Introduction
Eosinophilic colitis is a rare gastrointestinal pathology.1 The classic concept of primary eosinophilic diseases of the digestive tract (EGID) is that they share a common factor: the infiltration of eosinophils into the mucosa without having a known cause of peripheral eosinophilia. Although the latter is not usually present in many cases (as in eosinophilic esophagitis or eosinophilic colitis), the symptoms are essential to suspect these pathologies.2
Since the first publication in Germany in 1937 by Kaijser on EGID,3 isolated cases of these diseases have been presented in the literature, but currently, there are no clinical guidelines in this regard, particularly for eosinophilic colitis. The latter is a disease poorly studied and requires well-defined criteria for its diagnosis and treatment.
Presentation of the case
The following is a case of eosinophilic colitis with coexisting ascites, diagnosed and treated in a private hospital in Lima, Peru. We received a 23-year-old male patient who had been ill for six months and reported a clinical profile characterized by oppressive heart-type abdominal pain in the epigastrium, which subsequently migrated to the mesogastrium, associated with nausea, general malaise, and loss of appetite. After three months, liquid, mucus-free, bloodless, non-enteric stools began to appear approximately three to four times a day, only during daylight.
In our hospital, the first tests requested (on October 6, 2021) were the following: hemoglobin (13.7 g/dL), hematocrit (39%), leukocytes (8980 cell/μ L), eosinophils (38%), lymphocytes (15%), segmented neutrophils (40%), and platelets (208,000 cell/μL). From the main finding of peripheral eosinophilia, we began to look for etiologies, and the following results were obtained: Toxoplasma gondii immunoglobulin G (IgG) and M (IgM) negative, enzyme-linked immunosorbent assay (ELISA) for Fasciola hepatica negative, Toxocara IgG and IgM negative, immunoglobulin E (IgE) 67.3 IU/mL (normal), and antinuclear antibodies negative. The peripheral lamin described a red series of normal quantity and morphology, while the white series showed marked eosinophilia and normal morphology. The thrombocytic series resulted in a normal amount and morphology. Other outcomes were the following: lactate dehydrogenase (208 IU/L, normal), microglobulin β2 (1.33 mg/L, normal), HIV (negative), hepatitis A and B profile (negative), Epstein-Barr IgG and IgM (negative), cytomegalovirus (CMV) IgG and IgM (negative), antigenic test for severe acute respiratory syndrome coronavirus type 2 (SARS-CoV-2) (negative), vitamin B12 (482.1 pg/mL, normal), total protein (5.66 g/dL), albumin (4.03 g/dL), C-reactive protein (CRP) (9.8 mg/dl), and glucose (105 mg/dL). The functional stool examination was diarrheal, without steatorrhea, starches, food remains, red blood cells, negative Thevenon test, and negative inflammatory reaction. Parasites in feces (six samples) were negative.
The treating clinicians decided to start the search for the gastrointestinal etiology of eosinophilia associated with gastrointestinal symptoms presented by the patient. First, he underwent an endoscopy in which moderate erythematous gastritis (histopathological examination: mild superficial chronic gastritis, without Helicobacter pylori, atrophy, or intestinal metaplasia) and nonspecific duodenitis (histopathological examination: mild chronic duodenitis with intraepithelial lymphocytes 8-10/100 epithelial cells, without typical findings of a specific disease) were found. Then, he underwent a colonoscopy that presented the following findings: in the distal ileum, from approximately 15 cm from the ileocecal valve to the distal, marked mucosa congestion was observed with “punched” erosions, punctate erythema, and loss of mucosal vascularization (Figure 1). On histopathological examination, moderate chronic ileitis was found with no specific findings. In the cecum, at the level of the ileocecal valve and proximal to the appendicular orifice, erythema of the mucosa and partial loss of mucosal vascularization were observed (Figure 2). There was a significant increase in eosinophils at the level of the lamina propria and crypts, which reached approximately > 200 eosinophils in a high-power field in the histopathological examination (Figures 3 and 4). This eosinophilic infiltrate was also accompanied by plasma cells, scarce lymphocytes, and cryptic abscesses, which are findings related to eosinophilic colitis.

