INTRODUCTION
The coastal desert of the Peruvian and Chilean territory is a continuous, arid belt more than 3,500 km in length, from north to south, located on the western slopes of the Andes Mountains (Rundel et al., 1991). Throughout this region, different ecosystems are found, distributed as islands of abundant vegetation on hills and medium knolls exposed to winter fogs, known as coastal hills or "fog oases" (Dillon et al., 2003; Ferreyra, 1993; MINAM, 2019). Fog oases are xerophytic, seasonal vegetation formations rich in endemic plants, where herbaceous vegetation predominates, with an occasional tree found (MINAM, 2019). Fog oases are exclusive to South America, distributed from the Illescas peninsula in the Piura region, the Campana and Cabezón Hills in Trujillo, Peru (8 °S), to Huasco and Coquimbo, in Chile (30 °S), and are found between altitudes of zero and 1000 m.a.s.l. (Cuba-Melly and Odar, 2018; Sotomayor Melo and Jiménez Milón, 2008), mainly between 600 and 900 m.a.s.l. (Dillon et al., 2003; Ferreyra, 1993). In Peru, fog oases are distributed in the departments of Piura, Lambayeque, La Libertad, Ancash, Lima, Ica, Arequipa, Moquegua and Tacna (8 °S - 18 °S), covering an approximate area of 0.23% of the Peruvian territory (294,033.05 ha), one of the Peruvian ecosystems with the smallest area (MINAM, 2019).
Fog oases are formed by the influence of fogs coming from the South Pacific anticyclone that crosses the coastlines and intercept at the foothills of the Andes, especially on stony slopes and ravines oriented to the west (Ferreyra, 1993). Through this intercept, atmospheric water from mists condenses and precipitates, allowing vegetation to develop (Dillon et al., 2003; Muenchow et al., 2013a). This seasonal process typically occurs between the months of June and September (Ferreyra, 1986), when high levels of relative humidity are reached (>80%) (MINAM, 2019), along with greater plant diversity around rocky outcrops, including shrubs and trees (Lleellish et al., 2015). In contrast, between December and April, there is a decrease in atmospheric humidity, resulting in soils devoid of vegetation, except for some species of cacti, tillandsias, geophytes, xerophytic shrubs and trees that complete their flowering cycle in summer (MINAM, 2015).
The hilly formations of Chile and Peru have a high diversity of vascular plants compared to other cloud-dependent ecosystems in North America and southern Africa (Dillon et al., 2003; Schulz et al., 2011); they also have a high number of endemic genera and species of flora (Balaguer et al., 2011; Mostacero et al., 1996; Rundel et al., 1991). In Peru, these ecosystems have approximately 675 species of angiosperms, 30% of which are endemic (Moat et al., 2021). Among these endemics, species belonging to the genera Domeykoa (Apiaceae), Mathewsia (Brassicaceae), Nolana (Solanaceae), Palaua (Malvaceae) and Weberbauerella (Facabeae) are typical (Ferreyra, 1986). In the Mangomarca fog oasis, high levels of richness and endemism of vascular flora have been found, with 51 species, nine of which are endemic (Santa Cruz et al., 2020).
In fog oases, it is common to find maximum species richness in mid-elevation areas (Muenchow et al., 2013a; Rahbek, 2005), due to environmental conditions that allow for greater water availability (Borthagaray et al., 2010; Muenchow et al., 2013a), leading to the development of vascular plants (Krömer et al., 2006) and mosses (Ah-Peng et al., 2012). These zones of maximum richness are found at lower elevations and adjacent to the thermal inversion region. As one ascends altitudinally, the point of maximum fog condensation, and therefore the highest humidity, is reached in this region (Schemenauer and Cereceda, 1993). This region coincides with the average altitude of development of the hills in Peru, which is 500 m.a.s.l. (Moat et al., 2021). In Peru, studies analyzing species richness and composition along the altitudinal gradients of fog oases are scarce (Muenchow et al., 2013a, 2013b). However, there is only one previous study in the Mangomarca fog oasis, which evaluated the correlation between environmental variables such as slope, humidity and altitude, with species cover and richness (Padilla, 2018).
Fog oases are highly sensitive to climate change and anthropogenic impact, the latter which has drastically altered them throughout history (Beresford-Jones et al., 2015). Since colonial times, these ecosystems have been impacted by intensive cattle ranching activities (Galán de Mera et al., 2011; Muenchow et al., 2013a; Trinidad et al., 2012), and the introduction of European species (Galán de Mera et al., 2011), which locally extinguished species with lower adaptability to competition (Muenchow et al., 2013a). Five centuries ago, hillocks occupied more than 15,000 km2, when they supported important population centers (Engel, 1981). However, at present these plant communities cover only 6964 km2 in Peru and 1714 km2 in Chile (Moat et al., 2021). This dramatic decrease in their extent is primarily due to urban growth (Tovar et al., 2018), overgrazing, deforestation (Mostacero et al., 1996) and, to a lesser extent, mining (Miyasiro and Ortiz, 2016). On the other hand, land use change is the main threat to endemic species (Dillon et al., 2003; Whaley et al., 2010), as reported in two studies conducted in the Mangomarca fog oasis (Padilla, 2018; Santa Cruz et al., 2020).
