Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Revista colombiana de Gastroenterología
versión impresa ISSN 0120-9957
Rev Col Gastroenterol vol.33 no.3 Bogotá jul./set. 2018
Letter to the editor
Letter to the editor. Project URKUNINA 5000
1Director científico. Centro de Investigación de Enfermedades Digestivas y Nutricionales, Hospital Universitario Departamental de Nariño. Pasto, Colombia
San Juan de Pasto, June 20, 2018
Doctors
Colombian Journal of Gastroenterology
Bogotá
Please receive my warm greetings.
As a member of the Colombian Association of Gastroenterology and the Colombian Association of Digestive Endoscopy, I would like to respectfully ask that you consider publishing the following summary of the URKUNINA 5000 Project. This is a research project on the “prevalence of precursor lesions of Gastric malignancy and the effect of the eradication of Helicobacter pylori infections as primary prevention of gastric cancer in the department of Nariño.” It has been approved by Colciencias. Publication will disseminate information about this project throughout the scientific community. Special note should be taken that the project is already widely known in the international community although it is not yet sufficiently well-known at the national level.
I await your kind response, and I thank you in advance for your collaboration and attention.
Cordially,
Álvaro Bedoya U.
Scientific Director
Digestive and Nutritional Diseases Research Center,
Departmental University Hospital of Nariño
PROJECT URKUNINA 5000
Investigation of the prevalence of gastric malignancy precursor lesions and the effect of Helicobacter pylori infection eradication for primary prevention of gastric cancer in the Nariño department
Álvaro Bedoya, MD,1 Yomaira Yépez, MD,2 Diana Calvache, MD,2 Yenny Cifuentes, MD,2 Nubia Lucero, MD,2 Patricia González, MD,2 Álvaro Bedoya G., MD,2 Eliana Manosalva, MD,2 Teresa Martínez, MD, MSc,3 Arecio Peñalosa, MD,4 Blanca Piazuelo, MD5
1 Scientific Director of CIEDYN (Centro de Investigación de Enfermedades Digestivas y Nutricionales - Research Center for Digestive and Nutritional Diseases) at the Departmental University Hospital of Nariño
2 Researcher at (CIEDYN).
3 Master’s in Epidemiology from the National Institute of Cancer
4 Chief of Gastroenterology and Endoscopy at FUCS (Fundación Universitaria de Ciencias de la Salud - University Foundation of Health Sciences)
5 Research Pathologist at Vanderbilt University.
COOPERATING ENTITIES
CIEDYN (Centro de Investigación de Enfermedades Digestivas y Nutricionales - Research Center for Digestive and Nutritional Diseases)
Government of the Department of Nariño
Colombian National Institute of Cancer
Colombian Association of Surgery
FUCS (Fundación Universitaria de Ciencias de la Salud - University Foundation of Health Sciences)
INTRODUCTION
The URKUNINA 5000 project was approved by Agreement No. 57 of June 9, 2017 of the Collegiate Body of Administration and Decision of the Science, Technology and Innovation Fund of the General Royalty System of Colciencias (Departamento Administrativo de Ciencia, Tecnología e Innovación - Administrative Department of Science, Technology and Innovation).
Urkunina is an indigenous word that means “mountain of fire”. It was chosen in honor of the Galeras volcano, the sentinel of San Juan de Pasto in the department of Nariño, Colombia. The project is based in this city and has 5,000 voluntary participants in the 55 municipalities of the Andean area of Nariño, hence the name URKUNINA 5000.
