Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista Colombiana de Entomología
Print version ISSN 0120-0488
Rev. Colomb. Entomol. vol.39 no.2 Bogotá July/Dec. 2013
Effects of insecticides used in corn on immature stages of Trichogramma atopovirilia (Hymenoptera: Trichogrammatidae)
Efectos de insecticidas usados en el maíz sobre los estados inmaduros de Trichogramma atopovirilia (Hymenoptera: Trichogrammatidae)
JADER BRAGA MAIA1,4, GERALDO ANDRADE CARVALHO2,4, RODRIGO LOPES DE OLIVEIRA3,4, OLINTO LASMAR5 and MARIA ISABELLA SANTOS LEITE6
1 Ph. D. in Entomology, maiajader@yahoo.com.br. Corresponding author.
2 Ph. D. Associate Professor, gacarval@den.ufla.br.
3 Master Student in Entomology. rodrigo_lopes_oliveira@yahoo.com.br.
4 Department of Entomology, Federal University of Lavras/UFLA. Cx. Postal 3037, CEP: 37200-000 - Lavras, MG.
5 M. Sc. in Entomology at the Department of Entomology in Federal University of Lavras. University Estadual Paulista "Jülio de Mesquita Filho" - Campus de Jaboticabal- SP., CEP: 14884-900. lasmar84@yahoo.com.br.
6 M. Sc. Entomology at the Department of Entomology in Federal University of Lavras. Monsanto Brazil. maryisabbella@yahoo.com.br.
Received: 21-Nov-2012 - Accepted: 23-Oct-2013
Abstract: The chemical control of Spodopterafrugiperda (Lepidoptera: Noctuidae) is difficult because caterpillars live inside the corn (Zea mays) cob and they do not come into contact with insecticides. Research on the use of parasitoids of the genus Trichogramma (Hymenoptera: Trichogrammatidae) has contributed to advances in alternatives for the control of this pest. However, there is little information on the effects of pesticides used in corn on these parasitoids. Therefore, the objective of this study was to evaluate the physiological selectivity of insecticides for Trichogramma atopovirilia in the egg-larval, pre-pupal and pupal phases. The bioassays were carried out under controlled conditions, exposing eggs of Anagasta (= Ephestia) kuehniella (Lepidoptera: Pyralidae) to parasitism for a 24-h period. The insecticides were sprayed using a Potter tower on host eggs containing the parasitoids in the egg-larval period, pre-pupal and pupal phases. The insecticides evaluated in g a. i. L-1 were: imidacloprid/B-cyfluthrin (Connect 100 SC - 0.033), chlorfenapyr (Pirate 240TM SC - 0.6), chlorpyriphos (Astro 450 EW - 0.75), novaluron (Rimon 100 CE - 0.05), spinosad (Tracer 480 SC - 0.16) and triflumuron (Certero 480TM SC - 0.048). Water was used as a control. According to the results, the products were classified in the following toxicological classes for T. atopovirilia: imidacloprid/B-cyfluthrin, novaluron and triflumuron as harmless (class 1); spinosad and chlorfenapyr as slightly harmful (class 2); and Chlorpyrifos was harmful (class 4) to the parasitoid. Therefore, new studies under greenhouse and field conditions are necessary to confirm these toxicity results.
Key words: Zea mays. Parasitoids. Pesticides. Biological control. Physiological selectivity.
Resumen: El control químico de Spodoptera frugiperda (Lepidoptera: Noctuidae) es difícil debido a que las larvas quedan dentro de los cartuchos de las plantas de maíz (Zea mays), dificultando el contacto con los insecticidas. Las investigaciones sobre parasitoides del género Trichogramma (Hymenoptera: Trichogrammatidae) han contribuido a los avances en la búsqueda de alternativas de control. Sin embargo, existe poca información sobre los efectos de los plaguicidas usados en el maíz sobre estos parasitoides. Así, el objetivo de este estudio fue evaluar la selectividad fisiológica de insecticidas para Trichogramma atopovirilia durante las fases de huevo-larva, pre-pupal y pupal. El bioensayo se llevó a cabo en laboratorio bajo condiciones controladas, donde los huevos de Anagasta (= Ephestia) kuehniella (Lepidoptera: Pyralidae) fueron expuestos al parasitismo por 24 horas empleando el método de la torre de Potter, que contiene los parasitoides en sus diferentes fases. Los insecticidas evaluados, en g.L-1 de i.a., fueron: imidacloprida/B-ciflutrina (Connect 100® SC - 0,033), clorfenapir (Pirate 240® SC - 0,6), clorpirifos (Astro 450® EW - 0,75), novalurom (Rimon 100® CE - 0,05), spinosad (Tracer 480® SC - 0,16) y triflumurom (Certero 480® SC -0,048). Se usó agua como tratamiento control. Los productos se agruparon en clases toxicológicas de acuerdo a las recomendaciones de la OILB. Imidacloprida/B-ciflutrina, triflumurom y novalurom fueron inofensivos (clase 1) para T. atopovirilia; spinosad y clorfenapir fueron levemente perjudiciales (clase 2); y clorpirifos fue perjudicial (clase 4) al parasitoide. Se requieren más estudios en invernadero y de campo para determinar estos resultados de toxicidad.
