1. INTRODUCTION
Heavy crude oils are becoming an increasingly important energy resource worldwide. These crudes are characterized by extremely low mobility due to high viscosity at reservoir conditions and high content of undesirable components, such as Asphaltenes, heavy metals and sulfur (Speight, 2010). Asphaltenes are recognized as the least comprehended compounds of the petroleum industry; however it is well-known that Asphaltenes exhibit a colloidal structure in crude oils (Nellensteyn, 1938; Pfeiffer & Saal, 1940). The great significance of Asphaltenes in the petroleum industry is through its negative impact on several operations, such as oil production, transporting, and refining. Flocculation and subsequent precipitation of Asphaltene colloid particles under changes in thermodynamic conditions (Temperature, Pressure, Composition) in the crude oil matrix cause serious problems in all steps of the production chain. From a practical point of view, clarifying how the colloidal structure relates to Asphaltene precipitation is considered to be one of the main challenges in this field because, in the light of such knowledge, effective mitigation strategies will be optimized in petroleum industry.
For many years it has been proposed that Asphaltene precipitation is the result of colloid aggregation (Leontaritis & Mansoori, 1987). Within this approach, the initial molecular aggregation occurs when the aromatic cores of Asphaltene molecules interact by Π- Π stacking to form colloidal nanoaggregates, which then further associate into larger structures called "fractal clusters" (Figure 1) (Yen, Erdman, & Pollack, 1961; Dickie & Yen, 1967; Mullins, 2010; Eyssautier et al., 2011; Hoepfner, Vilas Bôas FáVero, Haji-Akbari, & Fogler, 2013). It is generally agreed that Asphaltene aggregates are in the colloidal length scale between 5 and 1000 Å (Sheu, 2006). Typical aggregates, depending on the type of their intermolecular interactions, are assumed to associate according to certain general models reported in the literature as Diffusion-Limited Custer Aggregation (DLCA) or Reaction-Limited Cluster Aggregation (RLCA) (Weitz, Huang, Lin, & Sung, 1984; Meakin & Family, 1987).
Various of the properties of scientific and industrial interest (e.g. phase behavior, viscosity and microstructure) are strongly influenced by the physical dimension of colloidal particles (Hiemenz & Rajagopalan, 1997). Nowadays, the size distribution of Asphaltene monomers and aggregates continue to be discussed. Besides the size, establishing other parameters such as the shape, structural factors, and polydispersity of Asphaltenes is required to develop phase behavior models in order to predict the thermodynamic conditions where Asphaltene precipitation takes place. To quantitatively characterize these colloidal parameters, small-angle scattering techniques have been applied on Asphaltene systems as a function of thermodynamic conditions (Tanaka et al, 2003; Sheu, 2006; Headen, Boek, Stellbrink, & Scheven, 2009; Amundaraín, Chodakowski, Long, & Shaw, 2011).
In this work we used SAXS to characterize, for the first time, the aggregated structure of Colombian Asphaltenes in Toluene solutions at a dilute concentration regime from 0.01 to 1 % w/v. Results showed large changes in the scattering behavior of Asphaltenes as a function of its concentration, mainly at low values of the scattering vector q. The key point of this article is the way as fractal morphology of the large scale aggregates change from surface-to-mass structures as concentration is increased. Besides, we demonstrated that Asphaltenes are under an aggregate form as a continuum of fractals and nanostructures in the concentration range studied.
2. SAXS DATA ANALYSIS
In the analysis of the SAXS spectra some typical regions and laws are identified: i) in the low-q region, a Guinier or Zimm behavior is common, from which the radius of gyration can be calculated and ii) for intermediate and large q a scaling dependence of type I(q) ≈ q-d from which a structural model is chosen. The Radius of gyration (Rg) is defined as the root mean squared distance from the center of the mass of the particle to the position of all atoms and is given by the formula, according to Guinier approximation (Guinier & Fournet, 1955):
The radius of gyration, Rg, is obtained from the slope of a line given by In I(q) vs q2 in the q region where qRg ≤ 1. Similarly, the Zimm approach (Zimm, 1948), defined by equation 2, has been found to be more appropiated than the classical Guinier formalism because its validity can be extended for complex samples as Asphaltene aggregates up to qRg ≤ 2. The 1/I(q) versus q2 plot gives directly the radius of gyration Rg of the particles:
For Polydispersed Systems, Rg corresponds to the z-average-squared radius of gyration:
Where ni, Mi and Rgi2 are the number per unit of volume, the molecular weight and the radius of gyration of aggregate i, respectively. Rg can be used to quantify the particle dimensions once the structure of the particle is known. Otherwise, the intensity of scattered radiation I(q) by aggregates usually behaves as a power law over the intermediate range of the scattering vector, q :
Where d is the power-law exponent and can be measured directly from the negative slope of the linear region of a log-log plot of I(q) vs q (Sakai, Alba-Simionesco & Chen, 2011). The power-law exponent is directly related to the fractal dimension of the particles. If 1 < d < 3, then a mass-fractal scaling is observed and d=dm, where dm is the mass fractal dimension. If 3 < d < 4, then a surface-fractal scaling is observed and d=6-ds, where ds is the surface fractal dimension (Martin & Hurd, 1987; Avnir, 1989). Different power-law exponents, though diversely interpretable, have been elucidated from small-angle scattering measurements at the low-q region. For example, an exponent of -4 refers to a dense structure (compact aggregates) in the fractal organization and also represents a smooth surface of the fractal objects. On the other hand, power-law exponents that are less than 3 are related to mass fractal aggregates that tend to become more porous and larger in size (Martin, J. E. & Hurd, A. J., 1987; Narayanan, 2008).
