Introduction
Aeration processes are a key factor for the treatment and restoration of wastewater, favoring bacterial growth, mineralization, and the removal of most contaminants present in wastewater [1]. These processes have been widely used for the transfer of dissolved oxygen (DO) in water. However, this phenomenon is subject to climatic [2,3], microbial [4], and physical changes [5,6]. It is also dependent on the presence of oxygen in the air hindering the transfer of oxygen present between the air and bodies of water [5,7].
Due to the need to restore normal DO concentrations in water, various aeration mechanisms have been developed. These systems normally have an energy consumption of 0.3 and 1.2 KgO2·kWh-1 in diffuse aeration systems and between 0.5 and 2.1 KgO2·kWh-1 in mechanical aeration devices [8]. Bubble diffusers have efficiency in oxygen transfer of 2 to 7 % per meter depending on the depth of the storage tank; however, the usage of mechanical aeration systems in several processes is preferred due to their higher efficiency [9].
Mechanisms such as centrifugal water agitators similar to those proposed by Inaba [10], have an operating cost 10 times lower than the paddle wheel aerators traditionally used in aquaculture [2]. Authors such as Brown & Trucker [11], show that the aeration efficiency by paddle wheel devices is directly related to the increase on the rotation speed and length of the paddles, generating an increase in costs and power required. Low RPMs in paddle aerators favor the volumetric transport of water when compared to conventional methods and equipment [12,13]. At high RPMs, there is a lower oxygen transference due to the volumes of air used and higher energy costs. This significantly impacts the efficiency and viability of this type of mechanism in remote environments. These agitation speeds also generate harmful disturbances [14,15], and increase the level of stress in ecosystems.
The purpose of this study seeks to measure the efficiency of DO transfer in shadow water simples concerning energy consumption of a rotating agitation system by varying the power supplied and the number of paddles within the device. Anoxic water was used as a standard while measuring the time elapsed and the electrical consumption required during the aeration process. Measurements of the concentration of oxygen in the water were taken and the data was analyzed to determine the efficiency and performance of the different variables.
Materials and experimental details
Setup
Measurements were made using 44 L simples of clean water without biological and particulate material. The water sample was placed in a container with a contact area of 0.1976 m2 with air and a depth of 0.25 m. The elimination of DO in the water was carried out by the addition of sodium sulfite (Na2SO3) with an excess of 0.3 % and 0.1 g·m-3 cobalt chloride as a catalyst [16,17]. The concentration of reagents used was established as follows:
The initial content of O2 in the water was measured by oximeter Hach-Flexi HQ30d US Pat. 6912050. The addition of reagents was carried out in one liter of water taken from the sample to ensure that it completely dissolved in the solution. It was then poured into the storage container and stirred for 10 minutes. The concentration of dissolved oxygen in the prepared sample was measured by an oximeter.
The application of sulfite in water favors the formation of sulfate ions (equation 3), resulting in a loss of oxygen from the water. The application of CoCl allows for the introduction of cobalt as a catalyst that accelerates the reaction kinetics. However, the presence of sulfate ions in the wáter favors the transfer of DO. As the concentration of dissolved sulfite ions increases, there is an increase in the retention of air for the transfer of DO due to its influence on the decrease in bubble sizes and generation of non-coalescence effects within the fluid [18].
Experimental process
Tests were performed using paddle wheel aerators with 6, 12, and 24 paddles, each paddle with a surface area of 0.009 m2. The design of the blades was developed based on what was reported by anco-Zuñiga et al. [19]. The application of a flat blade system was selected, which allows obtaining a higher DO transfer rate with energy consumption. Other studies have shown that the application of a low number of paddles, with flat or triangular morphology, presents optimal results on the efficiency of DO transfer [20]. The device was started by using a 0.8 W reduction geared motor and varying the voltage between 6, 9, and 12 V to alter the RPMs. The measurements were made at room temperature (19.4 °C), the atmospheric pressure of (740 hPa), an altitude of 2800 m.a.s.l., and a constant water volume of 44 L. Each combination of variables was repeated three times. DO transfer measurements in water were made by oximeters. Each one was placed inside and on opposite sides of the container, Figure 1. Measurements were made in 10-minute intervals until the dissolved oxygen measurements stabilized. These measurements will allow observing the behavior in the transfer of DO in the water samples from the following equation:
Where:
DOt = is the DO concentration measured at time t
DOsat = the saturation concentration of DO in water.
To determine the saturation concentration of DO in the water samples in this study, it was determined from the measurement of DO in samples of clean, unaltered water, i.e. before the removal of DO by the addition of sodium sulfite. Within this study, it was observed that the DO saturation value was 7.5 mg·L-1.
Nominal transfer capacity
Data obtained in the process of DO transfer for each of the measurements allowed the determination of energy consumption and the efficiency of the aeration process using the calculations reported in other studies [8,19,21].
