FAO estimates that 14% of the world's food is lost from post-harvest up to (but not including) the retail level (FAO, 2019). 25% of roots, tubers and oil-bearing crops are lost, followed by fruits and vegetables (22%), meat and animal products (12%) and cereals and pulses (9%). The most of the losses are due to microbiological and physiological deterioration as well as mechanical damage during harvesting, transportation and marketing stages.
Ecuador is an important world producer of fruits and vegetables. Among fruits, strawberry crop has been developed in Ecuador for the last years (Parra, 2018), with a monthly production of 300000 t. Microorganisms like Botrytis cinerea and Rhizopus stolonifer generate post-harvest losses of strawberry (Alcántara, 2009; Becerra et al., 2013). Additionally, this fruit is a carrier of some foodborne pathogens, e.g., Escherichia coli and Salmonella spp. (Carrasco and Caro, 2017). Therefore, to ensure the quality of strawberries, it is necessary to minimize the presence of pathogenic microorganisms that, at the same time, may affect consumer health (García-Robles et al., 2017).
There are various methods to reduce the microbiological load on the surface of fruits and vegetables. In general, the methods are based on physical processes such as mechanical removal, heat treatment, irradiation, and chemical methods. The use of an alkaline pH to control pathogenic microorganisms in food has not been widely studied. One of the reasons could be that most foods have a pH below 7. There are exceptions such as the lutefisk, an ancient tradition in Norway, Sweden and Finland of a fish prepared in lye, with a pH up to 12 (Lunestad et al., 2018).
In general, bacteria have an optimal growth pH close to neutrality; while fungi have a wider pH range, such as B. cinerea, which germinates in a pH range of 3 to 7 (Martínez and Moreno, 2008). There is a group of microorganisms, called alkaliphiles, that is developed at pH greater than 8, commonly between 9 and 10. These microorganisms are found in highly alkaline environments, such as soda lakes and carbonated soils (Lunestad et al., 2018).
Based on the previous information, the aim of this work was to evaluate alkaline solutions (pH 11, 12 and 13) via in vitro against growth of B. cinerea and R. stolonifer in strawberry. Additionally, a combination of alkaline solutions (pH 11, 12 and 13) and immersion times (20, 40 and 60 min) was used to inhibit the growth of Salmonella spp. and E. coli in strawberry.
MATERIALS AND METHODS
Strawberries (Fragaria x ananassa) were purchased in the central market of Manta city, Ecuador. Strawberries with an approximate weight of 20 g each, with no mechanical damage and with a maturity degree of 4, on a scale of zero to six, were chosen (NTC 4103, 1997) and washed with distilled water.
A total of 72 strawberries were used for microbiological analyses of Salmonella and E. coli, whereas 24 strawberries were used for B. cinerea and R. stolonifer. Two types of completely randomized designs were used. A unifactorial design to study of the effect of pH as a control of B. cinerea and R. stolonifer, where the independent variable was the pH at 3 levels (11, 12 and 13) and the dependent variables were microbial counts as CFU and the inhibitory effect against B. cinerea and R. stolonifer. A two-factor design was used to study the effect of pH as a control of Salmonella spp. and E. coli, being the independent variables pH (11, 12 and 13) and immersion time (20, 40 and 60 min) and the dependent variables were microbial counts as CFU and the inhibitory effect against Salmonella spp. and E. coli as mm of inhibition zone.
Control of B. cinerea and R. stolonifer by immersion in alkaline solutions
Strawberries were inoculated at 104 CFU mL-1 with B. cinerea and 105 CFU mL-1 with R. stolonifer (Camacho and Nieto, 2017). Sodium hydroxide solutions pH of 11, 12 or 13 were prepared by adding and dissolving NaOH in distilled water, under constant stirring, until the desired pH was reached. Afterwards, the fruits were placed in NaOH solutions pH 11, 12 or 13 and immediately were rinsed with distilled water. Strawberry surface swabbing was performed for microbiological analysis. Microbial growth was reported as CFU of B. cinerea and R. stolonifer, according to the methodology described by "Norma Técnica Ecuatoriana" NTE INEN 1529-10:2013 (INEN, 2013). All analyses were performed in triplicate.
Control of Salmonella spp. and E. coli by immersion in alkaline solutions
Strawberries were inoculated at 106 CFU mL-1 with both Salmonella and E. coli (Ledesma et al., 2018). Afterwards, the fruits were immersed in alkaline solutions pH 11, 12 or 13 during 20, 40 or 60 min. A strawberry surface sampling was performed (previously described) and microbial growth was reported as CFU.