Recent data estimate that 58% of Peru's population lives in coastal cities near these oases, where the highest levels of anthropogenic pressure are found in cities such as Lima, Chiclayo, Trujillo and Chimbote (Moat et al., 2021). Urban growth is a continuous threat to coastal hills near cities, which have been occupied by neighboring populations, causing accelerated reductions in their surface area (Pollack et al., 2020; Rodríguez, 2015; Santa Cruz et al., 2020; Trinidad et al., 2012). Although the greatest impact was evident in the 1980s, the anthropogenic threat continues to the present day (Pollack et al., 2020), affecting the extension, vegetation cover and floristic diversity of these ecosystems (Moat et al., 2021; Trinidad et al., 2012). Despite being classified as a fragile ecosystem (Tovar et al., 2018), scientists and authorities have paid little attention to the problem of fog oases (Pollack et al., 2020). Currently, only 3.3% of Peru's fog oases are conserved within a protected area (Moat et al., 2021).
Studies that relate variation in species richness and composition to environmental gradients are indispensable for understanding community ecology and biogeography (Macarthur, 1984; Muenchow et al., 2013a; Whittaker, 1967). This information can be used to help design conservation and management projects (Pomara et al., 2012). The Mangomarca fog oasis, and other hills of the Peruvian national territory, have been little studied in these aspects, and the notion of fog oases ecosystems as a homogeneous units predominates. Likewise, studies on floristic composition and its relationship with altitude continue to be scarce. These information gaps need to be filled to allow for the development of policies and plans aimed at the conservation of these ecosystems. Therefore, the objective of this study was to determine the floristic composition and richness of the vascular flora at three altitudinal ranges of the Mangomarca fog oasis during the period 2013 and 2014. The results show high levels of diversity and endemism in these hills. However, the altitude factor was not significant to determine that there are three different groups of plant communities, corresponding to the three altitudinal ranges.
METHODOLOGY
Study area
The study was carried out in the Mangomarca fog oasis (11° 58' 50" - 12° 01' 00 "S, 76° 59' 30" - 76° 57' 10" W), located on the western slopes of Balcón hill on both sides of the Mangomarca ravine in the district of San Juan de Lurigancho, department of Lima, Peru (Santa Cruz et al., 2020). The sampling area was located between 350 and 824 m.a.s.l., in the SW direction creek, located between the sectors of Los Sauces and Campoy, being accessible by the urbanization called Asociación de Propietarios de Vivienda de Mangomarca Alta (APTL) (Figure 1).
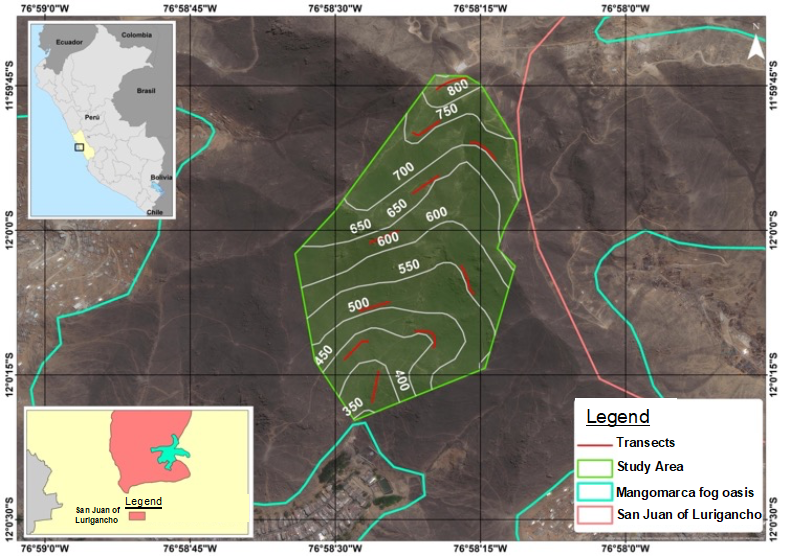
Figure 1 Study area in the Mangomarca fog oasis. The map shows both the stream where the transects were established and the contour lines in the study area, with their respective altitudes.
Sampling and characterization of altitudinal strata
Nineteen visits were made during the period from June 2013 to May 2014, carried out every 15 days during the wet season (June-December) and monthly during the dry season (January-May). Ten transects measuring 5 m × 200 m were installed, where species richness was evaluated. Three transects were installed in each altitudinal range: 350-500 m.a.s.l. (A1), 500-650 m.a.s.l. (A2) and 650-800 m.a.s.l. (A3). Additionally, a transect was installed at an altitude above 800 m.a.s.l., which was established in a zone of solid rock (Aguilar, 1976), which presents a plant community with a greater abundance of xerophytic and semi-xerophytic species (Aguilar, 1976; Brack and Mendiola, 2004). The transect established in the massive rock zone was excluded from the analysis of floristic composition and richness, in order not to alter the three replicates for each altitudinal range. However, this transect was considered within the A3 altitudinal range for the analyses of total richness of the hills, and of single and generalist species (Figure 1). Likewise, the delimitation of the altitudinal ranges was carried out by adapting the proposals of Ellenberg (1959) and Ono (1982) to the dimensions of the Mangomarca fog oasis.
Botanical collection and identification
The specimens were photographically registered, collected, and later identified by the Biologist Huber Sady Trinidad Patricio of the Museum of Natural History of the Universidad Nacional Mayor de San Marcos, an expert of the Herbarium San Marcos (USM), Biologist Rosmery Adela Robles León and Forestry Engineer Víctor Raúl Cornejo Badillo. Specialized taxonomic keys were used following the order of the "Angiosperm Phylogeny Group" update for the taxonomic classification of the orders and families of angiosperm species (APG, 2016). Likewise, the IPNI (2021) and "Plants of the World Online" (POWO, 2021) databases were reviewed to ensure that the scientific names of each species were current.