The efficiency of the aeration process was performed using a temperature correction factor on the transfer coefficient KLa. The transfer can be expressed as follows:
Where:
C = Average effective concentration of DO in the liquid phase.
C*͚ = Average final DO concentration achieved in infinite time (mg·L-1).
C0= Initial DO concentration, when the temperature.
KLᵃ= Volumetric mass transfer coefficient (°C-1).
Taking into account that the temperature varies during the experimentation process, the volumetric mass transfer coefficient KLa is corrected on a standard temperature (T= 20 °C) using the following equation:
Standard Oxygen Transfer Rate (SOTR) shows the amount of DO that has been transferred in the system per unit of time (kgO2·h-1), which is calculated as follows:
Where:
KLa20 = Volumetric mass transfer coefficient (°C-1), at 20 °C.
C S,20 = Saturation concentration for a steady-state at 20 °C.
Vol = Volume of liquid.
Finally, the standard aeration efficiency (N) is expressed as the oxygen transfer per unit of power, as indicated in the following equation:
Where:
SOTR = Standard oxygen transfer rate [kgO2·h-1].
P = Consumed potence [kW].
Results and discussion
The oxygen concentration of water
Figure 2, shows the percentage of DO transfer in the water by applying a rotational aerator of a) 6, b) 12 and, c) 24-paddle wheel. The results show that the application of a low voltage decreases the speed of rotation in each of the devices, decreasing the percentage of DO transfer in the water and resulting in a lower efficiency [20]. Likewise, the number of paddles and distance between them influence the transfer of DO depending on the time required to obtain the total oxygen transfer in the system.
Figure 2-c shows an oxygen saturation of 90 % for a device with 24 paddles obtained after 100 minutes when applying 12V. When applying 6V and 9V, this saturation occurred after 240 and 160 minutes, respectively. Greater efficiency was obtained with the application of a 24-paddle Wheel aerator with the application of 12V. The decrease in the number of paddle wheels inside the device generates an increase in the time required to reach the saturation point of the DO in the water simples (90 %). Also, Figures 2-a and 2-b show that the reduction of the applied voltage decreases the rotation speed of the paddle-wheels, making the DO transfer process less efficient in terms of time, obtaining a worse behavior during the transfer of DO the device of a 6-paddle wheel with an application of 6V. The behavior in the contribution of DO to the System reflects an Arrhenius-type curve, which depends on the difference between the partial pressure of DO in water and atmospheric oxygen [22,23]. This phenomenon causes the DO content supplied to the water body to decrease as the value approaches the DO saturation value in the system, requiring a greater amount of time to provide a lesser amount of oxygen to the wáter body [19,24].
Dissolved oxygen transfer
Figure 3, shows the percentage of oxygen transfer concerning. The measurements demonstrated stagnation in the DO transfer process during the first 50 minutes in the samples worked at 6V and 9V. This is then followed by an exponential increase in the DO transfer process until a saturation point is achieved, then immediately begins to decrease. This process is directly associated with the speed of rotation of the aerator, allowing an increase in the transfer of DO as the number of rotations of the device increases, thus reducing the time needed for the transfer of oxygen in the water.
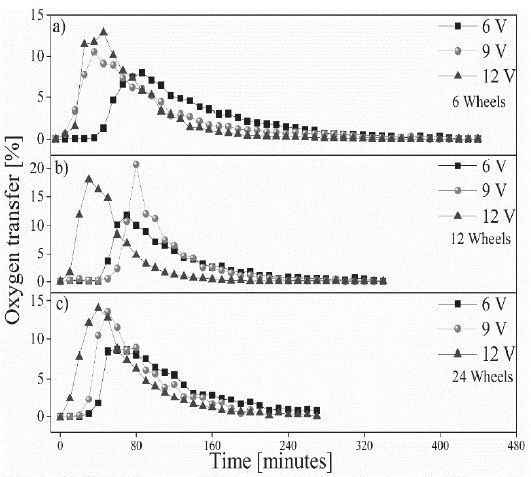
Figure 3 Percentage of oxygen transfer as a function of time for a) 6 wheels, b) 12 wheels and c) 24 wheels.
Similarly, the optimal distance between paddles allows a greater transfer of DO in the system. The results obtained within this study demonstrate a higher DO transfer rate value for the 12-paddle wheel system (Figure 3-b), which allows more space for a greater transfer of DO into the water. The reduction in the separation between paddles as shown in the device with 24 paddles (Figure 3-c), reduces the uptake of oxygen between each of the paddles, decreasing the transfer of DO depending on runtime. Likewise, the excessive separation between paddles prevents the introduction of a greater quantity of oxygen from the atmosphere as can be observed in the system with 6 paddles (Figure 3-a).
