Introduction
Pyloric syndrome or obstruction of the gastric outlet tract is an entity that can occur secondary to neoplastic involvement and adversely affect patients’ quality of life1,2. Metastatic involvement as an etiology is unusual and even less frequently associated with a primary breast tumor3,4, which makes it reasonable that in cases like the one described, various differential diagnoses should be suspected and explored.
Clinical case
A 69-year-old woman was diagnosed with infiltrating lobular carcinoma of the right breast one month before admission. She was admitted for a week of diffuse abdominal pain associated with nausea, postprandial emesis, and dyspnea at rest. On admission, she was found to be in a fair general condition, tachycardic and tachypneic, with ambient saturation of 57%, and she had no other relevant history. Management was given with supplemental oxygen, and a study for dyspnea and emetic syndrome was initiated. Contrast-enhanced CT studies of the chest and abdomen were performed, and the results were negative for pulmonary thromboembolism, with findings of basal subsegmental atelectasis and pericardial effusion without hemodynamic compromise. Additionally, bilateral breast neoplastic involvement and dilation of the gastric chamber with thickening of the pylorus were evident (Table 1).
Table 1 Paraclinical studies
| Study | Result |
|---|---|
| Transthoracic echocardiogram | Left ventricle of average size, concentric remodeling, preserved systolic function, EF of 60% without segmental disorders; moderate pericardial effusion with left pleural effusion and signs of collapsed or infiltrated lung segments; no significant valvular heart disease |
| Contrast-enhanced CT of the chest | Bilateral breast masses predominantly on the left concerning a known neoplastic history, with suspicious-looking lymph nodes in both axillary regions and mixed polyostotic involvement attributed to a secondary neoplastic disease; subsegmental atelectasis in the middle lobe and lingula |
| Contrast-enhanced CT of the abdomen | Moderate hepatic steatosis; dilation of the gastric chamber with apparent thickening of the pylorus walls; uncomplicated diverticular disease of the colon; polyostotic involvement concerning secondary neoplastic disease |
| Breast biopsy | Classic invasive lobular carcinoma |
EF: ejection fraction. Prepared by the authors.
With these findings, the hypoxemia was attributed to atelectatic involvement. Regarding the gastrointestinal condition, it was considered compatible with a pyloric syndrome, and management was initiated with a nasogastric tube and antiemetic, resulting in partial improvement. The patient was taken for esophagogastroduodenoscopy, which showed changes in esophagitis due to food retention and content in the gastric chamber, which, in turn, revealed an infiltrative-looking lesion with an ulcerated surface and easy bleeding at the postpyloric level that circumferentially compromised the duodenal lumen (Figure 1).
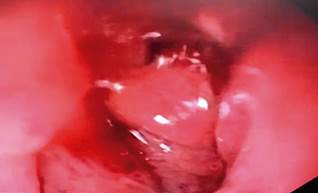
Source: Authors’ archive.
Figure 1 Prepyloric region with edematous, friable mucosa easily bleeding upon passage of the equipment with circumferential involvement.
Biopsies were taken from the lesion in the duodenum, which reported involvement by a group of atypical and discohesive cells with immunohistochemistry compatible with a lobular-type carcinoma of breast origin (negative for cytokeratin 20, CDX-2, and E-cadherin) (Figure 2). The findings were explained; however, the patient expressed advance directives not to receive invasive procedures, for which a 22 mm × 60 mm uncovered metal prosthesis was inserted to reestablish the feeding route and improve dyspnea and abdominal pain (Figure 3).
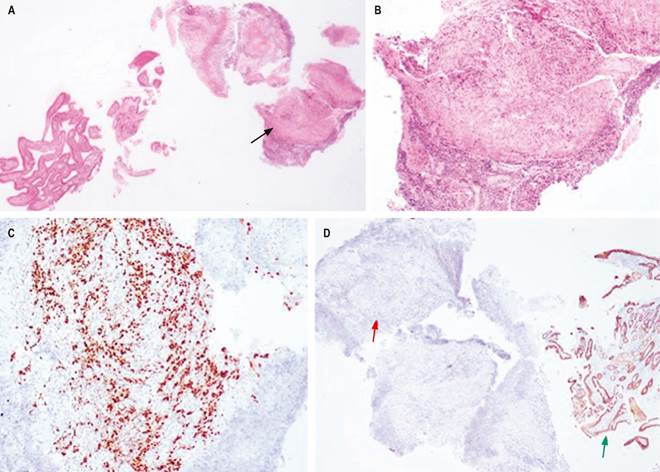
Source: Authors’ archive.
Figure 2 A. (Hematoxylin-eosin). Duodenal epithelium with preserved architecture and fragments of submucosa compromised by a tumor infiltrate (black arrow). B. (Hematoxylin-eosin). The tumor cells are discohesive, small to medium in size, with nuclear pleomorphism and hyperchromasia. C. (GATA-3). Positivity of tumor cells. D. (E-cadherin). Tumor cells are negative for E-cadherin (red arrow); there is adequate internal control in the intestinal epithelium (green arrow).