Figure 3 Biopsy of the cecum. Overview of the microscopic image. Numerous eosinophils surround the crypts. Source: Pathological Anatomy area of Clínica San Gabriel.
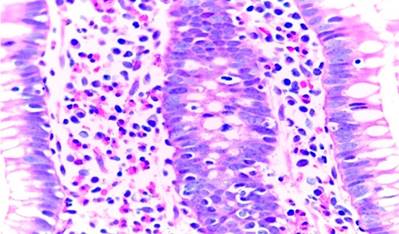
Figure 4 Cecum biopsy. It shows diffuse infiltration of eosinophils into the mucosa. Source: Pathological Anatomy area of Clínica San Gabriel.
The patient underwent an abdominal ultrasound in which an abdominopelvic free fluid was observed, approximately 1150 mL, determined by the four quadrants: the liver had its morphology preserved with no signs of cirrhosis, and the rest of the organs of the abdominal cavity were within normal parameters. Due to the not-so-abundant amount of ascitic fluid, it was decided to perform an abdominal paracentesis oriented under ultrasound guidance (by interventional radiology), in which turbid yellow fluid was obtained without any bad odor, from which approximately 120 mL were extracted for studies. The following were the rest of the results: ascitic fluid cytochemistry, cell count (3150 cell/UL), polymorphonuclear (90%), mononuclear (10%), Gram stain (negative), glucose (100 mg/dL), albumin (3.56 g/dL), serum-ascites albumin gradient (GASA) (1.18), culture (negative), and adenosine deaminase (ADA) (5.53 IU/L, normal).
With these findings, the diagnosis of subserous eosinophilic colitis was determined, so starting treatment with oral prednisone at doses of 40 mg per day for two weeks as induction therapy was decided. Blood count was monitored, and an improvement in symptoms and eosinophil count (successful clinical remission) was observed. Therefore, maintenance therapy was continued through progressive withdrawal of prednisone at a rate of 5 mg per week and reached up to 10 mg per day. The results were favorable (Table 1).
Table 1 Results of the patient’s laboratory tests
| 06/10 | 20/10 | 27/10 Initiation of induction therapy with prednisone | 11/11 After 14 days of induction therapy | 15/12 After 30 days of maintenance therapy | |
|---|---|---|---|---|---|
| Hb (g/dL) | 13,7 | 14,5 | 13,1 | 13,8 | 14,2 |
| Hto (%) | 39,3 | 40,1 | 37,2 | 40,6 | 41,9 |
| Leu (cel/μL) | 8980 | 11600 | 9710 | 10800 | 5300 |
| Eos (%) | 38 | 41 | 43 | 2 | 4 |
| Eos (cel/μL) | 3430 | 4830 | 4590 | 160 | 220 |
| Lymph (%) | 15 | 22 | 18 | 26 | |
| Seg (%) | 40 | 30 | 73 | 59 | |
| CRP (mg/L) | 9,8 | 0,49 | 0,7 |
Eos: eosinophils; Hb: hemoglobin; Hto: hematocrit; Leu: leukocytes; Lymph: lymphocytes; CRP: C-reactive protein; Seg: segmented. Source: Authors’ own research.
In the abdominal control ultrasound (15/12: after 30 days of maintenance therapy with prednisone), the liver of preserved morphology was observed, and there was no free fluid in the cavity. The rest of the organs had preserved characters.
