1. INTRODUCTION
Currently, numerous water sources throughout the country are highly contaminated due to various industrial activities, including the extraction of crude oil [1], According to Ecopetrol SA [2], for each barrel of crude extracted, an average of four barrels of contaminated water (production water) is generated, a proportion that increases along with the maturity of the well [3].
The contaminated water resulting from the extraction processes and water used for reinjection must be treated before being discharged into natural sources, reinjected, or used in agricultural activities [4] Its composition may vary depending on the reservoir, but it is estimated that this production water is composed of oil (500 to 5000 ppm), sulfates, bicarbonates, cyanide, carbon dioxide, and heavy metals such as cadmium, arsenic, chromium, lead, mercury vanadium and zinc. Such pollutants change the organoleptic characteristics of water, as the contact of their components with the ecosystem causes negative impacts on the environment; given that their high salinity and the presence of heavy metals are potentially toxic to the fauna present in water bodies [5].
Conventional treatment technologies produce chemically stable emulsions that are difficult to break [6] and non-conventional technologies, such as osmosis, electro dialysis, exchange for ion removal (OR, IOR), among others, require high investment [7]
This is the scenario where the Advanced Oxidation Processes (AOPs) become relevant as they involve the generation of hydroxyl radicals (OH·), very potent and non-selective oxidizing agents, which chemically modify or mineralize organic pollutants and inorganic substances present in water bodies and samples of all types [8]-[10]. The (OH ) are generated from physical-chemical processes nvolving chemical agents such as ozone [11], hydrogen peroxide [12], Fe2+ (along with hydrogen peroxide in the Fenton reagent) [13] and physicochemical processes such as the electronic excitation of semiconductor materials (excitation of titanium dioxide using UV radiation) [14].
The heterogeneous photocatalysis is an advanced oxidation technology, which consists of using a semiconductor, commonly titanium dioxide (Ti02), to photocatalyze chemical reactions of organic and inorganic substances and obtain lower toxicity substances or carbon dioxide [15]. Through the process, the incident radiation excites an electron from the valence band to the conduction band, generating an electron-hole pair, charge carriers, which when approaching the semiconductor surface can generate oxidation-reduction reactions with the adsorbed species [16]. The Ti02 is highly available, innocuous, inert, economically and chemically stable, and requires UV radiation (wavelength around 340 nm) to generate hole electron pairs, which directly or indirectly are responsible for the oxidation-reduction degradation reactions [17].
In this research, the photocatalytic degradation of hydrocarbons present in synthetic water mixtures prepared in the laboratory was performed from a known crude oil and in samples collected from the skimmer inflow (production water).
Ti02 modified with iron (III) oxide is used in order to sensitize the Ti02 and thus use the visible radiation wavelength (400-700nm) to perform the degradation, considering its industrial application by using the solar radiation high exposure in oil exploration areas. The percentage of total hydrocarbon degradation (HTP's) was estimated by measuring the absorbency of the samples as a function of the degradation time: Photometric detection and contracted services for quantification of HTP's mg / I (total hydrocarbons of petroleum origin) using the gravimetric partition method in the Laboratories of Prodycon SA [18].
2. EXPERIMENTAL DEVELOPMENT
Semiconductor Ti02 Degussa P-25 modified with iron (III), by means of the cyclic chemisorption-calcination method with iron (III) acetylacetonate [19], a dry impregnation is carried out with the iron precursor that was supported on polymethyl methacrylate plates by an adhesion method [20], It is calcined for 2 hours at a temperature of 500SC. Then, the subsequent characterizations showed that the precursor decomposed and formed the iron oxide; regarding the X-ray diffraction characterization, it was carried out to ensure that the percentages of anatase and rutile corresponded to 80% and 20% respectively, according to the data reported in the technical data sheet of the Degussa P-25; in terms of morphological characterization, it was carried out in previous works by means of scanning electron microscopy (SEM) using the JEOL NeoScope JCM equipment 5000, observing parameters such as homogeneity, roughness and a particle size of radii similar to 25 nm and agglomerates of some particles ranging between 200 nm and 300 nm.
The adherence of the photocatalyst to the films was evaluated by mass difference, before and after immersing, the plates in water for 72 h and losses of less than 10% were obtained on average, a minimum percentage that allows the use and disposal of the plates in the reactors.
Three plates were arranged per reactor in parallel configuration, in cylindrical amber glass reactors with 1.0l and 15.0 ml capacity, and Lighting systems with the following characteristics were installed:
In addition to air supply with an electric air pump, with 3 W power and 3L / min 20 kPa flow. AC Power 110 V / 50 Hz and dimensions: 7.5 x 4.6 x 3.7 cm, necessary for the photocatalytic process inside the reactors [17].
Synthetic water mixtures were prepared using API crude: 23.5 (campomax) and distilled water. The final concentrations are observed in Table 2
Table 2 Concentrations of synthetic samples (MSX) used in the evaluation of the total hydrocarbon degradation capacity of the reactors used.

