What do we know about this problem?
High-quality postoperative analgesia is essential in lower segment cesarean sections and a variety of multimodal analgesia approaches are used.
Regional anesthesia (both TAP and ESP blocks) is commonly used for postoperative pain relief as it reduces the need for systemic analgesia, provides a longer duration of analgesia, and is relatively safe.
What does this study contribute?
TAP and ESP blocks provide similar analgesia with comparable consumption of tramadol in the first 24 hours post-cesarean section and no difference in pain scores at rest/on movement.
INTRODUCTION
High-quality postoperative analgesia is essential in cesarean sections to ensure fast recovery to the mother, as well as bonding and caring for her newborn infant. Two of the components of post-cesarean pain are visceral pain due to uterine stimulus and a deep component of somatic pain caused by nociceptors in the abdominal wound. 1,2
Multimodal analgesia for postoperative pain relief after cesarean sections includes intrathecal, epidural and/or systemic opioids and regional techniques like truncal blocks. The administration of intrathecal morphine provides better pain relief but is associated with a range of side effects such as nausea, urinary retention, drowsiness, extensive pruritus, and respiratory depression; hence, regional anesthesia has gained popularity.3,4
Truncal blocks like transversus abdominis plane block (TAP) and erector spinae plane block (ESP) are two of the most commonly used techniques for postoperative pain relief after cesarean deliveries. Both these blocks reduce the need for systemic analgesics, provide analgesia for a longer period with higher quality, and are relatively safer with fewer complications.5,6
Conventional TAP block is given by injecting local anesthetic between the internal oblique and transversus abdominis muscle plane, thus affecting the sensory nerves of the anterolateral abdominal wall, delivering solely somatic analgesia with dubious visceral analgesia. 6-9 Forero et al. 10 proposed the unique ESP block, an interfascial plane block where a local anesthetic is injected deep into the erector spinae muscle. ESP block is said to have an analgesic effect on both somatic and visceral pain by blocking the ventral and dorsal rami, as well as rami communicantes of the thoracic and abdominal spinal neurons, which include sympathetic nerves, thus providing superior analgesia than TAP block.11-13
We hypothesized that bilateral ESP block after cesarean section would provide more effective and prolonged pain relief than TAP block. our primary objective was to compare the analgesic efficacy between ESP versus TAP blocks in terms of total tramadol consumption in the first 24 hours. Other secondary outcomes included duration of analgesia, the VAS scores for pain at rest/on movement, patient satisfaction, and adverse effects.
METHODS
After receiving "Institutional ethics committee clearance (Registration No. ECR/146/Inst/KA/2013/RR-19) (IEC: 618/2018)", a prospective double-blind randomized superiority trial was conducted at our tertiary care hospital from March 2019 to March 2020 and was prospectively registered with the "Clinical Trials Registry of India (Registration No. CTRI/2019/03/018103)". All subjects gave their informed consent, and the protocol followed the 2013 Declaration of Helsinki ethics norms. Pregnant women between the ages of 18 and 40 who were undergoing spinal anesthesia for elective cesarean sections with a BMI between 18.5-34.9 kg/ m2 were included in the study. Patients with coagulation abnormalities, local infection at the block site, a history of allergic reactions to the study drugs, or a BMI >35 kg/m2 were excluded.
one blinded investigator performed the preoperative evaluation, discussed the techniques, and demonstrated the use of the patient-controlled analgesia (PCA) pump and subsequently assessed the pain scores using the visual analogue scale (VAS). This investigator followed up the patients for 24 hours after the intervention, recording pain scores, tramadol use, patient satisfaction, and watched for any adverse reactions. Based on the group assigned, another investigator performed either a TAP or an ESP block. The patients could not be blinded as they were conscious and both the blocks required different positioning.
The fasting status was confirmed on the day of surgery, the patient was transferred to the operating room and initial vital parameters were recorded. Under strict aseptic precautions, all patients received spinal anesthesia in the left lateral position using 10mg of hyperbaric bupivacaine with 10 mcg of fentanyl. Transverse incision (Pfannenstiel) and lower segment cesarean section were performed after ensuring a sensory blockade of at least T10 level. At the end of the surgery, patients were assigned to either the TAP or ESP block group, based on a computer-generated random number table hidden in an envelope that was unsealed right before the cesarean section.
