Introduction
Granulomatosis with polyangiitis (GPA) is a small- and medium-vessel vasculitis with a preference for the airways and kidneys. It causes fibrinoid necrosis leading to organ dysfunction. Gastrointestinal (GI) involvement is observed in only 5% to 10% of cases and is a poor prognosis factor1. The small intestine and colon can be affected and cause life-threatening complications.
Case presentation
This is a 64-year-old male patient with a medical history of chronic kidney disease (CKD) diagnosed two months ago, with a 46.6 mL/min/1.73 m2 glomerular filtration rate (GFR). He consulted for a 15-day clinical picture of asthenia, adynamia, subjective oliguria, upper and lower limbs edema, hyporexia, and melena. In his admission physical examination, he was found with a 113/73 mm Hg blood pressure (BP), 88 beats per minute (bpm) heart rate (HR); during auscultation, decreased breath sounds in the lung bases and rhonchi bilaterally, limbs with grade II edema in the lower extremities and positive rectal examination for a mane.
Admission laboratory tests showed normocytic normochromic anemia (hemoglobin [Hb]: 7.7 g/dL, mean corpuscular volume [MCV]: 80.7 fL, mean corpuscular hemoglobin [MCH]: 25.8 pg), erythrocyte sedimentation rate (ESR): 15 mm/h, serum creatinine: 3.5 mg/dL, and non-nephrotic range proteinuria (1.05 g/24 hours). The urinalysis showed negative nitrites, negative proteins, blood hemoglobin 300 mg/dl, red blood cells 28 per field, leukocytes 10 per field, scarce squamous cells, and scarce bacteria.
Given the patient’s clinical picture and his CKD exacerbation, he underwent a urinary tract ultrasound in which an increase in the echogenicity of the right and left renal parenchyma compatible with changes due to acute bilateral nephropathy was found. Thus, he underwent a renal biopsy with an anti-myeloperoxidase (anti-MPO) and anti-proteinase 3 (anti-PR3) antibodies, compatible with rapidly progressive glomerulonephritis crescent formation, positive for anti-MPO. Therefore, the patient was diagnosed with vasculitis associated with antineutrophil cytoplasmic antibodies (ANCA) microscopic polyangiitis (MPA), so treatment with steroids was started. During his hospital stay, he presented a complication of persistent melena with anemia, requiring a transfusion of 2 units of packed red blood cells (PRBCs), so gastrointestinal bleeding was suspected, and an upper and lower gastrointestinal tract endoscopy was performed without relevant findings. However, due to the persistence of overt bleeding originating in the small intestine, an endoscopic video capsule was performed, showing multiple deep, fibrin-covered, transverse ulcers in the distal duodenum and proximal jejunum, with circumferential margin involvement, predominantly on the folds, with evidence of scarce active bleeding, mucosa with congestion and severe edema, with no signs of necrosis (Figure 1). Given the previously described findings, the patient was diagnosed with enteric vasculitis. Due to his clinical condition, high surgical risk, and associated complications, continued systemic management was decided to control his disease and its gastrointestinal manifestation.
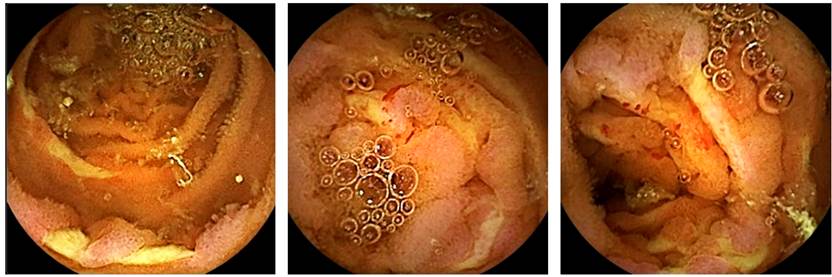
Figure 1 Multiple deep ulcers were observed, fibrin-coated with circumferential margin involvement and evidence of scarce active bleeding. Mucosal tissue with congestion and severe edema, without signs of necrosis.
During the hospital stay, he presented a torpid evolution because of persisting multisystemic compromise due to renal, pulmonary, neurological, and gastrointestinal involvement, requiring intensive care unit (ICU) treatment, where systemic steroid pulses and hemodialysis were initiated. The patient did not respond to multidisciplinary management and died 28 days after admission.
