THERORETICAL AND CALCULATIONS
Physicochemical properties (PCPs) of the liquid mixtures at various temperatures are required in many industrial process design and the infinite number of solvent compositions makes the experimental determination of PCP of all possible combination too difficulte. In such cases employing mathematical models may provide accurate tools for prediction of un-measured PCP data by using interpolation technique.
Vankar and Rana [1] reported the experimental density, viscosity and ultrasound velocity of binary mixtures of 3-bromoanisol + methanol at 303.15, 313.15 and 323.15 K along with the calculated molar volumes of the mixtures using the generated density data. They also provided some computational results for the generated PCP data. The aim of this communication is to point out the capabilities of the Jouyban-Acree model [2] for representing the experimental PCPs data using a unified version.
The excess PCPs of 3-bromoanisol + methanol mixtures at various temperatures were represented using a Redlich-Kister equation at each temperature by Vankar and Rana [1]. Although this representation could provide some useful information regarding PCP data, however, there is no possibility to predict the PCP data at other temperatures of interest which is required in many industrial applications. To provide such a capability, one may use an alternative model which was reported in an earlier work [2]. The model is:
where PCP m,T is the numerical values of the PCP of the mixture, x 1 and x 2 are the volume (weight or mole) fractions of liquids 1 and 2 in the binary mixture, α, β and J terms are the model constants.
The constants of equation (1) could be calculated via regressing ln PCP
1T
against T and ln PCP
2T
against
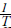 using least square analyses, then employing the α and β constants of the van't Hoff equation, the / terms could be calculated using regressing in
using least square analyses, then employing the α and β constants of the van't Hoff equation, the / terms could be calculated using regressing in
 [2]
[2]
The two first terms represent the PCP of neat liquids 1 and 2 at temperature of T, and the term
 represent the non-ideal mixing behaviour of the binary liquid mixture [2]. The accuracy of equation (1) to represent PCP data was evaluated by computing root mean square deviation (RMSD) and mean percentage deviations (MPD) by:
represent the non-ideal mixing behaviour of the binary liquid mixture [2]. The accuracy of equation (1) to represent PCP data was evaluated by computing root mean square deviation (RMSD) and mean percentage deviations (MPD) by:
and:
where N is the number of data points in each set.
The accuracy of the proposed model was compared with that of the Redlich-Kister equation which was used by Vankar and Rana [1]. The equation respective is [3, 4]:
in which the excess PCP (PCP Ex ) is calculated using:
at each temperature of interest. The model constants of equation (4) could be obtained via two types of least square analyses; 1) by regressing PCP
M
-(XI_PCPI+X
2
PCP
2
) against x1x2, x1x2 (x1-x2) and x1x2 (x1-x2)2 using a no intercept least square analsyis and 2) by regressing
 against (x1-x2) and (x1-x2)2 using a classical least square analsyis and power of (x1-x2) could be increased as much as the regression gives significant model constants.
against (x1-x2) and (x1-x2)2 using a classical least square analsyis and power of (x1-x2) could be increased as much as the regression gives significant model constants.
Table 1 listed the model constants and the MPD and RMSD values for the investigated PCPs. As MPD and RMSD values reveal, the model provided excellent correlation between mixture composition and temperature with ln PCP m,T and one may use the trained model for prediction of the PCP m,T at any mixture composition and temperature of interest without requirement of any further experimental effort. To show such a capability, PCP data of 3-bromoanisol and methanol at the highest and the lowest temperatures were used to calculate α and β values and the PCP data in x¡= 0.327, 0.5644 and 0.7446 at 313.15 K were used to calculate J terms of equation (1), then the rest of data points were predicted using the trained models in which the obtained MPDs were 1.1%, 1.6%, 0.7% and 0.4%, respectively for density, viscosity, speed of sound and molar volume data. To further investigate the applicability of the trained model using a minimum number of seven PCP data, the reported density, viscosity, molar volume and speed of sound data for the binary mixtures of 3-bromoanisole and methanol at 293.15 K [3] were also predicted, and then the MPD values for the predicted PCPs were 1.3%, 8.5%, 0.6% and 0.4%, respectively.
Table 1 Model constants, mean percentage deviation (MPD) and root mean square deviations (RMSD) of equation (1) for the investigated physicochemical properties of 3-bromoanisol + methanol mixtures.

a Not significant (p>0.10).
We tried to re-calculate the viscosity (and other PCPs) data of 3-bromoanisol + methanol at 303.15 K by using the reported model constants (Table 3 of Ref. [1]) of equation (4), but the re-calculated model constants and standard deviation value were slightly different from reported values and no more details were provided by Vankar and Rana [1]. To check the calculations, the model constants of equation (4) were re-calculated using two least square methods and the results for four investigated PCP data are listed in Table 2. There were significant differences between MPDs and RMSDs of viscosity and ultrasound velocity (paired t-test) and no significant differences for density and molar volume data when they analyzed using a no interceppt and classical least square analyses. According to MPD and RMSD values reported in Tables 1 and 2, equation (4) provides slightly more accurate calculations when compared with equation 1, however, it requires minimum number of experimental determination at each temperature to be able to predict the rest of PCP data at other mixture comositions. Equation (1) does not require any more experimental PCP data for 3-bromoanisol + methanol mixtures and is able to simulate the PCP data at any mixture composition and temperature of interest. In other words, it is more practical model in industrial applications. It should be added that the accuracies of both equations (1) and (3) are acceptable when the experimental unicertianity values are considered.




