Endemism and conservation status
The Guide to the flora of the Lima Hills (Lleellish et al., 2015), the Plants of the World Online database (POWO, 2021), and the red book of endemic plants of Peru (León et al., 2006) were used to determine whether the species were endemic, native, or introduced. Threat categories were classified according to Supreme Decree N° 043-2006-AG of the Peruvian Ministry of Agriculture.
Data analysis
Multivariate analyses were carried out by means of non-metric multidimensional scaling analysis (NMDS) to determine the clustering of altitudinal ranges according to the floristic composition of species and families. The aforementioned analysis was performed with R software (R Core Team, 2020) and the 'factoextra' (Kassambara and Mundt, 2020), and 'vegan' (Oksanen et al., 2020) packages.
RESULTS
Fifty-three species in 43 genera and 26 families were recorded (appendices 1 and 2), including a single fern species, Adiantum subvolubile Mett. Ex Kuhn (Pteridaceae) and a single species of the Gymnospermae clade, Ephedra americana Humb. and Bonpl. ex Willd. (Ephedraceae). The two most diverse families were Asteraceae and Solanaceae, with eight species in each, distributed in seven and five genera, respectively. These were followed by Amaranthaceae (3/3), Malvaceae (3/1), and Oxalidaceae (3/1). There were seven families that presented two species, while the remaining 14 families were monospecific. The Eudycotiledoneae clade is the richest, with 45 species in 35 genera and 20 families, while the Monocotyledoneae are represented by six species, in six genera and four families.
In terms of growth form, we identified 36 herbaceous species (68%), seven shrub species (13%), five subshrub species (9%), two cacti (4%), both located only in the A3 altitudinal range, two climbers (4%), both from the Cucurbitaceae family, and finally a single arboreal species (2%), Vasconcellea candicans (A.Gray) A.DC. (Caricaceae), located in the A2 and A3 altitudinal ranges.
In the study area 46 native species were found (86.8% of the total species), of which 12 are endemic (22.6%): Philoglossa peruviana DC. (Asteraceae), Haageocereus acranthus (Vaupel) Backeb. subsp. acranthus and Loxanthocereus acanthurus (Vaupel) Backeb. (Cactaceae), Drymaria paposana var. weberbaueri (Muschl.) J.A. Duke (Caryophyllaceae), Dicliptera montana Lindau (Acanthaceae), Oxalis bulbigera R. Knuth (Oxalidaceae), Exodeconus prostratus (Dombey ex L'Hér.) Raf. (Solanaceae), Nicotiana paniculata L. (Solanaceae), Nolana humifusa (Gouan) I.M. Johnst. (Solanaceae), Clinanthus coccineus (Ruiz and Pav.) Meerow (Amaryllidaceae), Ismene amancaes (Ruiz and Pav.) Herb. (Amaryllidaceae) and Tillandsia latifolia Meyen (Bromeliaceae). Seven introduced species (13.2 %) were also recorded: Acmella alba (L'Hér.) R. K.Jansen (Asteraceae), Sonchus oleraceus L. (Asteraceae), Sisymbrium officinale (L.) Scop. (Brassicaceae), Chenopodiastrum murale (L.) S. Fuentes, Uotila and Borsch (Amaranthaceae), Erodium malacoides (L.) L'Hér. (Geraniaceae), Fumaria capreolata L. (Papaveraceae) and Parietaria debilis G. Forst. (Urticaceae). On the other hand, six species were identified in the study area under some level of threat: Vasconcellea candicans, critically endangered (CE); Begonia octopetala L'Hér and Loxanthocereus acanthurus, both in the threatened category (T); Ismene Amancaes and H. acranthus subsp. acranthus, in the vulnerable category (VU); and Ephedra americana in the near-threatened category (NT).
The A2 altitudinal range presented the highest richness, with 41 species distributed in 34 genera and 22 families; while A3 and A1 presented 30 species (25/17) and 28 species (24/15), respectively (Table 1). Likewise, the A2 range presented the highest number of unique species, which were found only in that altitudinal range (Table 2). On the other hand, 16 generalist species were found to be present in the three altitudinal ranges, where Chenopodium petiolare Kunth (Amaranthaceae), Nasa urens (Jacq.) Weigend (Loasaceae), Oxalis megalorrhiza Jacq. (Oxalidaceae), Nicotiana paniculata, Nolana humifusa and Solanum peruvianum L. (Solanaceae) stand out because they are also found in the massive rock zone, located at altitudes above the A3 range.
Table 1 Number of species in each altitudinal range
| Altitude | Families | Gender | Species |
| A1 | 15 | 24 | 28 |
| A2 | 22 | 34 | 41 |
| A3 | 17 | 25 | 30 |
In the NMDS analysis (stress 0.06), carried out to test the characterization of the altitudinal ranges according to the species composition of their transects, we expected to find three separate groups representing the different species composition of each altitudinal range. However, we found that there are species in common among the three altitudinal ranges. Group A1 is intercepted by A2 and A3, while the latter two are not intercepted by each other (Figure 2), indicating that the A1 altitudinal range has the most varied and generalist species composition, while the A2 and A3 altitudinal ranges present species with a more restricted distribution.