Institutional protocol for pain management: As per our hospital practice, all the patients were on PCA tramadol and scheduled intravenous acetaminophen. A PCA 2ml (25mg) tramadol bolus, with a lockout interval of 10 minutes, was administered for breakthrough pain, as per our institutional protocol.
A TAP block was administered using a linear probe which was positioned in a transverse plane over the lateral abdominal wall on the midaxillary line, halfway between the iliac crest and the costal margin, and the block needle was directed in the plane from the medial to the lateral direction. Between the internal oblique and transversus abdominis muscle on each side, 20ml of 0.2 % ropivacaine with 25mcg of dexmedetomidine were injected.
The ESP block was administered using a linear probe which was positioned 2cm away from the midline, in a paramedian sagittal orientation. The trapezius, rhomboid major, and erector spinae muscles were identified and a 10cm block needle was inserted in-plane from cranial to caudal and the block was performed at the T9-T10 level. 20ml of 0.2% ropivacaine with 25mcg of dexmedetomidine were deposited between the erector spinae muscle and the transverse process on either side.
In the postoperative period, all patients were put on a PCA pump (20cc syringe with 12.5mg/ml of tramadol). A PCA bolus of 2ml (25mg) was set with a lockout interval of 10 minutes. Patients were followed up for the first 24 hours at 0, 1, 2, 4, 8, 12, 18, and 24 hours. VAS was used for pain assessment at rest and on movement which included a 10 cm horizontal line labeled "no pain" (score 0) and "unbearable pain" (score 10). A "three-point scale (1, excellent; 2, good; 3, poor)" was used to assess patient satisfaction. Any adverse effects or complications during the study in either group were recorded.
The primary outcome analyzed total consumption of PCA tramadol in the first 24 hours and secondary outcomes analyzed the time required for the first demand of analgesia, VAS score post-surgery for pain at rest and on movement, patient satisfaction with pain management and adverse effects such as nausea, vomiting, bradycardia, hematoma, intravascular injection, local anesthetic systemic toxicity, pneumothorax, and hypersensitivity.
Statistical analysis: The sample size was determined based on the results of a pilot study with 5 patients in each group that revealed a total tramadol consumption of 90±40.62 mg in the TAP group and 65±37.41 mg in the ESP group. With an 80-% power of the study and a 95 % confidence interval, 25 patients were needed in each group. The statistical analysis was carried out using the "SPSS statistical software package for Windows, version 20.0. (SPSS Inc., Chicago, IL, US)". The demographic characteristics were compared between both groups using the chi-square test for dichotomous variables. Descriptive statistics were used for continuous outcomes using "medians with their corresponding interquartile ranges (IQR)" and the "Mann-Whitney U test" was used for comparisons between the groups. A "P-value < 0.05" was considered statistically significant.
RESULTS
Our study group consisted of 50 mothers, 25 of whom were randomly assigned to each group, and no deviations from the study protocol were observed (Figure 1). Gender, weight, height, and BMI were distributed similarly in both groups (Table 1).
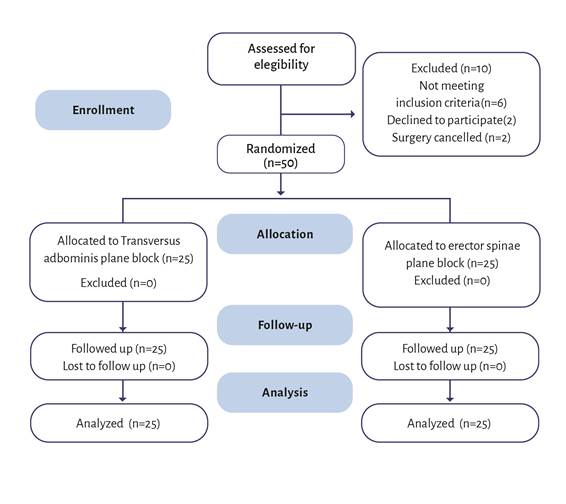
ESP block: Erector spinae plane block; TAP block: Transversus abdominis plane block. Source: Authors.
Figure 1 "CONSORTdiagram"of patient flowthrough the study.