Discussion
Eosinophilic colitis is a gastrointestinal pathology that is part of the EGID group, along with eosinophilic esophagitis and eosinophilic gastroenteritis, which are defined by an excess count of eosinophils in the thickness of the wall of the gastrointestinal tract, without having an established secondary etiology. It is such a rare disease that, for example, in the United States, an overall prevalence of 2.1 per 100,000 people is reported, and it is more frequent in adults (1.6/100,000), unlike most EGIDs, which have an epidemiological inclination to pediatric populations. However, it is important to highlight that eosinophilic colitis has a bimodal pattern of presentation: first, in neonates and infants and then in adults. Due to its preponderance in urban and suburban areas, it may be related to high socioeconomic status or people with better levels of education.4,5
The clinical profile of EGIDs depends on the level of involvement of eosinophilia in the gastrointestinal tract wall. According to the Klein classification, there are three types: mucosa (57.5%), including nausea, vomiting, abdominal pain, digestive bleeding, diarrhea, and others; muscular (30%), including intestinal obstruction (presented even as recurrent sigmoid volvulus) and gastrointestinal motility disorders; and serous (12.5%), including ascites, meteorism, peritonitis.6,7 Under this same criterion, we can say that the symptoms of eosinophilic colitis depend on the level of eosinophilic involvement, but they can co-occur in the same patient. Thus, in pediatric populations, there are clinical forms characterized by acute self-limited bloody diarrhea and, in adults, abdominal pain or chronic diarrhea, associated or not with the other symptoms described depending on the level of gastrointestinal involvement.8 In another review, three types of disease that do not differ much from the previous ones are described: mucosal, transmural, and subserous (the latter, which is the reason for this publication, with ascites, the most benign and, in turn, the most infrequent of the three).5
The finding of ascites by ultrasound, with the results of the sample exposed above, gives the form of subserous presentation to this disease so rare in our setting. Similarly, there have been cases reported in the literature with the presentation of eosinophilic ascites associated with gastroenteritis or colitis, such as the one reported by Cuko et al.,9 presenting a 37-year-old female patient whose leukocyte count in ascitic fluid was 8800, 94% polymorphonuclear (PMN), and with eosinophilic infiltrate in the duodenal mucosa. Likewise, in the case reported by Elsadeck,10 a 41-year-old male patient was described with gastroenteritis and colitis associated with infiltration of eosinophils, plasma cells, and lymphocytes at the mucosal level, in addition to the characteristic findings of eosinophilic ascites. Finally, a case reported by Amado et al.1 describes a 55-year-old woman presenting with subserous eosinophilic colitis potentially triggered by an herbaceous product (they do not describe it as an etiology of colitis).
To diagnose this disease, it is important to know its differential diseases, listed in Table 2, including some drugs such as gabapentin and pregabalin.11 It is relevant to note that eosinophilic colitis can mask colon adenocarcinoma, such as in the case reported by Milne et al.,12 or also be associated with another pathology such as Crohn’s disease (overlap), as in the case described by Katsanos et al.13 It is relevant to know that biopsies are important for the diagnosis of eosinophilic colitis. However, it is essential to differentiate it from the so-called colonic eosinophilia since, in the first one, the inflammatory infiltrate consists almost exclusively of eosinophils, unlike the second, in which there is a mixed inflammatory infiltrate and, generally, there is a known cause, such as allergies, drugs, inflammatory bowel disease, or infections, among others.14 In fact, the study conducted by Arévalo et al.15 found that, of 68 patients diagnosed with lymphocytic colitis (a subtype of microscopic colitis), there was a coexistence of 76.5% of elevated eosinophils in the colonic mucosa. The same study found that 51.4% of patients were diagnosed with eosinophilic colitis but only with biopsy, as the exclusion of other possible etiologies of eosinophilia is not described.
Table 2 Eosinophilic colitis: differential diagnoses2
| Parasitic colitis |
| Eosinophilic gastroenteritis |
| Hypereosinophilic syndrome |
| Inflammatory bowel disease |
| Drug-induced colitis: - Nonsteroidal anti-inflammatory drugs - Rifampicin - Clozapine - Tacrolimus - Gold salts |
| Allogeneic bone marrow transplant |
| Other: Toulouse-Hunt syndrome, vasculitis (e.g., Churg-Strauss syndrome) |
| Acute radiation colitis |
Taken from: Alfadda AA et al. Therap Adv Gastroenterol. 2011;4(5):301-9.
