Cocoa tree is cultivated in tropical countries with hot and humid environments. Three countries, Cote D’Ivoire, Ghana and Indonesia together cultivate 61% and produce 67% of global traded cocoa (Campos-Vega et al., 2018), considering the top ten countries account for approximately 93% of total world cocoa production (Table 1). The average yield in countries from Latin America and the Caribbean is 0.39 t ha-1 of cocoa and in the world is 0.44 t ha-1 of cocoa (Sánchez et al., 2019). The main varieties of cocoa produced and sold in Ecuador are cacao fino y de aroma (fine aroma cocoa) (Clones Nacionales, Pichilingue Tropical Experimental Station, Ecuador) and CCN-51 cocoa (Castro Naranjal Collection, Guerrero, 2015). Ecuador is an important producer of fine aroma cocoa worldwide and is currently recognized for providing more than 60% of its production. Fine aroma cocoa is recognized worldwide for its aroma, color and quality. The use of the fruit seed to produce chocolate, cosmetics, among others, triggers the generation of by-products. The agroindustry of cocoa generates between 17 and 20 kg of by-products for each 100 kg of cocoa fruit (López, 2014), which currently are used in soil fertilization (Munongo et al., 2017). Nonetheless, several phenolic compounds were identified by LC-MS/MS in cocoa pod-husk by Karim et al. (2014) such as phenolic acids, flavonoids, luteolin, apigenin and linarin. Despite cocoa pod-husk is an important source of phenolic compounds (Hii et al., 2009), with great potential for application in the field of nutrition, health and medicine, due to its antioxidant and antimicrobial characteristics (Niemenak et al., 2006), in many cases the by-products of cocoa are accumulated without any treatment, leading to the generation and transmission of diseases that may affect crops (FEDECACAO, 2013).
The fresh cheese is an Ecuadorian artisanal cheese with 37% moisture (Espinosa, 2012) and good acceptance among consumers, especially in the province of Manabí (Ecuadorian coast). The presence of pathogenic microorganisms such as Salmonella in this kind of cheese (Zambrano, 2014) together with commercialization of the cheese at inappropriate temperatures can affect the health of the consumer (Lee et al., 2015) and contribute to a high number of people suffering from Salmonellosis in the province of Manabí, Ecuador (Ministerio de Salud Pública, 2014, 2015, 2016, 2017, 2018).
Salmonella and other pathogenic microorganisms have been found in cheese of countries such as Colombia (Instituto Nacional de Salud, 2011), Turkey (Kahraman et al., 2010), France (Domínguez et al., 2009), Germany (Fretz et al., 2010) and Egypt (El-Baz et al., 2017). To deal pathogen microorganisms, natural antimicrobials can be an important alternative to preserve the microbiological quality of food and guarantee consumer safety (Abdalla et al., 2007).
Concerns about the use of synthetic chemical antimicrobials have renewed the interests of consumers using natural and safe alternatives (Nazzaro et al., 2009). The antimicrobial activity of phenolic compounds is well documented by several authors (Erdemoglu et al., 2007; Xia et al., 2011). The growth of strains of Salmonella and Escherichia coli is inhibited by phenolic compounds extracted from vegetables, fruits, herbs and spices (Cetin-Karaca and Newman, 2015), cocoa bean (Todorovic et al., 2017) and bean shells (Nsor-Atindana et al., 2012). However, no studies of the use of phenolic compounds of cocoa pod-husk against Salmonella growth have been performed.
In the present work, the inhibition of Salmonella spp. in fresh cheese was studied, after immersing the cheese in a solution of phenolic compounds previously extracted from the pericarp of fine aroma cocoa. Additionally, sensory analyzes were used to examine whether cheese treated with phenolic compounds showed organoleptic differences compared to an untreated cheese.
MATERIALS AND METHODS
Samples of fine aroma cocoa pericarp (Theobroma cacao L.) were collected at the Alegría farm, located in Junín, Manabí province, Ecuador, with 0.9277 south latitude and 80.2058 western longitude. The samples were washed with water and dried in an oven at 50 °C, ground and stored for future analyzes. Fresh cheese was purchased at the Nuevo Tarqui shopping center in the city of Manta, Ecuador.