Palabras clave: Zea mays. Parasitoides. Plaguicidas. Control biológico. Selectividad fisiológica.
Introduction
In 2012, Brazil expects to produce 68 million and five hundred and ten thousand tons of corn, considering the two seasons of the year, with planted area of approximately 14 million and 888 thousand hectares (IBGE 20l2). However, among the factors that may affect the yield and quality of production is the incidence of pests, which can cause damage to crops and production, with a significant economic impact (Fernandes 2003).
Among these pests, the fall armyworm, Spodoptera frugiperda (Smith & Abbot, 1797) (Lepidoptera: Noctuidae) is considered the main one. Its attack occurs from the plant emergence until bolting and silking. The damage caused by this pest can reach 60% depending on the cultivar, degree of infestation, stage of plant development and time of the attack (Cruz et al. 1999, 2002, 2006; Lourencao and Santos 2005). The chemical method is the most widely used against this pest, using several insecticide groups. Such control is difficult due to the behavior of the pest, which remains inside the plant cartridges, thus reducing the contact with insecticides. The failures in the application of the pesticides usually result in recurrence and selection of resistant populations of S. frugiperda, and may often cause biological imbalances (Gassen 1996).
Among the natural enemies of the fall armyworm, egg parasitoids stand out. These insects are known to be highly effective against a large number of agricultural pests. Those belonging to the genus Trichogramma (Hymenoptera: Trichogrammatidae) are widely distributed geographically and play an important role as natural enemies of lepidopteran pests in various agroecosystems (Hassan and Abdelgader 2001).
Beserra and Parra (2004) observed that the average parasitism rate and the number of adults emerged per egg of S. frugiperda were higher for Trichogramma atopovirilia Oatman & Platner, 1983, compared to Trichogramma pretiosum Riley, 1879, and inferred that the first species is more likely to increase their population in a shorter time than T. pretiosum, therefore, being more suitable for the biological control of the pest. However, there is little information on the effects of pesticides used in corn crops on these parasitoids. Thus, this study aims to evaluate the effect of new insecticides used in corn on T. atopovirilia during its immature stages.
Material and methods
The insecticides used with their technical names, trademarks, formulations, dosages and chemical groups were: imidacloprid/p-cyfluthrin (Connect 100TM SC - 0.033 g a.i. L-1, neonicotinoid/pyrethroid), chlorfenapyr (Pirate 240TM SC - 0.6 g a.i. L-1, pyrazole derivative), chlorpyrifos (Astro 450TM EW - 0.75 g a.i. L-1, organophosphate), novaluron (Rimon 100TM CE - 0.05 g a.i. L-1, benzoylurea), spinosad (Tracer 480TM SC - 0.16 g a.i. L-1, spinosyn) and triflumuron (Certero 480TM SC - 0.048 g a.i. L-1, benzoylurea). Water was used as control.
Assessment of the effects of insecticides on T. atopovirilia during its immature stages. Twenty T. atopovirilia females per treatment were individually placed in glass tubes (8 x 2.5 cm) and fed honey droplets deposited on the inner walls of the tubes and closed with PVC film. Approximately 125 eggs of Anagasta kuehniella Zeller, 1879 (Lepidoptera: Pyrali-dae), with up to 24 hours of age, were glued with arabic gum (50% diluted) onto a piece of blue cardboard (5 cm x 0.5 cm). Parasitism was allowed for 24 hours. The cards with parasitized eggs were kept in a climatic chamber at 24 ± 2 °C, RH 70 ± 10% and 14-h photophase until the parasitoids reached each stage of development desired for the bioassay.