Table 2 Unique species and families recorded for each altitudinal range
| Altitude | Unique species | Unique families |
| A1 | Adiantum subvolubile, Ismene amancaes, Fuertesimalva limensis, Fuertesimalva sp. Nicandra physalodes | Pteridaceae |
| A2 | Baccharis coridifolia, Calceolaria pinnata, Cyclanthera mathewsii, Dicliptera montana, Drymaria paposana var. weberbaueri, Echeandia eccremorrhiza, Exodeconus prostratus, Fumaria capreolata, Mimosa albida, Oxalis latifolia, Oziroe biflora, Villanova oppositifolia, Mimosa albida, Oxalis latifolia, Oziroe biflora, Villanova oppositifolia | Acanthaceae, Asparagaceae, Calceolariaceae, Caryophyllaceae, Fabaceae, Papaveraceae |
| A3 | Ephedra americana, Exodeconus maritimus, Haageocereus acranthus subsp. acranthus, Loasa nitidae, Loxanthocereus acanthurus, Tillandsia latifolia | Cactaceae, Bromeliaceae and Ephedraceae |
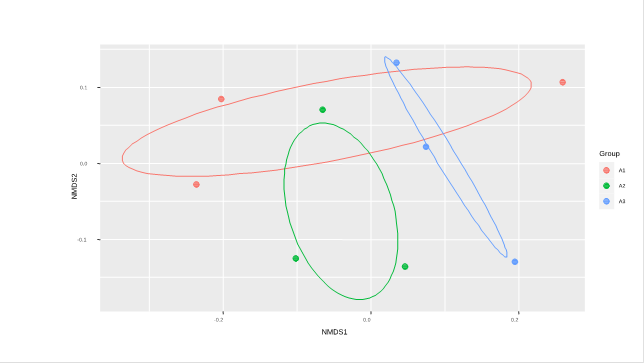
Figure 2 Grouping of the three altitudinal ranges (A1, A2, A3) of the Mangomarca fog oasis according to their species composition. Two altitudinal ranges are intercepted with A1, by means of a NMDS with "Bray" spacing and 0.06 stress. The points represent each of the 3 corresponding transects for each altitudinal range. A1: 350-500 m.a.s.l. A2: 500-650 m.a.s.l. A3: 650-800 m.a.s.l.
In a second NMDS analysis (stress 0.08), which shows the distribution of species and transects (Figure 3), it is evident that generalist species are agglomerated in the center of the graph, close to all transects. On the other hand, the unique species for each altitudinal range (A1, A2, and A3) are grouped close to the transects of their respective ranges, as observed with the species Baccharis coridifolia DC, Oxalis latifolia Kunth, Calceolaria pinnata L., Dicliptera montana, and Drymaria paposana var. weberbaueri, grouped and close to transects A2-T5 and A2-T6. Likewise, Loxanthocereus acanthurus, Haageocereus acranthus subsp. acranthus, Exodeconus maritimus (Benth.) D'Arcy (Solanaceae), Tillandsia latifolia, and Ephedra americana are observed grouped and close to transect A3-T9. The species mentioned coincide with the unique species for each altitudinal range in Table 2.
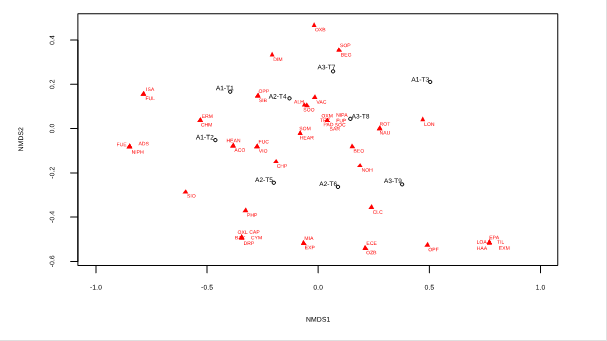
Figure 3 Distribution of the species of the Mangomarca fog oasis with respect to the transects of each altitudinal range. The NMDS was performed with a "Bray" spacing and had a stress of 0.08, where the triangles represent the relative location of each species, and the dots represent the transects (1-9) of each altitudinal range (A1, A2, A3). A1: 350-500 m.a.s.l. A2: 500-650 m.a.s.l. A3: 650-800 m.a.s.l. Acmella alba (ACO), Heliotropium angiospermum (HEAN), Heliotropium arborescens (HEAR), Sisymbrium officinale (SIO), Vasconcellea candicans (VAC), Alternanthera halimifolia (ALH), Chenopodiastrum murale (CHM), Chenopodium petiolare (CHP), Loxanthocereus acanthurus (LOA), Haageocereus acranthus subsp. acranthus (HAA), Drymaria paposana var. weberbaueri (DRP), Loasa nitida (LON), Nasa urens (NAU), Begonia geraniifolia (BEG), Begonia octopetala (BEO), Cyclanthera mathewsii (CYM), Sicyos baderoa (SIB), Mimosa albida (MIA), Erodium malacoides (ERM), Dicliptera montana (DIM), Calceolaria pinnata (CAP), Salvia rhombifolia (SAR), Fuertesimalva limensis (FUL), Fuertesimalva peruviana (FUP), Fuertesimalva sp. (FUE), Oxalis bulbigera (OXB), Oxalis latifolia (OXL), Oxalis megalorrhiza (OXM), Fumaria capreolata (FUC), Parietaria debilis (PAD), Exodeconus maritimus (EXM), Exodeconus prostratus (EXP), Nicandra physalodes (NIPH), Nicotiana paniculata (NIPA), Nolana humifusa (NOH), Solanum montanum (SOC), Solanum peruvianum (SOM), Solanum tuberosum (SOP), Clinanthus coccineus (CLC), Ismene amancaes (ISA), Echeandia eccremorrhiza (ECE), Oziroe biflora (OZB), Tillandsia latifolia (TIL), Rostraria trachyantha (ROT), Ephedra americana (EPA), Adiantum subvolubile (ADS).