Alcoholic extraction of phenolic compounds
The extraction of phenolic compounds was done according to Santacruz et al. (2020). An amount of 15 g of the previously ground cocoa pericarp was mixed with 150 mL of ethanol (95% v/v) and kept under stirring at 130 rpm for 24 h at 25 °C. Afterwards, the sample was centrifuged (SIGMA 2-16P, Germany) for 10 min at 4000 rpm and the supernatant was filtered by vacuum, saving the filtered liquid fraction.
Quantification of phenolic compounds
The quantification of phenolic compounds was done according to the Folin-Ciocalteu method proposed by Mahmood et al. (2011). A stock solution was prepared by mixing 10 mL of the previous filtered fraction with 5 mL of ethanol (95% v/v) and distilled water to complete a total volume of 100 mL. From the stock solution, 0.1 mL was mixed with 0.5 mL of the Folin-Ciocalteau reagent, allowing it to stand for 5 min. Afterwards, 1 mL of a sodium carbonate solution (5%) was added and made up to 25 mL with distilled water. The resulting solution was left in dark for 1 h and its absorbance at 760 nm was measured (Spectrophotometer Jenway 6320D, China). The quantification of phenolic compounds was performed using a calibration curve with gallic acid standards, and the results were expressed in mg of gallic acid equivalent (GAE) g-1 cocoa pericarp.
Inhibition of Salmonella spp. growing by phenolic compounds
The antibacterial activity of phenolic compounds against Salmonella spp. was performed according to Santacruz and Castro (2018) and the Clinical and Laboratory Standards Institute (CLSI, 2009). Salmonella spp. was inoculated into Petri dishes using Salmonella-Shigella agar (HiMedia Laboratories, India) as culture medium and incubated at 37 °C for 24 h. Filter paper disks (Fisher Scientific Q2) of 5 mm diameter were immersed in a solution of phenolic compounds at a specific concentration (see experimental design). The disks were dried and placed in the center of the Petri dishes containing Salmonella spp. and incubated at 37 °C for 24 h. Afterwards, the zone of inhibition of the growth by phenolic compounds was measured in triplicate.
Bacterial growth curve measurement
Bacterial growth was performed according to Puupponen et al. (2001). 10 mL of fresh growth medium (Salmonella- Shigella agar) were inoculated with 5% of culture (frozen Salmonella spp. was transferred to liquid media and incubated for 24 h). Phenolic compounds, from alcoholic extraction, were added to the culture media to give final concentrations of 0.5, 1 or 5%. The culture was manually shaken, and afterwards, it was incubated at 37 °C for 24 h. Bacterial growth was followed by taking seven samples from the culture during the incubation period of 24 h (0, 0.5, 1, 2, 4, 6, and 24 h). The samples were plated in Petri dishes containing Salmonella-Shigella agar and incubated at 37 °C for 24 h before bacterial count was recorded.
Bacterial growth in cheese
Fresh cheese was immersed in a 1% phenolic compounds solution for 15 min. Afterwards, cheese samples were stored for 8 days, overnight under refrigeration conditions and during the daytime at room temperature (approximately 25 °C).
Microbiological analysis of Salmonella in cheese was performed on samples after 0, 2, 4, 6 and 8 days of storage, according to ISO 6579: 2002 method (ISO, 2002). For the test, 10 g of cheese were weighed and placed in a sterile resealable plastic bag, before adding 90 mL of sterile peptone water. The sample was homogenized manually, and 1 mL of the liquid was transferred to a glass test tube before adding 9 mL of peptone water (10-1 dilution). The process was repeated to get a dilution of 10-4, and immediately 1 mL of the solution was inoculated into Petri dishes and incubated at 37 °C for 24 h prior CFU counting.