Endoscopic findings in patients with eosinophilic colitis are nonspecific. In fact, they may have a normal appearance in up to 70% of cases, so there may be overlap with diarrhea predominant-irritable bowel syndrome (IBS-D), as described by the study by Carmona-Sánchez et al.,16 which found a prevalence of 4.7% of patients with eosinophilic colitis who had also been diagnosed as IBS-D. Other common manifestations include erythema, edema, decreased mucosal vascularization, erosions, and ulcers. Regarding the location of the lesions, they can be found more frequently in the right and left colon and as pancolitis in up to 11% of cases.6 In our case, there were findings both in the distal ileum (“in punch” erosions, erythema, loss of mucosal vascularization) and in the cecum (erythema and loss of mucosal vascularization). In the other colon segments, there may have been the eosinophil counts necessary to diagnose eosinophilic colitis. However, the required samples were not obtained.
As already described above, a biopsy is essential. Nevertheless, eosinophils represent a normal component of inflammatory cells in the colon. Only they can vary in number in different colon segments, as they can also be part of the inflammatory infiltrate in various colon diseases.17 Eosinophilic density is usually estimated semiquantitatively and requires counting the number of eosinophils by high-power fields (HPF) and the mean count. This density is not yet clearly established to determine the diagnosis of eosinophilic colitis. However, some options have been proposed, such as more than 60 (or even 100) eosinophils/HPF, usually observed in the cecum18,19 or more than 40 eosinophils/HPF in at least two different segments of the colon.6 The finding was more than 200 eosinophils/HPF in our case. The other findings that can be found in histology are mentioned in Table 3.
Table 3 Microscopic findings in eosinophilic colitis6
| Histological characteristics | Prevalence in eosinophilic colitis |
|---|---|
| Inflammatory mucosal infiltrate | 100 |
| Intraepithelial eosinophils | 77 |
| Eosinophilic degranulation | 51 |
| Architectural distortion | 42 |
| Acute inflammation | 26 |
| Mucosal atrophy | 23 |
| Eosinophilic microabscesses | 21 |
Taken from: Macaigne G. Clin Res Hepatol Gastroenterol. 2020;44(5):630-637
Treating this disease is sometimes difficult, and no international guidelines define it. However, it has been reported that an improvement in gastrointestinal symptoms is observed with corticosteroids, mainly prednisone. Thus, its use in doses of 1-2 mg/kg/day orally for eight weeks with its progressive withdrawal (induction and maintenance therapies) has achieved effectiveness between 80% and 100% of cases.6,20 Clinical remission has also been achieved by induction therapy with oral prednisone (20-40 mg per day) for two weeks and subsequent maintenance therapy with progressive dose withdrawal, up to 5 mg daily.5 This last scheme was the one indicated in the patient and caused him to have remission of symptoms, as well as the normalization of eosinophilia after two weeks of induction with prednisone at 40 mg daily and later with progressive withdrawal, achieving a normal eosinophil count again at four weeks and absence of ascites. Furthermore, this form of treatment was indicated in some cases, such as the one described by Yep Gamarra et al.,21 which had a presentation of gastroenteritis with ascites, in that of Sánchez et al.,22 and as in the case presented by Páramo-Zunzunegui et al.,23 all with successful clinical remission.
In addition, there are other treatment schemes, such as the role of a hypoallergenic diet (even determined as a first therapeutic measure), anti-inflammatory agents such as mesalazine, immunomodulators such as azathioprine and antitumor necrosis factor (anti-TNF; infliximab and adalimumab) in refractory or steroid-dependent cases, and montelukast, among others.8,24
Conclusions
After having made a detailed description of our clinical case of subserous eosinophilic colitis, we can say that this is an uncommon disease and that management guidelines on this pathology are still pending. When a patient has eosinophilia associated with gastrointestinal symptoms, an exhaustive search should be carried out to rule out secondary etiologies and accurately diagnose eosinophilic colitis. Corticosteroids such as prednisone are important because they mitigate gastrointestinal symptoms and improve laboratory and imaging parameters.











 texto en
texto en 