As for the analyses at the family level, Cactaceae, Bromeliaceae and Ephedraceae were exclusive for altitudinal range A3. The A1 range only presented Pteridaceae as an exclusive family, and six families were found only in A2 (Table 2). Regarding the grouping of altitudinal ranges according to the family composition of their transects, the NMDS analysis (stress 0.06) shows that the three groups of the different altitudinal ranges intercept in the same area (Figure 4).

Figure 4 Grouping of the three altitudinal ranges (A1, A2, A3) of the Mangomarca fog oasis according to their family composition. It is observed that the three altitudinal ranges are intercepted by a NMDS with "Bray" spacing and 0.058 stress. The points represent each of the 3 corresponding transects for each altitudinal range. A1: 350-500 m.a.s.l. A2: 500-650 m.a.s.l. A3: 650-800 m.a.s.l.
Similar to what was obtained with the species-level analyses, the NMDS analysis (stress 0.07) shows that the families present in all altitudinal ranges were found agglomerated and close to all the transects evaluated, while those with restricted distribution were found closer to their corresponding transects (Figure 5).
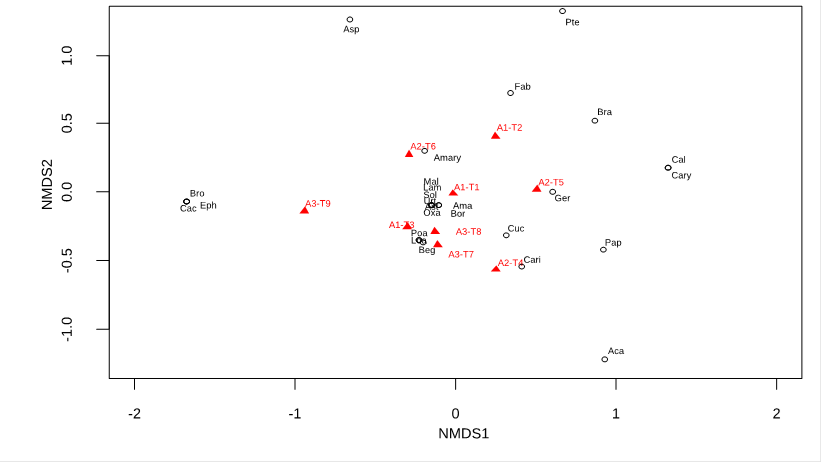
Figure 5 Distribution of the families of the Mangomarca fog oasis with respect to altitudinal ranges. The NMDS was performed with a "Bray" spacing and had a stress of 0.07, where the dots represent the relative location of each family, and the triangles represent the transects (1-9) of each altitudinal range (A1, A2, A3). A1: 350-500 m.a.s.l. A2: 500-650 m.a.s.l. A3: 650-800 m.a.s.l. Asteraceae (Ast), Boraginaceae (Bor), Brassicaceae (Bra), Caricaceae (Cari), Amaranthaceae (Ama), Cactaceae (Cac), Caryophyllaceae (Cary), Loasaceae (Loa), Begoniaceae (Beg), Cucurbitaceae (Cuc), Fabaceae (Fab), Geraniaceae (Ger), Acanthaceae (Aca), Calceolariaceae (Cal), Lamiaceae (Lam), Malvaceae (Mal), Oxalidaceae (Oxa), Papaveraceae (Pap), Urticaceae (Urt), Solanaceae (Sol), Amaryllidaceae (Amary), Asparagaceae (Asp), Bromeliaceae (Bro), Poaceae (Poa), Ephedraceae (Eph), Pteridaceae (Pte).
DISCUSSION
Previous studies that compiled lists of species of fog oases, among the most relevant is the list prepared by Dillon et al. (2011) for the hills of Peru and of Lleellish et al. (2015), for the hills of the department of Lima, where many of the species recorded in this research can be found. All of the species recorded in the present study are found in the species list of hills of Peru of Dillon et al. (2011), with the exception of: Sisymbrium officinale, Loxanthocereus acanthurus, Oxalis bulbigera and Adiantum subvolubile. 38 of the 53 species found in the present study were also found in the species list of Lima´s fog oases (Lleellish et al., 2015). The absence of some of the species recorded in the present study in the works of Dillon et al. (2011) and Lleellish et al. (2015), suggests that more floristic research is needed to contribute knowledge of the richness and diversity of the Mangomarca fog oasis, including increasing the sampling effort and considering more evaluation seasons.
Table 3 shows the high levels of richness and differences in flora diversity in the hills of Lima, Arequipa and Ancash, evidencing the uniqueness of these plant communities with native species (Dillon et al., 2011; Lleellish et al., 2015; Moat et al., 2021).
