Introduction
Preserving forage as silage is a common strategy used by farmers to feed animals during periods of low forage availability (Brito et al., 2020; Haselmann et al., 2020). Preservation of the aerial parts of cassava (Manihot esculenta, Crantz) in the form of silage could maximize the use of this resource (Fluck et al., 2017), considering that this material is usually discarded on the field at the time of root harvesting (Nunes irmão et al., 2008).
The aerial part of cassava presents, on average, 14.0 to 19.0% crude protein, 61.1% to 63.6% neutral detergent fiber, 6.7 to 8.3% ethereal extract, and 50.0 to 60.0% total digestible nutrients (Souza et al., 2011). These characteristics make the aerial part of cassava a low-cost forage alternative, which can be included in ruminant nutrition in its in natura hay or silage form (Souza et al., 2018; Nascimento et al., 2021).
Although it is possible to preserve the aerial part of cassava in the form of silage, as demonstrated in previous studies (Fluck et al., 2017; Oliveira et al., 2018; Nascimento et al., 2021), its low dry matter content (Modesto et al., 2008) may result in undesirable fermentation and increased effluent losses during silage, leading to a reduction in final silage quality. A possible way to circumvent this problem consists in mixing the silage of aerial part of cassava with dehydrated byproducts of the agro-industry.
On the other hand, viticulture is one of the most extended agro-economic activities worldwide, with over 60 million tons produced globally each year. An important part of the production of grapes is destined to the production of wine, which is a relevant traditional activity in several countries (Teixeira et al., 2014). Production of wines and juices involves the generation of large amounts of by-products, mainly organic waste, wastewater, greenhouse gas emissions, and inorganic waste (Musee et al., 2007). Grape pomace is a residue obtained from the grape pressing process, constituting on average 20% of the total used for the production of wine and juice (Teixeira et al., 2014; Flores et al., 2020). Inclusion of this residue in animal diets can reduce the environmental impact of this agricultural activity.
Dehydrated grape marc (Vitis vinifera) in silage form can be used as a feedstuff for ruminants (Flores et al., 2020). It has, approximately, 17.0% crude protein, 40.0% non-fibrous carbohydrates and 42.0 to 62.0% neutral detergent fiber (Basalan et al., 2011; Santos et al., 2014; Guerra-Rivas et al., 2016; Flores et al., 2020). Furthermore, depending on the dehydration process, the byproduct can have dry matter levels of 60 to 80%. Thus, using this by-product as an additive can elevate silage dry matter content to a desirable concentration during the fermentative process.
It should be noted that the proper amount of grape marc to be added to silage should be evaluated due to its high fiber values, which can negatively affect nutrient digestibility (Basalan et al., 2011; Guerra-Rivas et al., 2016). Therefore, our hypothesis was that adding 10% grape marc dehydrated to a silage from aerial parts of cassava would improve the chemical composition, degradability, and production of in vitro gases from the silage.
Materials and methods
Location of the experiment
The study was conducted at the Agricultural Sciences campus of Universidade Federal do Vale do São Francisco, municipality of Petrolina - Pernambuco, Brazil. Geographic coordinates: Latitude 9° 09' South and longitude: 40º 22' West. According to the Köppen classification, the climate in the region corresponds to a Bswh type (hot and dry region).
Experimental design and conduct of the test
The experimental design used was completely randomized and the treatments consisted on silage of the aerial part of cassava without additive (CAP), and with 10% dehydrated grape marc (DGM) in the natural matter, as an additive (CAP+DGM), with 14 repetitions per treatment. The upper third of the aerial part of cassava was used at approximately 12 months of age. The material was minced to approximately 5 mm in a stationary forage chopper. The DGM was the result of the extraction of natural grape juice (Cooperativa Agrícola Nova Aliança). This residue was disposed over an open area to the sun for five days and rotated daily until reaching approximately 80% DM. After this period, the residue was ground in a stationary forage chopper with 3-mm sieve.
The samples in natura from the aerial part of cassava and DGM were collected to determine dry matter (DM), crude protein (CP), ether extract (EE), neutral detergent fiber (NDF) and acid detergent fiber (ADF), as described in AOAC (2000). For the estimation of total digestible nutrients (TDN) we used the following equation proposed by Cappelle et al. (2001): TDN (%) = 99.39-0.7641*NDF (Table 1).
Table 1 Chemical composition of in natura ingredients used for silage production
| DM (%) | CP | EE (%) | NDF (%) | ADF (%) | TDN (%) | |
| Cassava aerial part | 29.61 | 13.93 | 5.90 | 56.08 | 31.11 | 56.54 |
| Dehydrated grape marc | 82.79 | 12.52 | 7.48 | 62.85 | 35.98 | 51.37 |
DM: Dry matter; CP: crude protein; EE: ether extract; NDF: neutral detergent fiber; ADF: acid detergent fiber; TDN: total digestible nutrients.
