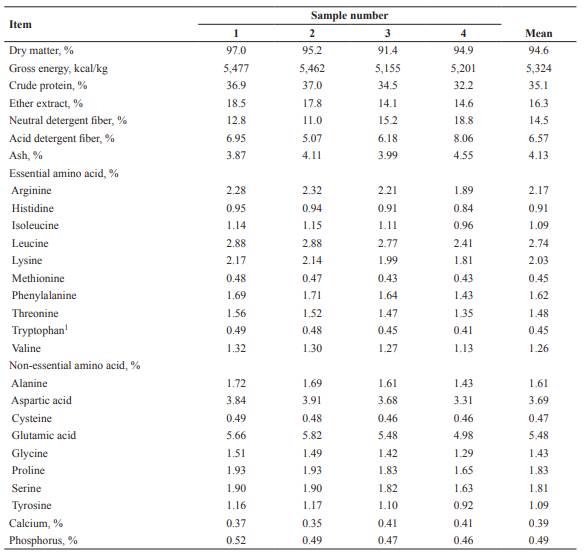
1Tryptophan was estimated using the average value of tryptophan to lysine ratio in soybean meal and full-fat soybean (Goebel and Stein, 2011; NRC, 2012).
Table 2 In vitro disappearance of soybean milk by-product1.
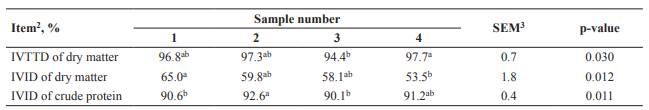
a-bLeast squares means within a row without a common superscript differ at p<0.05.
1Each least squares mean represents 3 observations except for IVID of crude protein in sample 4 (2 observations); 2IVTTD, in vitro total tract disappearance; IVID, in vitro ileal disappearance; 3SEM, standard error of the means.
Discussion
Soybean milk by-products produced from soybean milk-producing facilities have been regarded as potential substitutes for other protein sources in swine diets because of their high protein concentration (Li et al., 2012). However, very limited information is available on CP and essential amino acid digestibility of SMBP by pigs (Kortelainen et al., 2014), and energy values in SMBP for pigs, to our knowledge, have not been documented.
The analyzed composition of SMBP tested in the present work was within the range of reported values (Ma et al., 1997; Li et al., 2012; Kortelainen et al., 2014). The ratios of methionine and threonine to lysine of SMBP in the current study were very similar to the values in previous studies (Hermann and Honeyman, 2004; Kortelainen et al., 2014; Jiang et al., 2018).
In vitro total tract disappearance of DM represents the three-step digestion of the stomach, small intestine, and large intestine of pigs (Boisen and Fernández, 1997). In contrast to IVTTD of DM, IVID of DM and IVID of CP represent two-step digestion of the stomach and small intestine (Boisen and Fernández, 1995). The IVID of CP in SMBP in the present study was greater than the apparent ileal digestibility of CP (90.1 to 92.6 % vs. 81.5%) in SMBP by pigs determined by Kortelainen et al. (2014). This difference is likely due to endogenous losses of protein. Apparent ileal digestibility does not reflect endogenous losses of protein from the gastrointestinal tract (Park et al., 2013). In contrast to apparent values, the secretion of endogenous losses does not occur in the in vitro system, which is very similar to true digestibility (Boisen and Fernández, 1997). Another possible reason is the difference in trypsin inhibitor activity of SMBP. Trypsin inhibitor is a principal anti- nutritional factor in soybean and has been known to lower protein and DM digestibility due to a reduction in the activity of proteolytic enzymes (Goebel and Stein, 2011). In previous studies, in vitro disappearance of CP in raw legume grains ranged from 58 to 80%, and these values increased up to 88% when grains were adequately heat-treated (Khokhar and Chauhan, 1986; Nergiz and Gökgöz, 2007; Shimelis and Rakshit, 2007). Standardized ileal digestibility of essential amino acids reported by Kortelainen et al. (2014) and both IVTTD of DM and IVID of CP of SMBP in the present study exceeded 90%. Although trypsin inhibitor activity was not analyzed in both studies, negative influence of trypsin inhibitor in SMBP was not observed considering the high digestibility values.