Table 1 Demographicdetailsof patients.
| Characteristics | TAP block (Mean±SD) (n=25) | ESP block (Mean ± SD) (n=25) | p-value |
|---|---|---|---|
| Age (inyears) | 29.2 + 3.9 | 29.8+3.5 | 0.521 |
| Weight (kg) | 65.5 + 8.5 | 60.7 + 9.1 | 0.266 |
| Height(incm) | 153.0 + 8.7 | 154.0 + 7.5 | 0.660 |
| BMI (kg/m2) | 27.4 + 3.9 | 26.4 + 2.9 | 0.306 |
ESP block: Erector spinae plane block; SD: Standard deviation; TAP block: Transversus abdominis plane block.
Source: Authors.
Primary outcome
There was no significant difference in tramadol consumption between both groups (p=0.109) (Table 2). Patients in the TAP group received a median tramadol dose of 100mg (IQR 63-125) (10 morphine equivalents) in the first 24 hours and those in the ESP group received a median dose of 75mg of tramadol (IQR 38-100) (7.5 morphine equivalents).
Table 2 Primary and secondary outcomes.
| Variables | TAP block [Median (IQR)*] | ESP block [Median (IQR)*] | p-value** |
|---|---|---|---|
| Tramadol consumption (in mg) (in morphine equivalents) | 100 (63-125) 10 | 75(38-100) 7-5 | 0.109 |
| Time required for first demand of analgesia (min) | 210(135-315) | 270 (225-405) | 0.030 |
ESP block: Erector spinae plane block; TAP block: Transversus abdominis plane block. "*IQR-lnterquartile range; **Mann-Whitney U test.
Source: Authors.
Secondary outcomes
1. Time required for first demand of analgesia: The median time for first rescue analgesia after the procedure was 210 min (135-315) and 270 min (225-405) in the TAP block group and ESP block group respectively, which was statistically significant (p = 0.03) (Table 2).
2. Post-operative VAS score for pain at rest and on movement: There were no differences in pain scores at any time point within the first day after surgery (Table 3).
Table 3 Comparison of pain scores at rest and on movement.
| VAS Score at Rest | VAS Score on movement | |||||
|---|---|---|---|---|---|---|
| Time Interval | TAP block Median [IQR]* (n= 25) | ESP block Median [IQR]* (n= 25) | p-value** | TAP block Median [IQR]* (n= 25) | ESP block Median [IQR]* (n= 25) | p-value ** |
| 0 Hour | 0[0-0] | 0[0-0] | 0.641 | 0[0-0] | 0[0-0] | 0.253 |
| 1 Hour | 0[0-1] | 0[ 0-1] | 0.549 | 1[1-1] | 1[1-1] | 0.253 |
| 2 Hour | 1[0-3] | 2[ 0-2] | 0.741 | 2[1.5-4] | 3[2-3] | 0.764 |
| 4 Hour | 2[1-3] | 2[ 0.5-3] | 0.828 | 3[2-4] | 3[2-4] | 0.944 |
| 8 Hour | 2[1,3-5] | 3[1.5-3.5] | 0.744 | 3[2-4] | 3[2-4] | 0.477 |
| 12 Hour | 3[1-3] | 3[ 1-3] | 0.749 | 4[2.5-4] | 4[3-4] | 0.903 |
| 18 Hour | 3[1-3] | 3[ 1.5-3] | 0.713 | 3[2-4] | 4[3-4] | 0.872 |
| 24 Hour | 2[2-4] | 3[ 1.5-3] | 0.469 | 3[2-4] | 3[2-3] | 0.313 |
ESP block: Erector spinae plane block; TAP block: Transversus abdominis plane block; VAS: Score Visual analog score. *IQR: Interquartile range **Mann-Whitney U test.
Source: Authors.
Pain scores on movement were also similar between both groups in the first 24 hours after surgery. Figures 2 and 3 compare the trends of pain scores between both groups at rest and on movement, respectively.