Twenty-eight cylindrical PVC (Tigre®, Rio Claro, SP, Brazil) silos with 10-cm diameter and 50-cm length were used, with PVC caps equipped with a Bunsen-type valve to allow escape of fermentation gases. Compaction of the material was performed with wooden sockets and sealing with PVC covers (sealed with adhesive tape). The material was compacted to a specific mass of 650 kg/m3. Determination of chemical composition and in vitro evaluation of silages were carried out in triplicates.
Chemical composition and pH of silages
For silage evaluation, the silos were opened after 30 days of fermentation. The contents of the upper and lower three centimeters of each silo was disregarded and the rest of the contents were homogenized (initial, intermediate and final). After this procedure, silage samples were collected, packaged in plastic bags, and sent to the laboratory to determine DM, CP, EE, NDF and TDN in an analogous way to the evaluations performed to in natura material.
The pH of the silage was determined after dilution of 9 g of fresh silage into 60 mL distilled water; after 30 minutes of rest, an electrode was introduced in the solution waiting 15 seconds for stabilization of each sample (Jobim et al., 2007).
In vitro degradability
Evaluation of in vitro degradability followed methodology proposed by Tilley and Terry (1963), modified and adapted to Ankom Daisy System (Ankom Technology Corp., Macedon, NY, USA), described by Holden (1999). The procedure was performed by incubating samples of 0.500 g of silages in polypropylene tissue bags (5x5 cm, 100 µm). The samples were incubated in jugs with Bunsen valve using 1.600 mL of buffer solution composed of solutions A and B, and 400 mL of ruminal inoculum from sheep.
Incubation periods were 2, 6, 12, 24, 48, 72 and 96 h. The bags were positioned in different times and removed at the same time. After removal, bags were washed with cold water until the water ran clear. Later, the bags were dried in a forced-air circulation oven at 55 °C for 72 h, and then weighed. The DM content in the waste was determined according to AOAC (2000).
In vitro degradation kinetics was calculated using the nonlinear model proposed by Orskov and Mcdonald (1979), defined as:
P = A + B*(1-Exp(-C*t))
Where: P is the percentage of degraded substrate up to time t; A represents the rapidly soluble fraction; B is the fraction potentially degraded; C is the degradation rate of the B fraction, and t is the incubation time. With the estimation of the model parameters, it was possible to calculate the potential degradability (A + B), the non-degradable fraction (100 - (A + B)), and the effective degradability (ED), which was obtained from the following expression:
ED = A + B*C/(C+K)
Where, A, B and C are the same parameters as the model proposed by Orskov and Mcdonald (1979), and K is the rate of passage of solids in the rumen: 2, 5 and 8% per hour (AFRC, 1992).
Cumulative in vitro gas production
The in vitro semiautomatic gas production technique proposed by Maurício et al. (2003) and modified by Menezes et al. (2015) was applied to perform cumulative gas production evaluations. For each silage, three samples were incubated using ruminal fluid from sheep (Theodorou et al., 1994). The incubation solution was composed of 160 mL solution, 80% (128 mL) buffer and minerals, and 20% (32 mL) ruminal inoculum manually introduced using a graduated syringe in glass vials, previously washed and dried in an oven with forced ventilation and one gram of sample added. The ruminal inoculum, used in the in vitro degradation and gas production evaluations, was collected from ewes that were fed minced forage and concentrate when the ruminal fluid was collected, in a 70:30 proportion of roughage/concentrate.
In addition to the vials containing silage samples, two other vials contained only the inoculum and buffer solution for the purpose of measuring possible pressures that were not linked to the diets. The incubation solution was prepared as described by Theodorou et al. (1994) using cysteine-HCl as a reducing agent (Mould et al., 2005). After introducing the incubation solution and samples, CO2 was manually injected for seven seconds into each flask, later closed, and introduced into the forced-air ventilation oven at 39 °C during 96 hours for incubation.
To obtain the parameters of gas production kinetics, the bicompartmental logistic model proposed by Schofield et al. (1994) was used:
Y = A/(1+exp(2-4*B*(Lag-t))) + D/(1+exp(2-4*E*(Lag-t)))
Where: Y is the cumulative production of gas at time t (mL g/DM); A and D represent the volume of gas produced by the degradation of non-fibrous carbohydrates and soluble compounds, and fibrous carbohydrates, respectively; B and E are the respective degradation rates of non-fibrous carbohydrates and soluble compounds and fibrous carbohydrates; and Lag is the time (hours) of particle colonization by bacteria (lag time).