Table 3 Floristic studies in Lima´s, Arequipa and Ancash fog oases
| Name | Department | N° species | % of species in common with the present study | Reference |
|---|---|---|---|---|
| Mangomarca fog oasis | Lima | - | Present study | |
| Lachay fog oasis | Lima | 146 | 29% | Cano et al., 1999 |
| Lucumo fog oasis | Lima | 118 | 29% | Madrid-Ibarra and Cabanillas-Rodríguez, 2020 |
| Villa María fog oasis | Lima | 115 | 33% | Trinidad et al., 2012 |
| Amancae fog oasis | Lima | 53 | 51% | Trinidad et al., 2012 |
| Mangomarca fog oasis | Lima | 51 | 63% | Santa Cruz et al., 2020 |
| Pachacamac´fog oasis | Lima | 49 | 59% | MINAGRI, 2013b |
| Manchay fog oasis | Lima | 46 | 57% | MINAGRI, 2013d |
| Jicamarca fog oasis | Lima | 44 | 50% | Santa Cruz et al., 2021 |
| Lurin fog oasis | Lima | 42 | 57% | MINAGRI, 2013c |
| Retamal fog oasis | Lima | 30 | 63% | MINAGRI, 2013a |
| Atiquipa fog oasis | Arequipa | 339 | 8% | Talavera et al., 2017 |
| Mongon fog oasis | Áncash | 84 | 35% | Leiva et al., 2008 |
Four species stand out among the 13 studies and reports presented in Table 3 (Cano et al., 1999; Dillon et al., 2011; Leiva et al., 2008; Lleellish et al., 2015; Madrid-Ibarra and Cabanillas-Rodríguez, 2020; MINAGRI, 2013a, 2013b, 2013c, 2013d; Santa Cruz et al., 2020; Santa Cruz et al., 2021; Talavera et al., 2017; Trinidad et al., 2012), given that they were found in each of the studied hills, and in the present study. The species are: Sonchus oleraceus (Asteraceae), Fuertesimalva peruviana (Malvaceae), Nolana humifusa and Solanum montanum (both Solanaceae). Restricting the analysis to Lima only, we found the same species and additionally H. acranthus (Cactaceae) and Nicotiana paniculata (Solanaceae). These results agree with Dillon (2005), who recognizes the great representativeness of the Solanaceae family in the hills throughout Peru and Chile, presenting 128 species, distributed in 18 genera. Additionally, Exodeconus prostratus, Nicotiana paniculata and Nolana humifusa, are endemic to Peru (Dillon, 2005; Lleellish et al., 2015) and the genera they represent (Exodeconus, Nicotiana and Nolana) are part of the 11 genera of Solanaceae with endemic species in fog oases (Dillon, 2005).
A study of the biogeography and ecology of 39 hills located in central Peru showed the predominance of species of the genera Nolana and Solanum, which had the highest number of species, 17 and 11, respectively (Arana, 2019). The genus Nolana is the only one found in all hills throughout Peru and Chile, being the most widely distributed and the one with the highest number of endemic species of hills (70) within the Solanaceae family (Dillon, 2005), thus considered as indicator species of the hills (Lleellish et al., 2015). Likewise, the genus Solanum has species endemic to the fog oases (Dillon, 2005), as well as wild forms of cultivated varieties, or entered by settlers and subsequently found as wild forms, such as Solanum tuberosum L. (Santa Cruz et al., 2020). In the present study, Nolana humifusa was recorded in all altitudinal ranges of the Mangomarca fog oasis, even in the transect located above 800 m asl, and Solanum tuberosum as a feral species.
Another predominant species in the Mangomarca fog oasis and widely distributed in the hills of Peru is Fuertesimalva peruviana, a native species with stable populations, high seed production capacity (Lleellish et al., 2015) and rapid growth (Brako and Zarucchi, 1993), which in the present study was distributed throughout the three altitudinal ranges, being more abundant at low altitudes (A1).
The available information on the altitudinal distribution of the most predominant species in the Mangomarca fog oasis agreed with the distributions of the species found in the present study. As in the present study, extensive populations of Nasa urens, Salvia spp. (Lamiaceae), Sicyos baderoa (Cucurbitaceae), Solanum montanum (Solanaceae) and Fuertesimalva spp. (Malvaceae) have been recorded in the Mangomarca fog oasis, hills of Villa María and Amancaes, interspersed as patches on gently sloping hillsides (Santa Cruz et al., 2020; Trinidad et al., 2012). Likewise, the species Begonia octopetala (Begoniaceae) was recorded in rocky areas and stony ravines, and Nicotiana paniculata, Nolana humifusa, Solanum peruvianum, Loasa nitida Desr (Loasaceae), and species of the Cactaceae family at high altitudes, above 650 m asl, where humidity is lower (Padilla, 2018; Santa Cruz et al., 2020; Trinidad et al., 2012). Additionally, coinciding with the present study, Santa Cruz et al. (2020) recorded Tillandsia latifolia (Bromeliaceae) at high altitudes (A3), even in the massive rock zone, above 800 m.a.s.l., and Vasconcellea candicans and Solanum tuberosum, both at medium and high altitudes (A2 and A3).
On the other hand, Padilla (2018) mentions that Parietaria debilis presents greater coverage at low elevations. Solanum montanum and Fuertesimalva peruviana develop in ravines and medium slopes, while Nasa urens does so exclusively on high altitude slopes, coinciding with the distribution of these species in the present study. It is important to point out that these species appear on the hillsides progressively, first the predominant species of lower altitudes and towards the end of the wet season, those of higher altitudes.