The field sample was obtained from the inflow to a skimmer, from a plant located in the eastern plains region of Colombia. The initial concentration of total hydrocarbons in the sample was 145.9 mg / L, according to the TPH method (gravimetric partition) carried out by Prodycon SA.
The hydrocarbon degradation process was evaluated using two techniques. The turbidimetric technique was developed using a UV-vis spectrophotometer - Ocean Optics, at 400 nm (experimentally selected based on the R 2 factor, from a sweep of calibration curves from 350 to 650 nm every 50 nm). The total hydrocarbon analysis was carried out using the gravimetric partition technique, which consists of making successive extractions and the resulting organic fractions (containing the total hydrocarbons) are rigorously dried and their mass determined [21],[22].
The collection and preservation of water samples from the input flow to the skimmer required adjusting the pH of the samples with hydrochloric acid to pH below 2. All samples subjected to solvent extraction for gravimetric tests remained refrigerated between (04) ° C from the time of field collection. All samples were analyzed within 28 days after being collected, asset out in the procedure [23].
3. RESULTS
Figure 1 illustrates the hydrocarbon degradation experiments, present in the samples mentioned in Table 2, using a 15.0 ml_ reactor. The concentrations were determined by turbidimetry (wavelength = 400 nm). In general, it is observed that the photocatalytic activity is independent of the hydrocarbon concentration in the range of studied concentrations (200 - 1000 ppm). The degradation percentages specified in the figures correspond to a degradation time of 210 min. However, the concentration remains relatively constant after 100 min.

Figure 1 Hydrocarbon degradation curves in samples (a) MS2, (b) MS4 (c) MS6, (d) MS8, (e) MS10, using a 15 mL reactor. The degradation experiments were performed in triplicate for each initial concentration.
Thanks to previously developed investigations, it is known that the percentages presented of adsorption (before photocatalysis) in the absence of radiation can be considered negligible, since the curves remain stable and their change over time is not considerable.
Table 3 summarizes the percentages of average degradation and the variation coefficients for the different synthetic samples. Apparently, there is no correlation between the initial hydrocarbon concentration and the final degradation percentage.
Table 3 Degradation percentages and variation coefficient obtained from the experimental data corresponding to the degradation curves shown in Figure 1.
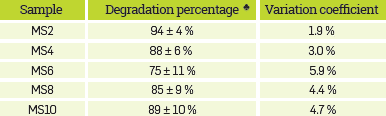
* The uncertainties reported were calculated based on statistical t-Student theory, with 95% confidence level [24].
Table 4 shows the results of the degradation process of the field sample (MC) obtained from the inflow to a skimmer of a plant Located in the eastern plains region of Colombia. The data obtained in degradation experiments are also shown for a 1: 2 dilution of the field sample (MCD) and a concentrated synthetic sample (MSC) The samples were subjected to the degradation process during (210 min), in the 1.0 L capacity photocatalytic reactor. The reported concentrations were obtained using the TPH method (gravimetric partition) by Prodycon S.A.
Table 4 Results of the degradation of hydrocarbons present in a field sample (MC), diluted at: 2 (MCD), and a concentrated synthetic sample (MSC).

The degradation percentages reported in Table 4 confirm that the photocatalytic processes proposed hereunder allow for a high yield in the reduction of hydrocarbon concentration in produced water.
These results open up the possibility of implementing this process at an industrial level, where production water represents is one of the main problems of oil production. By having the possibility of treating large volumes of water with a proportionally low cost method, operating costs would be reduced and this saving would translate into better economic profitability of the drilling and oil production project.
It is also worth pointing out that according to resolution 0631 of 2015 of the Ministry of Environment and Sustainable Development, the maximum permissible limit of TPH in production waters, to be discharged into surface water bodies is 10 mg /1. According to Table 4, the final concentration obtained in the field sample is 2.9 ± 0.5 mg /1 above the standard.
Bearing in mind that this research is a preliminary study on the photocatalytic removal of hydrocarbons with modified TI02, and the bibliography search carried out, no publications of this technological approach were found in the hydrocarbon industry, the results are promising and open the door to the treatment of this type of effluents nowadays.
Finally, Table 5 shows project costs:
It should be noted that the materials and reagents correspond to 39% of the total cost; as compared with the oil industry, hydrocarbon analysis and measurement techniques are either well known (61% of the costs of this project), implemented or part of routine activities, and are carried out independently of the treatments used for waste disposal. Even though the adaptation on an industrial scale of the system proposed in this paper depends on specific variables and conditions of the oil sector in particular, the implementation costs would include the same non-dimensional characteristics of the reactor described herein (See experimental session). It is worth highlighting that the photocatalyst base material (titanium dioxide) is economical and abundant and visible solar radiation can be obtained, without need for installation of lighting circuits. Hence, photocatalysis for the treatment of water in the oil industry is a potentially promising and economic technology.
CONCLUSIONS
The photocatalytic degradation of hydrocarbons in synthetic and field samples of production waters was studied Based on the results obtained at lab level, it was concluded that it is feasible to treat production waters to degrade present hydrocarbons, reaching degradation percentages above 91% and validating the experimental methodology. These results enabled us to conclude that the exposed treatment method is a modern and feasible alternative for the reduction of costs associated with the treatment and its application in the industry.
Photocatalytic degradation is a technique that, in addition to other factors, involves the use of artificial radiation sources to provide the necessary energy and thus enable degradation By sensitizing of Ti02 with iron oxide (III), these degradations are achieved using radiation in the visible spectrum, allowing for use of natural radiation and avoiding additional facilities to obtain the necessary energy. The intent is, therefore, to take advantage of solar radiation in oil fields for subsequent industrial implementation.
Evidence was obtained in this work of the removal capacity through sensitization of TiO2 and guidelines are ready for determining in subsequent studies the increase in efficiency Vs the amount of precursor in the cyclic chemisorption-calcination technique that was used.
Photocatalytic degradation methods have proven to be a real alternative for the treatment of production water by improving its physicochemical conditions, considering their subsequent discharge/ reinjection/disposal. This study makes it possible to propose an alternative treatment method to the oil industry, which must be continually enhanced due to its being constantly challenged by the environmental impacts it generates.

