Discussion
Systemic vasculitis can affect blood vessels of all sizes, causing necrosis and inflammation. The disease’s extent and clinical course depend on the affected vessel’s size and location. Intestinal manifestations are rare and may be indiscernible from those of mesenteric ischemia caused by emboli or thrombosis2.
Clinical presentations may be local or diffuse in the gastrointestinal tract, resulting in nonspecific paralytic ileus, mesenteric ischemia, submucosal edema and hemorrhage or intestinal perforation, or stenosis. In addition, they may lead to anemia and hypoproteinemia, abdominal pain, and ulcers3.
Radiological findings in various types of vasculitis often overlap, so the possibility of this pathological process should be considered whenever mesenteric ischemia occurs in young patients, is seen in unusual locations, tends to involve the small and large bowel concomitantly, and is associated with systemic clinical manifestations.
Capsule endoscopy has now been used to explore the entire small bowel. In addition, it is especially useful in treating occult and overt obscure gastrointestinal bleeding. Furthermore, it should be performed in all patients with this diagnosis after upper endoscopy and colonoscopy with ileonoscopy, performed by an expert examiner, do not find the cause of the bleeding.
This case is a female patient diagnosed with systemic vasculitis associated with ANCA MPO (microscopic polyangiitis) confirmed through renal biopsy. Understanding its systemic signs and symptoms may suggest that the result found in the endoscopic capsule at the distal duodenum and proximal jejunum as a first indication might be compatible with enteric vasculitis.
Common causes of small bowel bleeding (SBB) include vascular lesions, ulceration secondary to Crohn’s disease (CD), non-steroidal anti-inflammatory drugs (NSAIDs), Meckel’s diverticulum, Zollinger-Ellison syndrome, vasculitis, small intestinal tumors (gastrointestinal stromal tumors [GIST], neuroendocrine tumor, adenocarcinoma or small bowel lymphoma), aortoenteric fistula, or jejunal diverticulitis4. Reports have shown that the diagnostic sensitivity of capsule endoscopy for diagnosing the source of bleeding in SBB ranges from 38% to 93%5. Capsule endoscopy is significantly better than single-balloon enteroscopy2,6, small bowel series7,8, enteroclysis9, and computed axial tomography (CT)10) for identifying small intestine lesions in patients with SBB.
Small bowel ulcers are another common abnormality detected through capsule endoscopy. Although most small intestinal ulcers detected through capsule endoscopies are due to CD or NSAID enteropathy, other causes include infection, ischemia, trauma, or vasculitis11.
GPA is a small- and medium-vessel vasculitis affecting the respiratory tract and kidneys. Necrotizing granulomatous inflammation characterizes the GPA clinical picture. Between 5% to 10% of cases have gastrointestinal involvement1. The clinical spectrum is highly variable, and vasculitis can be systemic or localized. Gastrointestinal tract involvement is often part of a systemic inflammatory process and is a well-recognized manifestation of small and medium-sized vessel vasculitis. Patients with gastrointestinal involvement often have abdominal pain, nausea, vomiting, diarrhea, or gastrointestinal bleeding. These gastrointestinal complications, when manifested, negatively affect the prognosis and indicate disease severity12.
Although GPA occurs with the same frequency in men and women, gastrointestinal manifestations are most often seen in men. Lesions include ulcers, submucosal edema, hemorrhage, paralytic ileus, mesenteric ischemia, intestinal obstruction, and intestinal perforation13.
The active inflammatory process located in the gastrointestinal tract in the course of GPA is a rare complication; however, it occurs and causes a serious threat to patients’ lives. Differential diagnosis should cover other inflammatory diseases, especially CD. The type of vasculitis most commonly affecting mesenteric vessels includes systemic lupus erythematosus (SLE), Takayasu arteritis, polyarteritis nodosa, Wegner granulomatosis, and Kawasaki disease. When timely diagnosed, it significantly influences the prognosis, as the rapid initiation of steroid therapy can change the course of the disease14. Side effects of immunosuppressive therapy should also be considered a cause of intestinal complications. Therapeutic strategies widely accepted for treating GPA are effective in treating patients with gastrointestinal involvement in the course of the disease, although some complications require surgical intervention13.











 texto en
texto en 



