Case presentation
A 71-year-old female patient was admitted to the emergency room with a history of arterial hypertension, type 2 diabetes mellitus, and chronic use of acetylsalicylic acid and consulted for hematochezia on multiple occasions with no other symptoms. On admission, she was found to be hemodynamically stable, with paraclinical tests showing microcytic and hypochromic anemia and elevated urea nitrogen. Preparation for colonoscopy was indicated, in which traces of recent bleeding without a clear cause were found. Upon reaching the distal transverse colon, it was impossible to advance despite multiple attempts due to a marked angulation, so the procedure was suspended. During follow-up, she persisted with bleeding associated with hemodynamic instability and anemia of 3 grams (hemoglobin [Hb]: 7.1 g/dL). Transfusion therapy of 2 units of packed red blood cells (PRBC) was indicated, and she was later taken to upper endoscopy, with no evidence of injury or bleeding. Mesenteric angiography was advanced without evidence of active bleeding. The patient persisted with hemodynamic instability, which required starting vasopressor support and management in the intensive care unit (ICU).
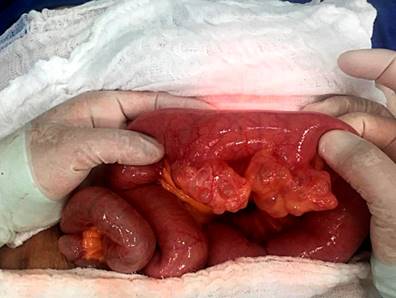
She was taken to surgery with a hemicolectomy plan due to suspicion of diverticular bleeding, with intraluminal blood in the small intestine. A manual inspection was performed, which revealed multiple multilobulated diverticula in the jejunum and a single diverticulum in the fourth portion of the duodenum. An intraoperative antegrade enteroscopy was performed with evidence of recent bleeding and multiple clots in the previously mentioned diverticula, requiring resection of the compromised jejunal segment and diverticulectomy of the distal duodenal diverticulum with adequate bleeding control. (Figures 1 y 2)
Discussion
The first reports of jejunal diverticulosis were described by Sommervit in 1794 and Cooper in 18071. There are only case series in the literature due to its low incidence, approximately 0.5% to 7.1%2. However, depending on the identification tool, it can vary from 0.2%-4.5% in autopsies, 0.5%-2.3% in studies with contrast, 0.16%-27% in endoscopic retrograde cholangiopancreatography (ERCP), and up to 7.5% in endoscopic ultrasound (EUS; periampullary)10. It mainly affects men between 60 and 702,9. Diverticula are acquired lesions with mucosal, submucosal, and serous involvement without the muscular layer, which is why they are considered false diverticula, which are fragile and thin. They frequently occur on the small intestine’s mesenteric side, where the arteries enter the intestine1,9. However, manifestation on the antimesenteric edge has also been described10. They are more frequent at the level of the duodenum and more rarely at the level of the jejunum and ileum9. Three-quarters are represented by the periampullary diverticula10. Approximately 79% are located in the duodenum, and distal to it; 80% manifest at the jejunal level, about 15% in the ileum, and 5% in both, with multiple manifestations at the jejunal level, decreasing in quantity towards the distal part and sometimes with a single diverticulum in the ileum9,10. Diverticula form during periods of increased intraluminal pressure, such as inflammation or intestinal dyskinesia, which produces areas of high pressure in segments of the small intestine10.
Most are asymptomatic and are found incidentally in studies for other causes1, such as imaging studies or during abdominal surgery9. It is important to take these diverticula into account since most of the clinical manifestations can be variable (abdominal pain, nausea, dyspepsia, emesis, rumbling, altered bowel habits) or resemble a picture of bacterial overgrowth that causes malabsorption9,10; however, and although less frequent, it can manifest with severe conditions with a high mortality rate such as obstruction, perforation, volvulus, infection, and bleeding1,8,9. When these diverticula display secondary gastrointestinal bleeding, they vary since they can present with pictures of melena, hematochezia, and, in some cases, hematemesis, leading to the patient’s clinical instability 3,6. Bleeding from jejunal diverticula ranges from 3% to 8%, and although the manifestation is variable, as mentioned, most occur as rectal bleeding, which is confusing. When a patient presents to the emergency department with rectal bleeding, the most probable cause is considered to be lower gastrointestinal bleeding, in addition to having a high risk of mortality due to the delay in diagnosis4; however, current gastrointestinal bleeding guidelines mention that if a patient presents with rectal bleeding that causes hemodynamic instability, upper gastrointestinal bleeding should be considered10. It is believed that the bleeding mechanism of the jejunal diverticula is ulceration secondary to inflammation that affects an artery and causes bleeding1.
Some reports described using drugs, such as warfarin and low-dose aspirin, as risk factors1. In the case of our patient who uses acetylsalicylic acid (ASA) for secondary cardiovascular prevention, it is advisable not to suspend it. Remember that its mechanism of action occurs through the irreversible inhibition of cyclooxygenase-1, which mediates the synthesis of thromboxane; when ingesting the ASA, the synthesis of thromboxane returns to an average between 7 and 10 days; therefore, stopping ASA will have little impact on the initial clinical course due to the persistent antiplatelet effect of ASA in the first day or two after manifestation. By then, the patient will have undergone an endoscopic study with hemostasis. If ASA is suspended, it must be restarted once hemostasis is achieved since its effect will still be present. These two current recommendations have made it possible to demonstrate a decrease in mortality11.
The mechanisms that could explain the increase in bleeding secondary to non-steroidal anti-inflammatory drugs (NSAIDs) are direct damage to the mucosa, disruption of intercellular integrity, and activation of neutrophils by bacteria7. Diagnosis is a challenge because initial studies such as upper endoscopy and colonoscopy have limitations in finding the site of bleeding at this level, so the algorithm for suspicion of bleeding from the small intestine must be continued, and studies such as videocapsule endoscopy or enteroscopy when the patient is hemodynamically stable should be advanced. Angiography is a valuable tool, especially in the context of instability, since it can help locate the lesion and make differential diagnoses based on the characteristics of the extravasation. It is considered the gold standard5. Management is generally surgical since embolization by arteriography is not practical in most cases; however, if the cause of the bleeding is not found, the recommendation is to perform a subtotal colectomy, but if found, it performing a resection of the affected segment with primary anastomosis is suggested. If it is found in several parts, an intraoperative enteroscopy can be helpful to find the source of bleeding and resect only this segment9.
Conclusion
Bleeding jejunal diverticula are rare and difficult to diagnose as they can mimic signs and symptoms of upper or lower gastrointestinal bleeding. In this case, it presented with massive bleeding that the chronic intake of NSAIDs could have favored. Within the studies, selective mesenteric angiography is the gold standard for diagnosing active and fatal bleeding, but as in our case, it may not reveal the site. So, surgical management can be an essential tool for making a diagnosis and definitive treatment.











 texto en
texto en