Unlike IVTTD of DM or IVID of CP, the value of IVID of DM was relatively low (53.5 to 65.0%). The low values of IVID of DM in SMBP compared with IVTTD of DM are supported by previous studies. In the studies by Park et al. (2016) and Navarro et al. (2018), IVID of DM in soybean meal was less than IVTTD of DM by 20 and 15% unit, respectively. This difference may be due to the absence of simulation of fiber-degrading enzymes that are secreted by microbes in the large intestine in the IVID procedure. In contrast to the IVID procedure, Viscozyme (V2010, Viscozyme® L, Sigma-Aldrich, St. Louis, MO, USA) is added in the third step when determining IVTTD. As Viscozyme contains various carbohydrases, such as arabanase, cellulase, beta-glucanase, hemicellulose, and xylanase, most dietary fiber in SMBP is expected to be digested in the third step.
The reason for less IVID of DM in sample 4 compared with sample 1 may be due to greater NDF concentration in sample 4 (18.8 vs. 12.8%). However, no difference in IVID of CP between sample 1 and 4 was observed. Generally, a greater amount of undigested fiber in the small intestine could interfere with protein utilization, but dietary fiber may not always be a promising predictor of IVID of CP (Huang et al., 2003; Fang et al., 2007). In the study of Huang et al. (2003), NDF concentration (11.0 to 15.1%) was negatively correlated with IVID of DM in various barley cultivars (r=−0.97). In contrast, the correlation between NDF and IVID of CP was not significantly strong. Another possible reason is that variability in NDF concentrations (11.0 to 18.8%) of SMBP in the current study might not be sufficient to verify a consistent negative impact of fiber on protein utilization.
The equation employed for estimating DE:GE ratio of SMBP in the present study was developed by Park et al. (2012). In the study by Park et al. (2012), the prediction equation was developed by a regression analysis based on a high correlation (r2=0.91) between IVTTD of DM and DE:GE ratio of by-products in pigs. The reason for the least ME of sample 3 is attributed to low GE concentration and DE:GE ratio. The average value of estimated ME in SMBP (4,490 kcal/kg as-is basis) is greater than ME in soybean meal (approximately CP 47% and 3,283 kcal/kg as-is basis) reported by Sauvant et al. (2004) and NRC (2012). This difference could be attributed to a higher ether extract concentration in SMBP (14.1 to 18.5% as-is basis) compared with values in soybean meal (1.5%; Sauvant et al., 2004; NRC, 2012). As oil extraction does not occur during the production of soybean milk, SMBP contains high ether extract concentration.
Hermann and Honeyman (2004) fed a diet containing 25% SMBP replacing corn and soybean meal to nursery pigs with 13 kg initial body weight. This inclusion did not show compromised growth performance compared with pigs fed a corn-soybean meal-based diet. Considering the inclusion rate and energy and protein utilization of SMBP determined in the present study, SMBP could be used as a high- quality feed ingredient for swine. However, as potential difference in energy values and protein digestibility among SMBP samples were found in the present study, energy and nutrient utilization should be determined before using SMBP in swine diets. Furthermore, available energy and protein values in SMBP should be carefully applied to the industry. As stated earlier, trypsin inhibitor of SMBP was regarded inactive considering the high in vitro disappearance value of DM and CP. However, active trypsin inhibitor may exist in SMBP, resulting in the necessity of down adjusting the nutritional values of SMBP found in the present study.
In conclusion, estimated ME ranged from 4,311 to 4,619 kcal/kg as-is basis, and in vitro protein utilization of soybean milk by-product exceeded 90%. Considering these values, soybean milk by-product can be a good protein source for pigs. However, as energy values and protein utilization of soybean milk by-products are variable, different energy and protein values should be used for each soybean milk by-product considering the trypsin inhibitor effects.