Sensory analyzes
Cheese samples were previously analyzed to verify the absence of Salmonella. Afterwards, the cheese was immersed for 15 min in a 1% phenolic compounds solution before sensory analyzes. First, a duo-trio test ISO 10399: 2004 (ISO, 2004) with 16 semi-trained panelists was done to determine organoleptic differences between cheese samples treated with phenolic compounds and a control sample (cheese with no immersion). Subsequently, attributes of firmness, color, odor and flavor were evaluated using a five-point hedonic scale, with scores ranging from "I like it a lot" (5) to "I dislike it a lot" (1).
Texture analysis
Puncture tests were performed according to Castro et al. (2017), using cheese cubes of edge 2 cm, with slight modifications. A Shimadzu texturometer (Model EZ-LX, Japan) together with a stainless-steel probe of 3 mm diameter and 8 cm length were utilized. The probe speed was 10 mm s-1 and the maximum penetration force was recorded. Results were the average of three measurements.
Experimental design and statistical analyzes
A completely randomized design with a unifactorial arrangement was used. The concentration of phenolic compounds obtained by alcoholic extraction at 3 levels (0.5, 1 and 1.5%) was established as the independent variable (Medrano, 2019), whereas the zone of inhibition and the bacterial growth measurement were the dependent variables. The concentration of phenolic compounds with the best Salmonella inhibition was applied to cheese samples, before sensory analysis.
ANOVA and the significance of the difference between means was determined by Tukey test (P<0.05) using InfoStat statistics software (Infostat version 2014, Argentina). All measurements were performed in triplicate.
RESULTS AND DISCUSSION
The results of the inhibition of the growth of Salmonella spp. by phenolic compounds showed that there was a difference between Salmonella inhibition zones at the three concentrations of phenolic compounds (P<0.05) with values between 7.78 and 11.78 mm (Table 2). A positive correlation was also found between the inhibitory effect and the concentration of phenolic compounds by Hernández et al. (2012). The results of the present study agree with those obtained by Nsor-Atindana et al. (2012) for an ethanolic extract of cocoa pericarp at 100 mg mL-1 with zones of inhibition between 10.98 and 12.21 mm against Salmonella, E. coli, S. aureus and B. cereus. The inhibitory effect can be influenced by the type of phenolic compounds (Orihuela, 2016) that acts to a greater or lesser degree on the cell wall of bacteria, resulting in the loss of cell structure and the death of the microorganism. Another explanation for the inhibitory effect may be the diffusion of hydrophilic compounds through the channel proteins of the outer membrane that surrounds the cell wall of gram-negative bacteria (Santos et al., 2014) This cell wall may contribute to the entry of phenolic compounds, which may have resulted in the observed cell death.
Table 2 Inhibition of growth of Salmonella spp. by phenolic compounds extracted from the pericarp of cocoa.

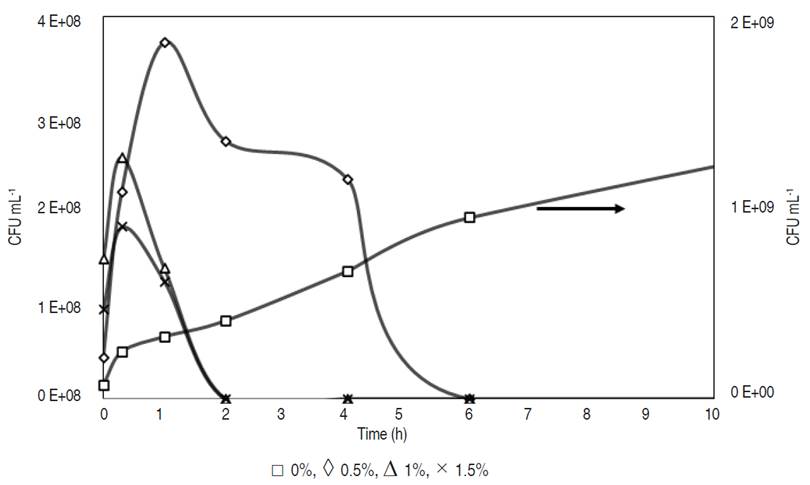
The measurement of bacterial growth curve (Figure 1) shows an increase in CFU mL-1 for the control sample along storage time. A complete inhibition of Salmonella spp. was observed after 2 h in the presence of phenolic compounds at 1 and 1.5%. The concentration of 0.5% showed a complete inhibition of Salmonella after 6 h. Phenolic compounds presented a dose-dependent antibacterial activity (Khan et al., 2021). Puupponen et al. (2001) showed that berry extracts at concentrations between 1 and 5 mg mL-1 were strong inhibitors of Salm. enteric. The inhibitory effect over time may be due to affectations of the bacteria cell wall because of the presence of the phenolic group (Puupponen et al., 2001). However, diffusion of phenolic compounds through the cell wall may allow an initial growth of bacteria (Figure 1) (Khan et al., 2021).