Discussion
Obstruction of the gastric outlet tract can be secondary to intrinsic or extrinsic causes that explain the mechanical blockage of gastric emptying; it can be located at the distal level of the stomach, pylorus, or duodenum and usually presents with nausea, vomiting, and abdominal pain1.
The etiology is usually divided into malignant (62%) and benign (38%), including gastric neoplasms, duodenal adenocarcinoma, carcinomas of the ampulla of Vater, neoplasms of the gallbladder, lymphomas, and metastases, among the malignant causes1,2. Benign etiologies include peptic ulcers, acute and chronic pancreatitis, inflammatory entities (such as Crohn’s disease), infiltrative entities (such as amyloidosis), and foreign bodies2-5.
Regarding infiltrating lobular carcinoma of the breast, it is characterized by discohesive cells and mutations in genes that code for E-cadherin since its absence supports the diagnosis6-8. However, there seems to be no association between this characteristic and the appearance of metastasis7,9. Multiple metastatic involvements of the bone, liver, and lung have been described but are rare in the peritoneum (14.6%) and the stomach (2.8%)10.
In the case of the patient, duodenal metastatic involvement due to already-known breast carcinoma was documented. While metastases are not frequent within tumor lesions of the duodenum, there are case series that describe prevalences of 32%, of which up to 12% are metastases from squamous cell carcinoma, clear cell carcinoma (8%), melanoma (8%), and pleomorphic liposarcoma (4%)3,11. Yim et al., in a series of cases of patients with pyloric syndrome, reported a metastatic etiology close to 27.6%12. Unusual manifestations also include metastatic involvement due to adenocarcinoma of the cecum, squamous cell carcinoma of the cervix and anus12-14, and pleomorphic lung carcinoma15,16. Regarding duodenal involvement due to breast carcinomas, episodes of gastric outlet obstruction secondary to or as the first manifestation of neoplasia are described, such as that reported by Khairy et al.17.
Among the therapeutic alternatives for outlet obstructions of benign etiology, pharmacological therapy and some endoscopic procedures, such as balloon dilations, are listed, leaving surgical management as the last option. Regarding the management of malignant obstructions, the insertion of antroduodenal prostheses, gastrojejunostomies (GJ), and endoscopic ultrasound-guided gastroenterostomy (EUS-GE) are described2,17-20.
Antroduodenal prostheses are metallic, whether or not covered by plastic or silicone membranes. The covered ones have a higher risk of migration but a lower stricture rate. The leading cause of stricture is intra-stent tumor growth (8-25.4%), and argon plasma therapy or a new intraluminal stent is required. Prosthesis migration occurs in 0% to 19.4%, mainly in covered ones; they are rearranged or removed endoscopically or surgically. Bleeding associated with the insertion occurs in 1%, and conservative or endoscopic management is usually given. They have a limited useful life and an in situ duration of up to 85 days. They are generally employed in patients with a poor short-term prognosis to improve quality of life and bring the oral route back, which allows longer relief of symptoms and lower frequency of reintervention and costs12,20.
GJ can be performed using the conventional approach or by laparoscopy in those patients with a life expectancy greater than or equal to two months19,20.
EUS-GE is a safe and effective alternative, which uses metallic prostheses that oppose the lumen covered with a device that prevents migration. The insertion is carried out through the stomach, communicating with the distal duodenum or the proximal jejunum; a technical success of 87% to 96% and clinical success of 81% to 92% have been reported. Complications include pneumoperitoneum, gastric fistula, bleeding, peritonitis, or abdominal pain, which are usually infrequent. When comparing this technique with antroduodenal prostheses, both have similar technical success, hospital stay, and safety19,20.
GJ has better technical success with symptom recurrence at a frequency similar to the endoscopic approach20. EUS-GE has a higher clinical success rate with greater obstruction and is a minimally invasive alternative with technical difficulty and less availability, which favors the other options19,20.
In the case of our patient, who expressed her advance directive to refuse invasive maneuvers and, taking into account her clinical condition, the insertion of an uncovered duodenal prosthesis was considered as management.
Conclusion
The pyloric syndrome is a widely known entity with a significant percentage of malignant etiology. It can be suspected in patients like the one mentioned, taking into account their advanced age and previously known neoplastic involvement. Although metastatic involvement of breast neoplasms to the digestive tract is uncommon, and even less so for lobular carcinoma, immunohistochemical markers can help establish an etiological diagnosis.
Management will depend on the patient’s context and clinical condition. Performing a GJ could be considered in patients with adequate functionality and medium-term prognosis. In those with an ominous prognosis, an enteral prosthesis could be inserted to improve the patient’s quality of life.











 texto en
texto en 