3. EXPERIMENTAL SECTION
Sample Preparation.
Asphaltenes were extracted from a Colombian heavy crude oil following the ASTM D6560-12 standard method. Initially, a portion of the crude was mixed with n-heptane in a 1:25 ratio (g/mL) and the mixture heated under reflux. The precipitated Asphaltenes were collected on a filter paper and then Soxhlet-washed with hot «-heptane to remove the waxy substances co-precipitated. After removal of the waxy matter, the Asphaltenes were separated from the inorganic material by dissolution in hot Toluene in a Soxhlet extractor. Finally, the extraction solvent was roto-evaporated and Asphaltenes were weighed, ground and stored. The yield of Asphaltenes (precipitate) was 15% w/w.
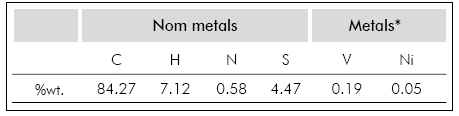
Elemental analysis for C, H, N and S was carried out using a Thermo Finnigan elemental analyzer. Metal content (V, Ni) was analyzed using a XRF Bruker S8 TIGER spectrometer (Karlsruhe, Germany) with an X-ray tube with rhodium target (Kα1 0.6136 Å) on finely ground powder of Asphaltenes. Results are presented as percent by weight (%w.) for each element in Table 1.
A 1% w/v Asphaltene solution was prepared by dispersing the appropriate amount of solid Asphaltenes in the solvent (Toluene) by sonication and subsequent magnetic stirring during 24 h. Then, less concentrated solutions of Asphaltenes (0.1 and 0.01% w/v) were prepared from the 1% solution by successive dilutions. All samples used in the SAXS studies were allowed to stand overnight before any experiment.
Small-Angle X-ray Scattering
The SAXS work presented here was performed using the Small Angle X-Ray facility at the Laboratory of Universidad Autónoma Metropolitana (Iztapalapa, Ciudad de México, México). The analysis was conducted by loading 2 mm quartz capillaries with aliquots of the 1, 0.1, and 0.01 w/v % Asphaltene solutions in Toluene.
Data were collected at the laboratory conditions, room temperature and atmospheric pressure, by using a high resolution small-angle X-ray scattering, Xeuss (SAXS) equipment from XENOCS, with radiation Kα(Cu) with a wavelength of (λCu = 0.1541 nm) and a detector in two dimensions (2D, Pilatus 300 K). We used a sampleto- detector distance of 1.14 m, covering a q (scattering vector) range from 7.7 x 10-3 to 0.25 Å-1.
Fit2D® software was used for image analysis and processing (Hammersley, 1997). The total scattered intensity from a liquid solution (Asphaltene-solvent) is a contribution of the camera background, cell or capillary background, and the solvent signal. To obtain the scattering arising from the Asphaltene particles alone, a subtraction must be performed: data from the sample minus data from the pure solvent. The effect of background scattering was evaluated using SAXS intensity for empty sample tubes and solvent-filled tubes. In this way, all dispersion data from each sample were corrected before being converted to an absolute scale [cm-1].