Twenty cards with eggs of A. kuehniella per treatment, containing the parasitoid in the egg-larval period or in the pre-pupal or pupal phases, in a total of 140 cards for each developmental stage, were sprayed with the products using a Potter tower adjusted to the pressure of 15 lb./pol2 with a volume application of 1.5 ± 0.5 LiL/cm2 (Vogt 2001). Then, they were distributed into glass tubes, which were kept in a climatic chamber under the same conditions described above. Each treatment consisted of five replications, where each one was constituted of four cards with eggs of A. kuehniella containing the parasitoid in its different immature phases.
The effects of pesticides on parasitoids of the F1 generation were assessed for emergence rate [(number of eggs with an outlet orifice of the parasitoid /total number of parasitized eggs) x 100].
The effects of the compounds on the emerged insects from the F1 generation were evaluated. Twenty females per treatment were individually placed in glass tubes and fed honey, deposited on the inner wall of the tubes, which were closed with PVC film. Around 125 eggs of A. kuehniella, untreated and fixed in a piece of blue paper, were offered to these females for a period of 24 hours.
The effects of the compounds tested on the parasitoids were measured by evaluating the parasitism rate (number of eggs/female of generation F1/24 hours), and according to the emergence rate.
A completely randomized experimental design was used in a 3 x 7 factorial arrangement (3 periods of development x 7 compounds, totaling 21 treatments), the data collected were submitted to analysis of variance, and means were compared by the Scott-Knott test at 5% significance level (Scott and Knott 1974).
According to the reduction in the survival rate and beneficial features of the parasitoids, the insecticides evaluated were grouped into toxicological categories, according to the IOBC recommendations: class 1 = harmless (< 30% reduction), class 2 = slightly harmful (30% to 79% reduction), class 3 = moderately harmful (80% to 99% reduction) and class 4 = harmful (> 99% reduction) (Sterk et al. 1999; van de Veire et al. 2002). The average reduction in the beneficial capacity of parasitoids (parasitism and emergence) was calculated with the following equation: % reduction = 100 - [(% general mean of treatment with pesticide/% general mean of the control treatment) x 100]. The average obtained in the control treatment was used as a reference to calculate the reduction in the beneficial capacity of parasitoids.
Results and discussion
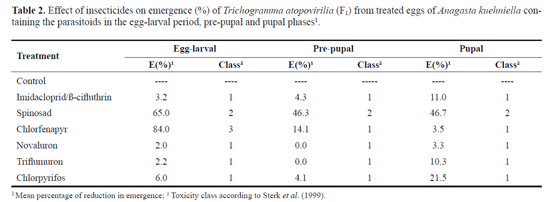
Parasitoid emergence (F1 generation). Imidacloprid/ß-cyfluthrin, novaluron and triflumuron did not affect the emergence rate of the F1- generation parasitoids from the maternal generation treated in the egg-larval period, and can be considered harmless (class 1) (Tables 1 and 2). Our results are similar to those of Matos (2007) who evaluated the selectivity of products used in citrus to T. atopovirilia and found that imidacloprid/ß-cyfluthrin and triflumuron were slightly toxic.
Spinosad reduced the emergence of parasitoids independent of the developmental stage, being framed in class 2 (slightly harmful) (Table 2). Bueno et al. (2008) studied the effect of insecticides on immature stages of T. pretiosum and classified spinosad as toxic (class 4). Comparing the compound spinosad with other products tested within the egg-larval period, it was found that it had the second lowest emergence rate, e.g. 33.0% and, for the pre-pupal and pupal stages, spinosad caused the greatest reduction in adult emergence, of about 51.0% and 51.8%, respectively (Table 1). The major toxicity observed for the product spinosad may be related to the high value of octanol-water (Log Kow), which was 4.91. High values of Log Kow provide greater lipophilicity and a further penetration of the product through the corium (Guedes et al. 1992).