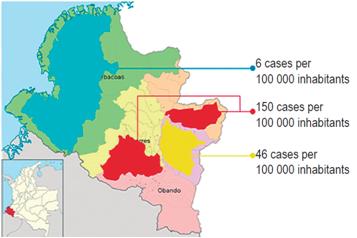
The department of Nariño is a high risk area for gastric cancer. It is located in southwestern Colombia on Pacific coast bordering Ecuador. It has approximately 1,800,000 inhabitants and 64 municipalities. The fifty-five of these that are located in the Andean zone contain 81.7% of the population of Nariño. The department is characterized by a special geographical, social and ethnic situation: its high mountainous Andean area has a high incidence of gastric cancer while its low coastal area has a low incidence of this type of cancer. This small territory of 32 265 km2 has three well defined gastric cancer zones, a situation that makes it special, and perhaps unique in the world:
Two red zones are located to the north and south-west. They have incidences of gastric cancer that are among the highest in the world (150 cases per 100,000 inhabitants), and the prevalences of precursor lesions of malignancy and Helicobacter pylori infections are both high.
A yellow zone in the capital, San Juan de Pasto, and surrounding municipalities. The incidence of gastric cancer in this area is 46 cases per 100,000 inhabitants.
A green zone on the Pacific coast whose incidence is six cases per 100,000 inhabitants and which has a lower prevalence of precursor lesions of malignancy but a high prevalence of H. pylori infections. This is currently known as the Nariñense or Colombian enigma (Figure 1). 1,2,3
This was reflected in a study published in 2008 by Dr. Sánchez that confirmed that the epidemiology of gastric cancer in our country has not changed significantly. 4 That study states that gastric cancer is predominantly Andean and the areas with the highest mortality rates are the departments of Nariño and Cauca (Figure 2).
Based on this information, we compared our gastric cancer mortality rate to rates elsewhere in Colombia, Latin America and the rest of the world. We used five-years rates as an indicator. According to the medical literature, in Japan it is around 60%, in the United States and Europe it as about 30%, in Chile and Brazil it is about 18%, in Colombia it is 16% and in Pasto it is 13%. This very poor survival rate confirms that we are neither working logically nor rationally (Figure 3).
The results above lead us to believe that one of the most important causes of our low survival rate is delayed diagnosis of cancer. The intervention made in Japan to diagnose cancer early has led to 73% of the cases being diagnosed early while in our country early diagnoses account for only 2% to 5% of all cases. This great difference directly influences the high gastric cancer mortality rate (Figure 4). 7,8,9.
The facts that we have lived in the Andean zone for a long time and have monitored the natural history of this disease for more than twenty years have allowed us to better understand the carcinogenesis of this disease. It is unquestionably the product of several factors. In our region we have found five principal risk factors including high prevalences of H. pylori infections ranging from 60% in children to 90% in adults, 5 high prevalences of malignancy precursor lesions (38.6%), 3 high salt intakes, low micronutrient and antioxidant intakes, and genetic susceptibility. These factors are responsible for 90% of the cases of gastric cancer. Of this 90%, seventy percent is accounted for by the association of malignancy precursor lesions and H. pylori infections. They have become a deadly duo in relation to the appearance of gastric cancer (Figure 5). (5,10
Taking advantage of the Law of Science, Technology and Innovation Royalties, and accompanied by other institutions, we presented the URKUNINA 5000 project to Colciencias. The project’s general objective was to evaluate the prevalence of gastric malignancy precursor lesions and the determinants of initial eradication of H. pylori infections in a voluntary population drawn from the 55 municipalities of the Andean area of the department of Nariño. Of the four specific objectives, the most important is the creation of a first reserve of biological material of the population at high risk for gastric cancer and precursor lesions of malignancy in the department of Nariño for future national and international research (Figure 6).
The protocol for taking biopsies requires ten biopsies: five from the antrum, two from the angular incisure and three from the corpus.
Two antrum biopsies at 3 cm from the pylorus, one angular incisure biopsy and two corpus biopsies at 8 cm from the cardia are intended for conventional histology study with hematoxylin-eosin staining, modified Giemsa staining for the detection of H pylori, and histochemical staining with Alcian blue for confirmation and classification of intestinal metaplasia.
One antrum, one angular incisure and one corpus biopsy intended for extraction of nucleic acids in dry NUC at minus 80 ºC.