4. RESULTS AND DISCUSSION
SAXS technique was used to obtain structural information of Asphaltene aggregates in model systems (Asphaltene solutions in Toluene) as a function of its concentration. Figure 2 shows the SAXS profiles for all the solutions at 25 °C as a double-logarithmic plot of I(q) versus q. A qualitative analysis of the scattering data in Figure 2 is enough to conclude that all of the samples are colloidal systems with aggregates having polydisperse sizes. Although solutions at 0.01 and 0.1 w/v % present very similar scattering curves, the derived structural parameters are slightly different when both samples are compared as will be discussed later. In these spectra two regions can easily be distinguished: a first one corresponding to small q-values (q < 3 x 102 Å 1) in which the scattered intensity is a fastly decreasing function of q (or power-law scattering), and a second one at large q-values (q > 3 x 102 Å 1)where the scattered intensity shows a plateau (or Guinier region) followed by a slight decrease of the intensity at higher q. Similar scattering profiles have been obtained previously in small angle scattering studies for some concentrated Asphaltene solutions and crudes (Espinat et al., 1998; Roux, Broseta, & Deme, 2001; Sheu, 2006; Gawrys, Blankenship, & Kilpatrick, 2006; Zielinski, Saha, Freed, Hürlimann, & Liu, 2010).
In a different way, the SAXS profile for the 1% Asphaltene solution just exhibits a power-law scattering behavior in which the scattered intensity decays as q-d, and therefore, only the structure parameter d can be extracted from the data. This particular scattering behavior is typical of particles with fractal dimensionality in which the power-law exponent (d) provides an insight into the highly complex structural morphology giving rise to the scattering (Beaucage, 1996). We will discuss this behavior at the end of this section.
Low-q region (q < 3 x 10-2 Å-1). According to the literature, a large upturn in the scattering intensity at very low-q is related to density fluctuations of small aggregates or coexistence of large aggregates with smaller ones in the solution (Espinat, 1998). In our opinion, the power-law scattering at low-q for all samples indicates the presence of large scale aggregates with fractal dimensionality whose radius of gyration could not be established with the present experimental setup (the Guinier region was not accessible in this q range). This q-dependence is proof of structures are much larger than 1/qmin (i.e., Rg > 13 nm).
As mentioned earlier, the fractal dimension d is related to the nature of the structures present in the medium. If these structures are interacting in the solvent through any association process, they could form amass fractal structure or they could have fractal surfaces. Our measurements showed that for the 0.01 and 0.1 % (w/v) Asphaltene solutions, the power-law scattering is derived from a surface-fractal scaling characterized by fractal dimensions around 2.8 and 2.6, respectively. However, forthe most concentrated solution (1% w/v), the fractal dimension dropped off to ~1.9 which corresponds to a mass-fractal scaling. The fractal dimension of aggregates was estimated from the fitting of power-law exponents in the spectra and the results are given in Table 2. Results show a decreasing of the fractal dimension as Asphaltene concentration is increased. This result is attributed to morphological changes of the Asphaltene aggregate structures due to a singular surface-to-mass fractal transition probably controlled by the increase of the inter-particle interactions. We have to emphasize that this type of fractal evolution has not been previously reported for Asphaltenes in solvents. Also, the decrease in the fractal dimension indicates that the aggregates formed at the highest Asphaltenes concentration (1% w/v) have a substantially less dense structure, whereas aggregates formed at lower concentrations (0.01 and 0.1 w/v %) are more compact in its fractal organization. Such concentration-dependent structural effects are summarized in Figure 3.
High-q region (q > 3 x 102 Å1). We have observed in Figure 2 that in this region the intensity levels drop off to a (quasi)plateau followed again by power-law behavior at the largest q. These features are due to the presence of small heterogeneities with a size in the order of a few nanometers corresponding to the primary Asphaltene aggregates known as "nanoaggregates". Note that there are not robust changes in the scattering intensities when Asphaltene concentration raises up to 0.1% w/v, indicating that this concentration is not sufficiently high to modify the aggregation state of nanoaggregates. We suggest that this specific behavior is because these small aggregates behave as independent particles (i.e., uncorrelated objects) probably due to packing constraints, however, when the Asphaltenes concentration is increased up to 1% w/v, the scattering intensity is strongly modified because of nanoaggregates are increasingly in contact favoring some type of interaction.