Additionally, in the studies of Santa Cruz et al. (2020) and Padilla (2018) in the Mangomarca fog oasis, differences were found with respect to the present study, in terms of the distribution of some species. In the present study Dicliptera montana was found in the A2 altitudinal range, while Santa Cruz et al. (2020) recorded it in higher elevations. Likewise, Santa Cruz et al. (2020) recorded individuals of Baccharis sp. at low altitudes, while in the present study they were found only at intermediate altitudes. As for Trixis cacalioides (Asteraceae), Santa Cruz et al. (2020) mention that it is found scattered at low altitudes in a predominantly vegetative state and among rocks of different sizes, accompanied by Heliotropium angiospermum Murray, Heliotropium arborescens L. (Boraginaceae), and Ismene amancaes (Amaryllidaceae). Similarly, Padilla (2018) recorded the development of Trixis cacalioides in ravines and slopes at medium altitudes, and even greater coverage at high altitudes. However, in the present study it was distributed in a clustered manner at low altitudes (A1) and in a more dispersed manner at elevations higher than 500 m.a.s.l., being present in the three altitudinal ranges.
Among the multiple environmental factors that condition the development of this particular hilly vegetation are factors such as terrain, climate, marine temperature fluctuation (Dillon et al., 2011; Lleellish et al., 2015; Moat et al., 2021) and altitude (Muenchow et al., 2013a, 2013b).
Using models to predict the distribution of species in the hills, it was observed that altitude is one of the variables that most influences the prediction of species richness in the different zones of the hills (Muenchow et al., 2013a). Likewise, another study found that altitude alone explained 96% of the floristic composition of the different altitudinal strata in the Mongón fog oasis, showing four strata (belts) of vegetation, and reflecting the levels of water availability and soil properties of each stratum (Muenchow et al., 2013b).
In the present study, the A2 altitudinal range (500-650 m.a.s.l.) presented the highest richness (41 species), observing a well-documented pattern that holds that the highest species richness occurs at intermediate altitudes (Muenchow et al., 2013a, 2013b). This pattern would be a consequence of the greater availability of water in the hills, determined by the phenomenon of thermal inversion, which prevents the ascent of moisture from the fog at high altitudes (Muenchow et al., 2013a, 2013b). Thus, the maximum value of water availability occurs at altitudes immediately below the thermal inversion point, approximately 750-850 m.a.s.l. (Muenchow et al., 2013a, 2013b), as shown in Figure 6.

Figure 6 Model of the altitudinal profile of the Mangomarca fog oasis. The limits of the altitudinal ranges A1, A2, and A3 and the altitude at which the thermal inversion point and the area of greatest richness are found are shown, adapted from Muenchow et al. (2013a, 2013b) and Rundel et al. (1991).
The presence of higher levels of richness at intermediate altitudes in the Mangomarca Hills was evidenced in a previous study, where the highest development of species was found at altitudes close to 500 m.a.s.l., coinciding with the average altitude of development of the hills in Peru (Padilla, 2018). Thus, during the wet season (August and September), the highest species richness and relative cover, such as the highest levels of diversity, were recorded in three transects located between 491 m.a.s.l. and 607 m.a.s.l. (Padilla, 2018). Additionally, between altitudes of 600 and 750 m.a.s.l., the highest coverages were found. The pattern was also observed in the Mongón (Áncash) fog oasis, with the range of highest richness being 700-800 m.a.s.l. (Muenchow et al., 2013a).
Additional factors have been identified that influence the development of species in the hills, in addition to altitude. Brack and Mendiola (2004) mention that the density and development of plant communities depends on coastal humidity and the continuity of cloudy days during the winter months in the coastal region of the Pacific desert, while Rolando et al. (2017) mention that in addition to altitude, gravimetric soil moisture and its organic matter positively influence the assemblage of plant communities. Likewise, according to Muenchow et al. (2013a), at a smaller spatial scale, topographic surface water uptake and slope, play an important role in the richness of each hill site. Additionally, soil texture and salinity have been recorded to determine 88% of the floristic composition and species distribution at the local scale (Muenchow et al., 2013a, 2013b). Finally, it was recently found that the presence of vegetation on hillocks presents a significant relationship with the slope of the site, as well as with the direction of wind flow, influenced by ground level topography (Moat et al., 2021). These wind movements across the terrain produce significant microclimatic changes and can alter the patterns of the species niche and habitat (Moat et al., 2021).
Regarding the Mangomarca fog oasis, Padilla (2018) did not find significant correlations between humidity, altitude and slope of the transects. Humidity would be more correlated with slope than with altitude. As for the accumulated cover, it did not present significant correlation for the categories of slope, position or altitude. Similarly, according to Padilla (2018), it is not possible to conclude that altitude determines the composition of the plant community, however, indicates a certain tendency for the relationship between these variables to become significant in wetter seasons, attributing it to the greater availability of data at that time. Similarly, the composition of the plant community is not influenced by the orientation of the slopes of the evaluation points, however, low altitudes (450 m.a.s.l.) favor the development of herbaceous species and higher altitudes (approx. 700 m.a.s.l.) favor the development of shrub species, cacti and certain herbaceous species (Padilla, 2018).
On the other hand, Santa Cruz et al. (2020) identified altitude, the slope and the presence of rocky areas as the factors that influence the distribution of its plant communities. However, more studies are needed to determine with certainty the environmental factors that positively influence the development of the vegetation of the Mangomarca fog oasis. Although this study presents the floristic richness and composition existing in each altitudinal range of these hills, it is not possible to determine which variables most influence the distribution of the species of the Mangomarca fog oasis in the period studied. This is due to the method used, since only data on the presence and absence of species in each altitudinal range were generated and the environmental variables associated with the species in each established transect were not evaluated.