An antral biopsy placed in glyceryl guayacolate for culturing and typing of the bacteria; and
An antral biopsy for rapid urease test validation.
The histology will be reported on a visual scale similar to the updated version of the Sydney system. This will allow us to establish the status in the OLGA/OLGUIN system (Operative link for gastritis assessment) (Figure 7).
Logistics procedures will be carried out and coordinated by CIEDYN researchers. The supervision and development of the project’s administrative activities will be occur during its realization. The first phase will be presentation and explanation of the project to the mayors, managers of state social enterprises, and the doctors of the 55 municipalities of the Andean area. The government of Nariño and CIEDYN will participate in this work and in dissemination of information and publicity aimed at the target population. The second phase will consist of field work with the 5,000 volunteers. This will include performance of upper digestive endoscopies for collection of biological material, taking blood samples and separating them into blood components, and taking urine, saliva and fecal matter samples for further studies. The blood components and biopsies in dry NUC will be placed in containers at temperatures at minus 80 ºC and will be sent to the tumor biobank of the National Institute of Cancer in Bogotá which will keep them at minus 80 ºC.
Biopsies for histological study will be sent to the pathology department of Hospital San José de Bogotá which will keep them available virtually. The diagnostic report for quality control will be done by Dr. Blanca Piazuelo of Vanderbilt University.
The results will allow us to divide the group of volunteers into three groups:
Those without malignancy precursor lesions will be referred to the first level of care with recommendations.
Those diagnosed with gastric cancer, high-grade dysplasia and other gastric lesions who will be referred to the third level of care.
Those with confirmed diagnoses of malignancy precursor lesions. If they test negative for H. pylori, they will be provided nutritional education and subsequent endoscopic follow-up. If they test positive for H. pylori, they will undergo sequential triple treatment with 40 mg esomeprazole/day divided into two doses for 10 days, 2 grams of amoxicillin in 2 doses/day for 5 days, and 1 g clarithromycin in 2 doses/day for 5 days. Twelve weeks later a breath test will be performed. The results will be classified as positive if they indicate the presence of H. pylori and negative if H. pylori is absent. A positive result after treatment will indicate that the therapy has failed. This group will be meticulously studied by the research group to discover the causes of these failures, and they will be referred to the respective EPS for rescue treatment with recommendations of therapy to follow. These volunteers will undergo endoscopic and histological follow-ups to analyze changes in the gastric mucosa with respect to the first examination (Figure 8).
Final study goals:
Characterization of the population: After Japan, Nariño will be the only other part of the world that has histologically studied its population and has determined the prevalence of precursor lesions of malignancy and H. pylori. These data will allow orientation of pertinent actions of promotion and prevention in the municipalities with the greatest risks.
Measurement of the effectiveness of H. pylori eradication treatment with breath tests in order to reorient strengthen depending upon whether or not it is effective.
Typing of bacterial strains by municipality and identification of possible mutations to analyze the susceptibility to conventional treatment.
Creation of a biobank for future national and international research. There will be 45,000 samples of gastric tissue, 20,000 blood samples, more than 3,000 genomes of H. pylori strains, and urine, saliva and fecal matter samples. This area will become the only region in the world with a genetic reserve of such magnitude.
Development of low cost and easy-to-use biomarker technology is projected for phase II.
Development of a vaccine is our dream, but with the appropriate genetic samples, a low-cost, safe and effective, either curatively or prophylactically, vaccine against H. pylori will be sought in phase III. This will require research and financial support from multiple centers.
This project will contribute to strengthening public policies for early detection of gastric cancer and malignancy precursor lesions.
Finally, the results of this research will contribute to the social appropriation of the knowledge of the medical community regarding diagnosis, risk, follow-up, severity and treatment of malignancy precursor lesions.