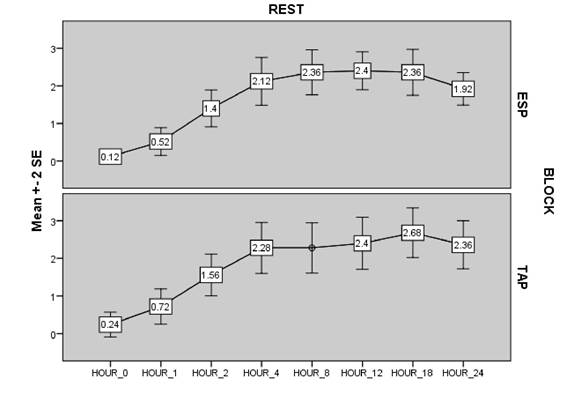
ESP block: Erector spinae plane block; TAP block: Transversus abdominis plane block; VAS: Score-Visual analog score. Source: Authors.
Figure 2 Mean ± SD VAS scores at rest.
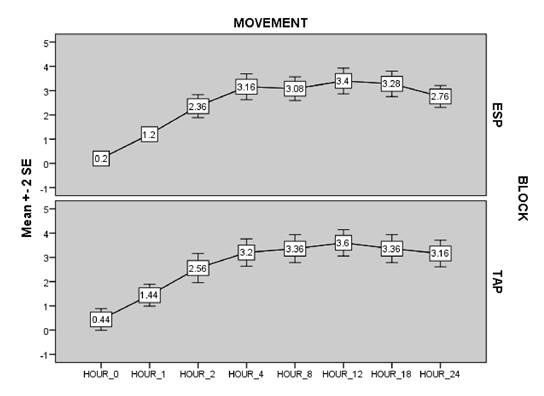
ESP block: Erector spinae plane block; TAP block: Transversus abdominis plane block; VAS: Score-Visual analog score. Source: Authors.
Figure 3 Mean ± SD VAS scores on movement.
3. Patient satisfaction with pain management: Patient satisfaction was divided into "poor, good, and excellent" categories. There were no significant differences between the groups in patient satisfaction (p=0.72). Among TAP block patients, 4 had "excellent" satisfaction, 16 had "good" satisfaction and 5 had "poor" satisfaction, whereas among ESP block patients 5 had "excellent" satisfaction, 17 had "good" satisfaction and 3 had "poor" satisfaction.
4. Adverse events: Neither group experienced any adverse effects such as nausea, vomiting, bradycardia, hematoma, intravascular injection, local anesthetic systemic toxicity, pneumothorax, and hypersensitivity.
DISCUSSION
Postoperative pain management in patients who undergo elective cesarean section is key to ensure a fast recovery. This study focused on two different regional anesthetic techniques in caesarean section. Our results showed no clinically significant differences between TAP and ESP blocks in pain control and analgesic consumption. Patient satisfaction was comparable between both groups.
Intravenous opioids, non-steroidal anti-inflammatory medications, rectal analgesic suppositories, and epidural analgesia are usual modalities employed for pain management in cesarean sections, each with its own set of drawbacks such as frequent dosing and constant monitoring, to name a few. Regional techniques involving ultrasound-guided TAP and ESP blocks have been gaining popularity in recent years for providing superior postoperative analgesia with fewer side effects. 1-5
In our study, there was a median tramadol consumption of 100mg and 75mg in the TAP block group and ESP block group respectively in 24 hours, which notwithstanding the intergroup variation, was not statistically significant (p = 0.109). Our findings are similar to those of Srivastava et al.14, who used TAP block with 0.25 % bupivacaine for postoperative analgesia in elective cesarean deliveries (n=62) and found that tramadol use was reduced by 50% in patients who received versus those who did not receive a TAP block, 48 hours after surgery (p = 0.001). Multiple studies have demonstrated that TAP block provides postoperative analgesia in patients following cesarean section, with the added benefit of procedural simplicity, a consistent level of analgesia, and longer duration.15-20 However, a recent study by Boules et al.21 compared the efficacy of TAP block with ESP block post-cesarean deliveries under spinal anesthesia (n=60) and found that the median total tramadol consumption in the first 24 hours was significantly higher in the TAP group (n=30) (125mg) than in the ESP group(n=30) (100mg) (p=0.003), which can be explained by the lack of retrograde spread of the TAP block into the paravertebral space, thus providing poorer analgesic effect when compared to ESP block. Over the last decade, several studies involving ESP block have concluded that it has a longer duration of analgesic activity, gives more effective pain relief, extends the time to initial analgesic necessity, and decreases tramadol consumption. 22-25
In this study, the median time elapsed until the demand for the first rescue analgesia after the procedure was 270 min (4.5 hours) in the ESP group and 210 min (3.5 hours) in the TAP block, respectively; this was statistically but not clinically significant (p-value = 0.03).