Paddles that were submerged during the aeration process yielded an increase in the standard oxygen transfer rate (SOTR) in the water. This also provided general agitation of the tank allowing an increase of the DO of the water [8,19]. This allows the utilization of low RPMs for the paddles decreasing the degree of disturbances that are normally generated by aeration devices and their impact on aquatic ecosystems [15,25]. This presents a possible application in aquatic ecosystems that present low DO concentrations. Figure 4 shows a decrease in the standard oxygen transfer efficiency (SOTE) when very low RPM is applied. As the speed of rotation increases, the amount of DO in the water and the SOTE increase directly within a conventional aeration system [26]. However, the results obtained by using a 12-paddle wheel aerator at 6V, show a reduction in DO transfer speed due to the low RPM. Thus, making it difficult to maintain a good transfer of oxygen from the atmosphere to the water inside the tank. A low level of water agitation, caused by the movement of the paddles inside the rotational aeration device, delays the transfer of DO as time passes [27]. Figure 4, shows an increase in the oxygen transfer rate as this value approaches the saturation point of DO in the water, mainly for the systems running at high RPM (applying 12 V). However, once the system gets close to the saturation point it becomes more difficult to introduce oxygen into the water regardless of RPM [28].
The efficiency of the process
Table 1 shows the nominal transfer capacity of the system (N), expressed in kgO2/kWh. The tests carried out for each of the conditions continued until the saturation point of DO in the water was reached. The time required to reach this saturation point depended mainly on the rotational speed of the wheel. The values obtained show greater efficiency for devices that ran at lower RPMs. This allowed for a lower energy consumption expressed as a function of power and the DO transfer obtained.
Table 1 Nominal transfer capacity of the system (N).
| Wheels | Voltage [V] | RPMs [a.u.] | N [kgO2/kWh] | S.D. |
|---|---|---|---|---|
| 6 | 5.5 | 2.88622 | 0.13943 | |
| 6 | 9 | 8.16 | 2.40631 | 0.12032 |
| 12 | 10.93 | 1.48427 | 0.07421 | |
| 6 | 6.12 | 2.14457 | 0.12722 | |
| 12 | 9 | 7.8 | 1.79911 | 0.119 |
| 12 | 10.5 | 1.07103 | 0.12536 | |
| 6 | 4.97 | 1.42939 | 0.12961 | |
| 24 | 9 | 7.39 | 1.13136 | 0.11566 |
| 12 | 9.2 | 0.57176 | 0.11786 |
Figure 5, shows the energy efficiency of each of the devices at different RPMs. A greater DO transfer capacity can be observed for the 6-paddle wheel aerator at 6V (N = 4.19 KgO2·kWh-1). An increase in paddles inside the device requires an increase in the power required for its operation, thus reducing its efficiency [29]. Likewise, higher RPMs also require an increase in the power supply, thus reducing efficiency. The data shows a direct relationship between the number of paddles and RPMs and the energy efficiency of the process. Although the transfer of DO as a function of time is more efficient for the 12-paddle wheel aeration system, at 12V, its efficiency based on the energy required during the process is less than the 6-paddle wheel aerator device at 6V.
Figure 6, shows the physical behavior of the 6-paddle wheel system at different RPMs. The images allow to observation of the generation of bubbles by the paddles at relatively high RPMs applying different voltages. Figure 6-a shows a high amount of bubbles due to a high rotation of the paddle-wheels, using 12V of power, while Figures 6-b and 6-c show the performance of the system with the application of 9 and 12V, respectively. This creates disturbances in the water caused by the formation of a turbulent flow within the system [30]. As the number of RPMs in the system decreases, the speed of rotation of the wheel decreases (Figure 6-b), reducing the quantity and size of the bubbles generated which fail to form at low speeds, Figure 6-a. The degree of disturbance of the water present in the system is reduced, this being favorable for bodies of water destined for aquaculture [14]. Also, Figure 5 shows that an increase in RPMs generates an increase in energy consumption. This application of high-speed devices and a large number of paddles reduces their efficiency in terms of power supplied. The application of systems that work at high rotation speeds produces a decline in the efficiency of the process, generating movements such as spills and splashes, which are not very efficient in DO transfer processes [31].
Conclusions
The influence between the rotational speed and the number of paddle wheels of an aeration device on the transfer of DO in shadow water samples is analyzed in this study. Results indicated a better performance for a 12-paddle wheel aerator when applying 12 V, allowing for a greater transfer of oxygen into the water in a shorter period of time. However, energy consumption data showed a greater efficiency for an aeration device with a low number of paddle wheels, using in this study a 6-paddle wheel aerator at 6 V, presenting greater energy efficiency according to the power supplied and the percentage of oxygen transfer.



