The control sample (cheese with no immersion) showed presence of Salmonella spp. According to Ecuadorian regulations, NTE INEN 1529-15: 2013 (INEN, 2013), Salmonella should not be present in cheese. Cheese samples previously immersed in phenolic compound solutions at 1% showed an absence of Salmonella at 0 day of storage. In fact, Figure 1 shows no presence of Salmonella in cheese treated with phenolic compounds at 0.5% after 2 h of storage.
Sensory analyzes
Results of the duo-trio test showed that 15 of 16 panelists did not find difference between cheese previously immersed in a solution of phenolic compounds and the control sample. Texture analysis displayed that penetration force of control sample was 1.02 N, which was lower than cheese treated with phenolic compounds, showing values between 1.92 and 1.99 N (P<0.05). Instrumental texture of cheese does not have a single dominant characteristic and exhibit at least two primary characteristics (e.g. chewiness and cohesiveness) (Meullenet et al., 1998).
Figure 2 shows the responses of the panelists to the five-point hedonic test about cheese treated with phenolic compounds. Firmness and odor obtained the highest ratings among the evaluated characteristics, since 56.3% of the panelists classified them as "I like it". For the flavor, 37.5% of the panelists gave a rating of "I like it" and an equal percentage for the color of the cheese with the label of "I neither like nor dislike". The lower color rating could be due to the fact that phenolic compounds developed a yellow color in cheese, color that did not change along storage.
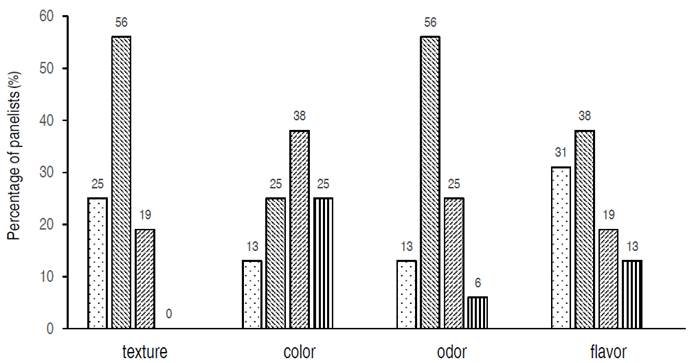
Figure 2 Sensory analyzes. Percentage of panelists and their evaluation of firmness, color, odor and flavor on cheese using a five-point hedonic scale, with scores ranging from "I like it a lot"  , "I like it"
, "I like it"  , "I neither like nor dislike"
, "I neither like nor dislike"  , "I dislike it"
, "I dislike it"  ,"I dislike it a lot"
,"I dislike it a lot"  .
.
Based on present results, it is difficult to conclude if color had influence either in odor or flavor. Moreover, visual and auditory appreciations can modify the flavor of food but are not intrinsic to it (Auvray and Spence, 2008). Koch and Koch (2008) stated that "In fact, it may be that color has nothing to do with the taste of food or drink". Meanwhile, Bayarri et al. (2001) suggested that "the possible influence of color on flavor perception is under discussion and no clear conclusions have been attained yet".
CONCLUSIONS
The solutions of phenolic compounds at concentrations of 1 and 1.5% showed the highest in vitro inhibition of Salmonella spp., requiring 2 h to achieve a complete inhibition of the bacteria.
Sensory analysis showed there was no difference between the cheese treated with phenolic compounds and a control cheese. Firmness and odor had high ratings whereas color had the lowest rating among the evaluated attributes.