Our findings are consistent with with the study by Kamel et al.22 wherein he analyzed the time required for first morphine demand in patients (n=48) receiving either TAP or ESP block after complete abdominal hysterectomy and found that the time for first morphine demand was 10.58 ± 2.35 hours in the TAP group and 14.81 ± 3.52 hours in the ESP group, which was statistically significant (p = 0.001). This difference has been linked to the epidural, neural foramina, and intercostal dissemination of local anesthetics in the ESP block, which is more widespread and covers a larger dermatomal region than the TAP block.26
This study found that the mean VAS score in both groups at rest and on movement at different time intervals were similar, implying that ESP and TAP blocks provided equivalent pain control. This was similar to the metanalysis by Cai Q et al 27 where studies showed that although pain scores in patients with ESP block were lower in the first 24 postoperative hours than in controls, whether at rest or during movement, the average pain score reduction was only between 1.2 and 2.27, which apparently didn't change dramatically based on the 0-10 scale, which had no clinical significance as shown in the previous study where the average pain score change of 1.3 had only minimal clinical change in pain.28
According to our study, there was no discernible difference in the patient satisfaction scores obtained 24 hours after the procedure; this is indicative of a comparable quality in terms of pain relief among both groups, which is similar to the systematic review on the efficacy of ESP block in post-cesarean section patients by Rebeiro et al.29 wherein ESP block resulted in reduced tramadol intake and a longer blockade duration; however, there was no significant difference in patient satisfaction between the two groups in the studies analyzed. The possible reason is that although pain control is an important consideration, there is a wide spectrum of factors that affect overall patient satisfaction post-cesarean deliveries.
Overall, our study did not show any evidence of superiority of ESP over TAP block; this is consistent with the metanalysis by Rebeiro et al.29 wherein most of the RCTs analyzed had spinal block as the primary anesthesia in both groups - which is identical to our study - and it showed that the spinal neuraxial block per se provided a protective factor against pain among patients.
LIMITATIONS OF THE STUDY
A control group was not included to show the effectiveness of these blocks as it could have proved the efficacy of these fascial plane blocks.
Although dexmedetomidine has not yet been approved for use in analgesic fascial blocks, there are several published Western and Indian studies confirming its use in fascial blocks.
Patient blinding was not feasible as patients were awake and both blocks required different positioning.
We could not compare the time to recovery as no data was available on the length of hospital stay or time to mobilization.
CONCLUSION
TAP and ESP blocks offered similar pain control and tramadol consumption within the first 24 hours post-cesarean section, with no difference in overall patient satisfaction. Both regional anesthetic techniques are adequate alternatives for pain management in cesarean section.
ETHICAL DISCLOSURES
Ethics committee approval
After receiving "Institutional ethics committee clearance (Registration No. ECR/146/Inst/KA/2013/RR-19) (IEC: 618/2018)", a prospective, double-blind, randomized, superiority trial was conducted at our tertiary care hospital from March 2019 to March 2020 and was prospectively registered with the "Clinical Trials Registry of India (Registration No. CTRI/2019/03/018103)".
Protection of human and animal subjects
The authors declare that no experiments were performed on humans or animals for this study. The authors declare that the procedures followed were in accordance with the regulations of the relevant clinical research ethics committee and with those of the Code of Ethics of the World Medical Association (Declaration of Helsinki).
CONFIDENTIALITY OF DATA
The authors declare that they have followed the protocols of their work center on the publication of patient data.
ACKNOWLEDGEMENTS
Authors' contributions
NVKR: Study planning, data collection, interpretation of results, and initial writing of the manuscript.
MP: Conception of the original project, Study planning, data collection, interpretation of results, data analysis, and final writing of the manuscript.
STK: Conception of the original project, study planning, interpretation of results, and final writing and approval of the manuscript.
SM: Study planning, data collection, interpretation of results, and initial writing of the manuscript.











 text in
text in 