Statistical analysis
The parameters of the Orskov and Mcdonald models (1979), and bicompartmental logistics, were estimated using the modified Gauss-Newton method using the SAS NLIN procedure (SAS University Edition -Version 12-, SAS Institute Inc. Cary, CA, USA). The maximum number of iterations used was 100.
All data were subjected to analysis of variance using a completely randomized design by the PROC GLM command, and the averages were compared by the Fischer test, analyzed by the SAS statistical package (SAS University Edition -Version 12-, SAS Institute Inc. Cary, CA, USA). A significance level of 5% was adopted in all statistical analyses.
Results
Chemical composition and pH of silages
Addition of DGM did not change CP, EE, NDF, nor the TDN levels in the silage (Table 2). However, DGM promoted an increase in DM contents of the silage. Likewise, the pH of silage plus DGM was 15.79% lower than the value observed in silage without DGM (Table 2).
Table 2 Chemical composition of cassava aerial part silage (CAP) with or without dehydrated grape marc (DGM).
| Variables | Silage | SEM | p-Value | |
|---|---|---|---|---|
| CAP | CAP + DGM | |||
| DM (%) | 30.31b | 32.61a | 0.360 | <0.001 |
| CP (%) | 14.75a | 14.75a | 0.130 | 0.980 |
| EE (%) | 5.62a | 5.38a | 0.160 | 0.290 |
| NDF (%) | 53.98a | 55.34a | 0.440 | 0.060 |
| TDN (%) | 57.10a | 58.14a | 0.370 | 0.060 |
| pH | 4.75a | 4.00b | 0.110 | <0.001 |
DM: Dry matter; CP: crude protein; EE: ether extract; NDF: neutral detergent fiber; TDN: Total Digestible Nutrients; SEM: standard error of the mean. Different letters within the same line differ at 5% probability by the Fischer test (p<0.05). SEM: standard error of the mean.
In vitro degradability
The rate of DM degradation did not change with the addition of DGM. However, the silage that received DGM presented higher degradability coefficients of soluble and potentially degradable fractions, which led to higher values of potential and effective degradability of this silage (Table 3). On the other hand, the non-degradable fraction was higher in the silage composed only by aerial parts of cassava.
Table 3 In vitro degradability parameters of dry matter of cassava aerial part silage (CAP) with or without dehydrated grape marc (DGM).
| Degradability coefficients | Silage | SEM | p-Value | |
|---|---|---|---|---|
| CAP | CAP + DGM | |||
| Soluble fraction (%) | 13.84b | 15.06a | 0.334 | <0.001 |
| Potentially degradable fraction (%) | 31.46b | 37.13a | 1.245 | <0.001 |
| Degradation rate (%/h) | 0.045 | 0.044 | 0.004 | 0.0218 |
| Non-degradable fraction (%) | 54.71a | 47.82b | 1.452 | <0.001 |
| Potential degradability (%) | 45.29b | 52.18a | 1.362 | 0.0341 |
| Effective degradability (%) | 24.23b | 27.80a | 0.631 | 0.0172 |
Different letters within the same line differ at 5% probability by the Fisher test (p<0.05). SEM: standard error of the mean.
Cumulative in vitro gas production
Inclusion of DGM had no effect on the volume of gas (mL) produced from degradation of non-fibrous (A) and fibrous (D) carbohydrates (Table 4). There was also no difference in the values of non-fibrous (B) and fibrous (E) carbohydrate degradation rates. However, the inclusion of DGM promoted a reduction in particle colonization time (Lag time) and increased cumulative gas (Y) production.
Table 4 In vitro gas production kinetics parameters of cassava aerial part silage (CAP) with and without dehydrated grape marc (DGM).
| Parameters | Silage | SEM | p-Value | |
|---|---|---|---|---|
| CAP | CAP + DGM | |||
| Y (mL/g DM) | 57.82b | 62.03a | 0.750 | 0.001 |
| A (mL g/DM) | 31.29 | 30.18 | 1.280 | 0.740 |
| B (h) | 0.066 | 0.061 | 0.002 | 0.310 |
| Lag time (h) | 10.01a | 6.74b | 0.581 | <0.001 |
| D (mL g/DM) | 26.52 | 31.86 | 1.621 | 0.180 |
| E (h) | 0.021 | 0.021 | 0.001 | 0.640 |
Y = cumulative gas production at time t (extent of degradation); A = gas volume (mL g/DM) produced by degradation of non-fibrous carbohydrates and soluble compounds; B = degradation rate of non-fibrous carbohydrates and soluble compounds (h); Lag = time taken by the bacteria to colonize particle (h); D = gas volume (mL g/DM) produced by degradation of fibrous carbohydrates; E = fibrous carbohydrate degradation rate (h); SEM: standard error of the mean. Distinct letters on the same line differ at 5% probability by the Fischer test (p<0.05).