Acknowledgments
We would like to express our appreciation to the advisors:
Dr. Pelayo Correa
Professor of Medicine,
“Anne Potter Wilson” Chair of Cancer Research Division of Gastroenterology
Vanderbilt University
Dra. María Constanza Camargo, PhD, MSc, MHA
Infectious Diseases, Immunology and Epidemiology Branch
Division of Cancer Epidemiology and Genetics
National Cancer Institute
Dr. Guillermo Peres Peres
Associate Professor of Medicine and Microbiology
New York University School of Medicine
Department of Medicine
Dra. Alba Lucía Trespalacios, PhD, MSc
Titular Professor and Director of Research Line
Pontificia Universidad Javeriana
Dr. Germán Campusano
Associate Professor of Hematology and Pathology
University of Antioquia
Dr. Arley Gómez López
Doctor, Master in Molecular Biology and Biotechnology
Doctorate in Medical Sciences, Doctorate in Nanomedicine
University Foundation of Health Sciences - FUCS Bogotá
Dra. Anabella Fajardo
Bacteriologist, Master in Biomedical Sciences
University Foundation of Health Sciences - FUCS Bogotá
Dr. Segundo Morán Villota
Senior Researcher, Mexican Institute of Social Security
National Researcher Level III
Laboratory of Gastrohepatology Research
Hospital of Pediatrics, Centro Médico Nacional Siglo XXI México
Dr. Richard M. Peek Jr.
Director, Division of Gastroenterology
Professor of Medicine Cobb Mine Wallace Chair in Immunology, Professor of Cancer Biology
Vanderbilt University
Dr. Keith T. Wilson
Professor of Medicine, Professor of Pathology, Microbiology and Immunology, Thomas F. Frist Sr. Chair in Medicine, Professor of Cancer Biology
Vanderbilt University
Referencias
1. Correa P, Piazuelo MB. Cáncer gástrico: el enigma colombiano. Rev Col Gastroenterol. 2010;25(4):334-7. [ Links ]
2. Correa P. Cáncer gástrico: una enfermedad Infecciosa. Rev Col Cir. 2011;26(2):111-7. [ Links ]
3. Bedoya A, Sansón F, Yépez Y, Santacruz C, Cifuentes C, Calvache D, et al. Prevalencia y severidad de las lesiones precursoras de malignidad en un área de alto riesgo de cáncer gástrico. Pasto 2012. Rev Col Gastroenterol. 2012;27(4):275-81. [ Links ]
4. Sanches W, Peña D, García D. Mortalidad por cáncer gástrico en Colombia. Informe epidemiológico. Rev Col Cir. 2008;23(3):41. [ Links ]
5. Bedoya A. Prevención del cáncer gástrico en Nariño. Una tarea difícil, pero factible. Rev DGSM. 2012;1(1):28-9. [ Links ]
6. Bedoya A. Urkunina 5000 en Colombia. Conferencia del VIII Simposio Internacional de Cáncer gástrico y Helicobacter pylori. Bello Horizonte, Brasil. 2018. [ Links ]
7. Kenji Y. Hospital Universitario de Chikushi - Fukuoka. Comunicación personal. [ Links ]
8. Bedoya A. El CIED contra el cáncer gástrico en Nariño. Rev Debates. 2009;32:30-1. [ Links ]
9. Otero W. Cáncer gástrico en Colombia: un diagnóstico tardío que amerita el compromiso del Estado. Rev Col Gastroenterol. 2008;23(4):302-4. [ Links ]
10. Bedoya A. Lesiones precursoras de malignidad y su asociación con infección por Helicobacter pylori: dúo mortal. Conferencia del 18.o Congreso de Ciencias Básicas y Especialidades Clínicas. Congreso intermedio de la ACC. Bogota, abril, 19-21 de 2018. Publicado en: Órgano para la difusión del programa de formación continuada del médico. Facultad de Medicina. AEXMUN. 2018;23(1):11. [ Links ]
Received: June 20, 2018; Accepted: June 25, 2018











 texto en
texto en