With respect to the species composition of the different altitudinal ranges, overlaps were observed between the A2 and A3 altitudinal range and the A1 altitudinal range. This implies that a significant part of the species present in the A1 altitudinal range are distributed in the other altitudinal ranges. On the other hand, the floristic compositions of the A2 and A3 altitudinal ranges are apparently distant from each other, since they do not overlap, while at the family level an overlap between the three altitudinal ranges was observed. It could be said that altitude has not determined species and family compositions sufficiently different from each other to form distinct groups between altitudinal ranges, which agrees with the literature, where altitude is not mentioned as a main determinant of richness or species composition of hills (Moat et al., 2021; Muenchow et al., 2013a, 2013b). Although there are unique species and families for each altitudinal range, they have not been sufficient to differentiate groups, suggesting that there is a predominance of species occurring in two or three altitudinal ranges.
On the other hand, the altitudinal distribution of herbaceous, shrub, tree and cactus species in the Mangomar fog oasis coincided with that described by Ellenberg (1959) and Ono (1982), who identified herbaceous species between 200 and 450 m.a.s.l., woody and tall herbaceous species at altitudes between 300 and 650 m.a.s.l., and cacti and bromeliads between 400 and altitudes above 700 m.a.s.l.. In the present study, it was possible to appreciate a tendency for cacti to develop at altitudes of more than 650 m.a.s.l. (A3), while the distribution of woody species (trees, shrubs and sub-shrubs) was greater in the A2 and A3 ranges. The above coincides with what was found in the bromeliad stratum of the Cerro Campana fog oasis, which is described as the altitudinal stratum of highest humidity, vegetation cover and with the highest species richness (Muenchow et al., 2013b), even called the stratum of woody species (Dillon and Rundel, 1990). However, the low abundance of arboreal individuals in these strata is common (Dillon and Rundel, 1990; Muenchow et al., 2013b), which could be attributed to anthropogenic impact, such as burning for agricultural purposes (Dillon et al., 2003). Whereas, in the hills of Quebrada Verde (Pachacamac), herbaceous species were dominant at low altitudes and woody species were found in the highest stratum, where there was more moisture and organic matter in the soil during the year (Rolando et al., 2017). However, the appreciation of the present study should be taken as preliminary results for this aspect, given the absence of sufficient data to determine with certainty the relationship between growth forms and altitude (NMDS analysis stress: zero, indicating insufficient data).
The Mangomarca fog oasis is severely threatened by urban expansion associated with anthropic pressures, such as the introduction of exotic species (Manrique et al., 2014; Pollack et al., 2020; Santa Cruz et al., 2020; Trinidad et al., 2012). Only Mangomarca Creek is conserved, unlike the adjacent Campoy and Los Sauces creeks, which are severely reduced due to urban encroachment (Santa Cruz et al., 2020). The rate of degradation of this ecosystem makes it necessary to promote research on the effects of these disturbances on the flora of this ecosystem. In addition, it is necessary to contribute information on the conservation status of hill species and their resilience to environmental changes resulting from increasing anthropogenic influence, the coastal El Niño phenomenon, and climate change (Muenchow et al., 2013a). In addition, it is especially important to update the conservation status species from the hills, given that it is suspected that the levels of threatened or endangered species are higher than they are at present (Moat et al., 2021).
It has been recorded that in the most impacted hillsides there are fewer individuals of shrub and tree species (Dillon et al., 2003; Muenchow et al., 2013a; Trinidad et al., 2012). The loss of tree species cover in arid and semi-arid ecosystems profoundly changes their processes and functionality (Breshears, 2006), due to the fact that trees have the capacity to capture greater amounts of moisture from the mists of hills, fulfilling the function of a wet nurse for species that develop under the canopy (Oka, 1986; Sotomayor and Drezner, 2019; Walter, 1973). This aspect, for example, must be taken into account for future studies of the nurse effect and moisture retention by woody species, which benefit the development of herbaceous species of hills, which would make visible the fundamental role of woody species in the ecosystem, and thus, promote their use in conservation and restoration plans. Likewise, there is an urgent need for conservation plans that contemplate adequate delimitation and land use planning in areas close to hilly vegetation, as well as initiatives for the implementation of sustainable tourism programs (Santa Cruz et al., 2020; Trinidad et al., 2012). If this is not done, it is possible that the vegetation of the less conserved creeks of the Mangomarca fog oasis will disappear in its entirety in the coming years, depriving nearby populations, and even the population of Lima, of the ecosystem services they provide (Santa Cruz et al., 2020). Therefore, it becomes crucial to raise awareness and promote knowledge about the importance and benefits provided by this ecosystem to the population, as has been done successfully in the Lúcumo fog oasis, through tourism (Nieuwland and Mamani, 2017).
CONCLUSIONS
The Mangomarca fog oasis is an ecosystem with high levels of richness, diversity, and endemism, presenting 53 species, of which 12 are endemic. There were 28 species in the lower altitudinal range, 41 in the middle range and 30 in the upper range, with unique species in each range. Based on the analysis of floristic composition and altitude, it can be affirmed that the effect of altitude in the three altitudinal ranges is not significant to affirm that there are three groups of plant communities significantly differentiated by altitude in the Mangomarca fog oasis.






