As a first step in the data treatment, we have used the Zimm approximation (Eq. 2) from a lower limit where scattering from large scale aggregates comes to be significant to an upper limit defined by qmaxRg ≤ 2. From this analysis, the scattering intensities at q = 0 and the radii of gyration (Rg) were calculated. However, forthe 1% Asphaltene solution, the extraction of these structural parameters was rendered difficult due to the quasi-plateau corresponding to the Guinier region overlaps with the intense scattering at low q. The fitted parameters are given in Table 3. Our results are not completely in agreement with previous studies, where higher radii of gyration for Asphaltene aggregates in solvents were obtained. For example, Roux, J. N., Broseta, D. & Déme, B., (2001) studied the structure of Asphaltene aggregates in Toluene solutions by SANS (small-angle neutron scattering) observing radii of gyration larger than 30 Å in a dilute concentration regime (from ~0.3 to ~3%) at 20 °C. (Morimoto, Morita, Takanohashi, & Nishikawa, 2013) also reported SAXS measurements on Asphaltene solutions in Toluene finding aggregates with radii of gyration between ~30-60 Å for concentrations as low as 20 mg/L. Other reports, on the other hand, showed a range of ~20-23 Å for Rg from SAXS measurements on Asphaltene/Toluene systems (Sheu, 2006; Yarranton et al. 2013). The discrepancies in the gyration radii derived from several small-angle scattering measurements could be ascribed to the diverse compositions of Asphaltenes.
In a second step, Asphaltene nanoaggregates were assumed to be spherical particles. Several small angle scattering studies of Asphaltenes from different geological sources have reported that Asphaltene colloids form more-or-less spherical aggregates (Sheu, 1998; Sheu & Acevedo, 2001). Therefore, it is acceptable to use a spherical shape to derive the particle dimension from SAXS data. In this sense, the average radius R of the sphere can be calculated from the following equation:
Where Rg is the radius of gyration obtained from the Zimm analysis. R values are also given in Table 3. As one can see, the results confirm that Asphaltenes assemble to form colloidal structures conforming to the hierarchical aggregation model proposed by Mullins (Mullins, 2010) in which particle dimensions range between 50-100 Å for nanoaggregates. Additionally, we proved the Asphaltenes nanoaggregates are fully formed in a solvent such as Toluene at concentrations similar to the previously reported "critical nanoaggregate concentration" (CNAC ~100 mg/L) (Andreatta, Bostrom, & Mullins, 2005)
As we mentioned at the beginning of this section, the scattering curve for 1% Asphaltene solution is characterized by the appearance of a crossover between two successive power-law regimes which correspond to mass fractals. Evidence for succession of power-law regimes on Asphaltenic Systems from SANS data have been earlier reported (Headen, 2009). This type of scattering is commonly related with a complex multilevel structure, i.e. a large-scale particle composed of smaller sub-particles. From the values of the scattering exponents of both power-law regimes (see Figure 4), we have inferred that this complex structure is formed by one mass fractal embedded into another mass fractal. However, a more accurate description of this fractal system should also involve the size polydispersity of the aggregates. On the other hand, as the mass fractal dimension decreases the structures appear sparser until at d= 1 (the lowest possible value for a 'connected' fractal) the object essentially becomes a line (Martin & Hurd, 1987). According to the mass-fractal morphologies defined by Beaucage (1996), the large-scale structure may be assumed as a multi-particle diffusion limited aggregate, whereas the substructures may be described like randomly oriented rods. Thus, we suggest that structures present in the 1% Asphaltene solution are low-dimensional subunits that form a large-scale structure by some growth process.
5. CONCLUSIONS
Asphaltene-Toluene solutions were investigated by SAXS as a function of Asphaltene concentration. Scattering curves were separated in two regions to be analyzed: low-q values and high-q values. In the low-q range studied, Asphaltenes displayed power-law scattering indicating the presence of large aggregates with fractal properties. We demonstrated the structural evolution of these large heterogeneities as concentration was increased. The most consistent interpretation of this evolution is the modification of the aggregation state of Asphaltenes due to some fractal association processes, resulting in the transition from surface aggregates to mass aggregates. Besides, Asphaltenes showed to form loosely structured aggregates (porous or diffuse) at the highest concentration (1% w/v), whereas aggregates that formed at lower concentrations (0.01% and 0.1% w/v) are denser.
At high-q values, we proved that in a solvent such as Toluene, large scale aggregates coexist with Asphaltene nanoaggregates in the order of a few nanometers at relatively low concentrations (≤ 1% w/v), i.e. that not all nanoaggregates are part of the larger aggregates. These results are in agreement with the general view of the modified Yen model. On the other hand, the scattering intensity for the 1% Asphaltene solution is described by the presence of two successive mass fractal regimes with two different power law exponents (q-1.9 → q-1.0) which is consistent with a multi-phase system of mass fractals. Finally, we expect that Asphaltenes in Colombian heavy crude oils or the various industrially processed petroleum fractions also exhibit a similar concentration dependence from which the hydrocarbon phase behavior can be explained.