Inhibition of the growth of B. cinerea, R. stolonifer, Salmonella spp. and E. coli by alkaline solutions
Analysis of inhibition was determined according to EUCAST (2013) with slight modifications. Petri dishes were inoculated at 104 CFU mL-1 with B. cinerea and 105 CFU mL-1 with R. stolonifer (Camacho and Nieto, 2017) using Sabouraud dextrose culture medium, whereas Salmonella spp. and E. coli were inoculated both at 106 CFU mL-1 in Salmonella-Shigella agar (HiMedia Laboratories, India). Afterwards, Petri dishes were incubated at 37 °C for 2 days. An amount of 20 µL of alkaline solution was added to filter paper disks (Fisher Scientific Q2) of 5 mm diameter. The disks were placed in the centre of the Petri dish, previously prepared and incubated at 25 °C with both B. cinerea and R. stolonifer for 24 h and Salmonella spp. and E. coli at 37 °C for 24 h. The zones of inhibition of microorganisms growth were measured after incubation and reported as mm of inhibition. Analyses were performed in triplicate.
RESULTS AND DISCUSSION
Control of B. cinerea and R. stolonifer by immersion in alkaline solutions
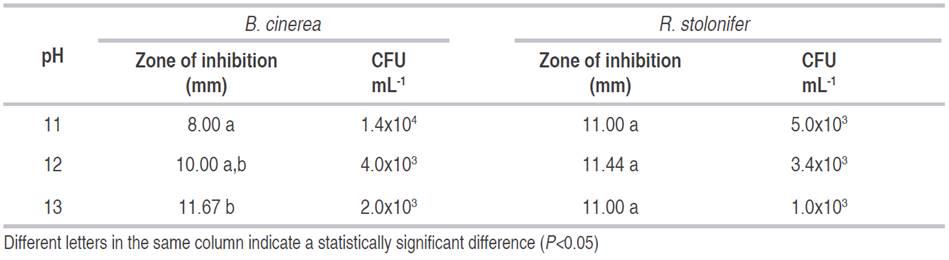
Results of the effect of alkaline solutions against B. cinerea showed differences among the three pH (P<0.05). The smallest zone of inhibition was obtained by a pH 11 solution, with a diameter of 8 mm, whereas the largest zone by a pH of 13 with a diameter of 11.67 mm (Table 1). An increase of pH led to 1-log reduction from pH 11 to 12. Regarding R. stolonifer, there was no difference on the inhibition zone among the three pH (P<0.05) and additionally, the increase of pH did not cause a log reduction in CFU mL-1.
Similar results of inhibition were obtained by Ahlem et al. (2012), showing that an alkaline pH 10 gave a better inhibition of B. cinerea than a lower pH. Besides, Qin et al. (2010) showed the effectiveness of NaOH solution to control B. cinerea on table grapes.
Inhibition of B. cinerea and R. stolonifer in the presence of an alkaline solution could be due to a drying effect of the microorganism resulting from osmotic dehydration. In fact, salinity affects microbes via osmotic effect by drawing water out of cells which may kill microbes through plasmolysis (Oren, 1999).
Control of Salmonella spp. and E. coli by immersion in alkaline solutions
Effect of pH. There were no differences on the zones of inhibition of Salmonella spp. (Table 2), whereas differences were found for E. coli (P<0.05), with zones of inhibition of 11.56 and 12.11 mm, when solutions at pH 11 and 12, respectively, were used. Smaller zones (10.33 mm) were obtained using solutions pH 13. The highest pH values led to a higher inhibition in Salmonella spp. In fact, salts have been used to control Salmonella spp. in food (Aspridou et al., 2018; Tiganitas et al., 2009). Zhou et al. (2011) observed that Salmonella suffers an initial decline in cell numbers when inoculated into a high salt concentration medium. However, when the stress is not lethal, the cells could adapt and subsequently grow under the new condition. Similar studies in sub-lethally stressful environments reported that cell populations suffered an initial loss followed by a recovery (Mellefont et al., 2005). Differences of zone of inhibition between pH 13 and lower pH (11 and 12) may not reflect real differences since longer times of analyses may be needed to guarantee a full recovery of cell population.
Effect of immersion time. Table 3 shows that there was no growth of Salmonella spp. for the three immersion times in the pH 13 solution. The 20 min immersion in a pH 11 solution showed the highest CFU counting. Regarding E. coli, there were differences of CFU counting among different immersion times (P<0.05). Treatment of pH 13 for 20 min showed the highest CFU counting (5.92x106 CFU mL-1) and pH 13 for 60 min showed no growth. Sampathkumar et al. (2003) showed a reduction of CFU of Salmonella enterica when pH was increased of 10 to 11 within 20 min of exposure to alkaline solutions, whereas Gill et al. (2019) observed a reduction of Salmonella enterica population after exposure to NaOH solution pH 11 for 2 h. Different results may be due to the use of a different strain. The difference in growth among the bacterial species examined, could be due to different strategies to cope with osmotic stress (Wood, 2007).
CONCLUSIONS
The present study showed that alkaline solutions at pH 13 can control the growth of B. cinerea and R. stolonifer in strawberries. The immersion of strawberries in alkaline solutions of pH 13 for 60 min inhibited completely the growth of Salmonella spp. and E. coli in strawberries. Complementary studies of dehydration of strawberries after immersion in alkaline solutions should be performed along with the use of other alkalis.