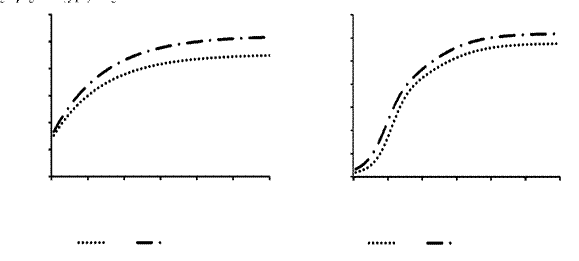
The differences observed in DM degradability parameters (Table 3) and in vitro gas production kinetics (Table 4) promoted changes in the respective curves projected from the parameters estimated by the Orskov models (Figure 1A) and bicompartmental logistic (Figure 1B).
Discussion
Addition of 10% DGM was not sufficient to increase or reduce CP, EE, NDF and TDN levels of silage, which can be attributed to the fact that the DGM did not present a discrepant difference in the concentration of these nutrients in relation to the aerial part of cassava (Table 1). Moreover, when silage processing is performed according to established basic procedures, no significant change in the concentration of these nutrients in silage is detected (Zardin et al., 2017; Santana et al., 2019).
However, inclusion of DGM promoted an increase in DM of silage, as this residue went through the drying process with the objective of raising DM to approximately 80%. The aerial part of cassava has a high moisture content (Modesto et al., 2008; Fluck et al., 2017), which may potentiate clostridia activity and cause undesirable fermentation, significantly increasing losses in nutritional quality due to proteolysis (Liu et al., 2011; Xie et al., 2012; Wilkinson & Muck, 2019). Therefore, the use of absorbent additives, such as agro-industrial waste, can be an alternative to improve the fermentation process of wet materials (Gurgel et al., 2019), such as the aerial part of cassava.
The higher DM content observed in CAP + DGM silage reflected in better compaction and, consequently, in lower silage pH (Table 2). In addition, the lower the DM content of ensiled forage, the greater the resistance that the forage mass has to the pH decrease (Jobim et al., 2007). The pH value of silage is one of the most important indicators of quality (Wilkinson & Davies, 2013), in this sense, the pH values found in DGM additive silage are satisfactory for the conservation of forage, since pH between 3.6 and 4.2 are considered ideal (Mcdonald et al., 1991).
The higher degradability coefficients of soluble, potentially degradable, potential and effective fractions observed in CAP + DGM silage resulted in lower non-degradable fraction of this silage, suggesting that DGM may have provided more substrates to micro-organisms and elevated the degradation of the incubated material. It should be noted that although DGM has high fiber contents (Flores et al., 2020), which could hinder access by microorganisms, inclusion of 10% of this additive was not sufficient to change the fibrous fraction of silage (Table 2).
Agroindustry residues are rich in starch, simple sugars, and pectin, the latter being a structural carbohydrate of rapid ruminal degradation (Ítavo et al., 2020). Pectin is a non-starch carbohydrate considered as soluble fiber readily available, despite being part of the cell wall. Considering its better-quality fiber, agroindustry residues such as DGM are alternative additives that can improve ruminal degradability of silages.
High cumulative production of gas in CAP + DGM silage was observed. Gas production potential of total carbohydrates was prolonged due to possible increase of fermentable substrates by DGM addition. The shorter particle colonization time (lag time) shows, in parts, that DGM addition increased substrate supply to microorganisms. Lag time is related to the presence of readily fermentable substrates and physical and chemical properties of food, capable of facilitating microbial fermentation (Schofield et al. 1994). In addition, there is a direct relationship between food degradability and cumulative gas production (Leal et al., 2020, Santana et al., 2020).
Addition of DGM to silage of aerial parts of cassava proved to be a promising alternative. The DGM promoted an increase in DM levels and reduction of silage pH, improving fermentative parameters. Therefore, the hypothesis that adding 10% DGM to silage from aerial parts of cassava would improve the chemical composition, degradability and production of in vitro gases from silage was confirmed by the results observed.
In conclusion, addition of 10% dehydrated grape marc in the silage of aerial parts of cassava is recommended to reduce pH, increase dry matter contents, and improve parameters of silage fermentation kinetics.