Chlorfenapyr was more toxic than the other compounds tested when sprayed on the host eggs containing parasitoids in the egg-larval stage, causing a lower emergence rate, approximately 15.0%, being classified as moderately harmful (class 3); for the remaining developmental stages of the parasitoid, chlorfenapyr was considered harmless (class 1) (Tables 1 and 2). This effect might be explained in terms of the longer exposure period of the product through the host egg chorion, increasing contamination of the embryo due to their increased activity and contact with the product that penetrated the egg (Carvalho et al. 2001; Nürnberg et al. 2009).
Moura et al. (2005) found that chlorfenapyr caused a reduction in the emergence of the F;-generation T. pretiosum from eggs of A. kuehniella treated in the egg larval period and in the pre-pupal and pupal phases, averaging 19.4%, 2.4% and 37.3%, respectively. The greater toxicity of this product may be related to the high Log Kow value (4.83) and to the amount of active ingredient used by these authors (0.12 g a.i. L-1), since it is twice the amount used in this study.
Insects treated with chlorpyrifos showed an average of 85.61% regardless the developmental stage of the insect, so it was placed in class 1 (harmless) (Tables 1 and 2), however it caused the immediate death of all insects after emergence. It is likely that some residue of the product remained on the surface of the egg chorion of A. kuehniella, leading the death of the parasitoids that touch it. This is a clear clue of the high toxicity of chlorpyrifos for the parasitoid species. These data are similar to those found by Nürnberg et al. (2009), when assessing the effect of Lorsban 480 Br on immature stages of T. pretiosum.
Giolo et al. (2006) studied the effects of organophosphate (phosmet, malathion and fenthion) products on immature stages of T. pretiosum and classified them as harmless (class 1). The selectivity of these compounds probably occurred in the concentrations used by these authors, which were 0.1, 0.2 e 0.05 g a.i. 100 L-1, respectively, less than the dose of chlor-pyrifos used in this study.
Manzoni et al. (2007) evaluated the effects of carbaryl (0.00173 g a.i. L-1) and of the organophosphates fenitrothi-on (0.001 g a.i. L-1) and trichlorfon (0.0015 g a.i. L-1) on T. atopovirilia in its immature stage, and found that all compounds were harmful when sprayed at the egg-larval period and, as well as inthe pre-pupal and pupal phases.
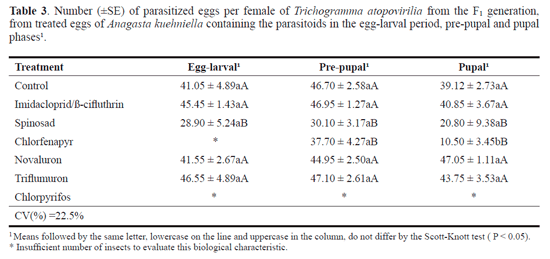
Egg-larval/parasitized eggs. At the egg-larval period, the lowest number of parasitized eggs was recorded for spinosad, with an average of 28.9 parasitized eggs per T. atopovirilia females. In the pre-pupal and pupal phases, the lowest parasitism averages were found for spinosad and chlorfenapyr (Table 3).
Imidacloprid/p-cyfluthrin, novaluron and triflumuron did not reduce the number of parasitized eggs per female of the F1 generation, and these insecticides were considered harmless (class 1) (Tables 3 and 4). Novaluron and triflumuron are selective to T. pretiosum. However, imidacloprid/p-cyfluthrin is slightly harmful (class 2) when the parasitoid was treated at the egg-larval period (Carvalho et al. 2010). The results found in this study are consistent with those of Matos (2007). This author found that triflumuron is harmless or slightly harmful in three immature stages of T. atopovirilia.
The selectivity of novaluron and triflumuron in the parasitism of T. atopovirilia may be related to the mode of action of these insect growth regulator insecticides.
Pre-pupal and pupal stage/parasitized eggs. Chlorfenapyr was harmless (class 1) to the parasitoid in the pre-pupal phase, but slightly harmful (class 2) to the pupa. It was not possible to analyze the ability of parasitism for the egg-larval period, due to the low number of insects that emerged from the F1 generation and the high mortality of this generation after hatching (Tables 3 and 4). Moura et al. (2005) evaluated the effect of chlorfenapyr (0.12 g a.i. L-1) on the parasitism capacity of T. pretiosum of the F1 generation from eggs of A. kuehniella treated in the egg-larval period and in the pre-pupal and pupal phases. Those authors found that this insecticide caused a reduction in parasitism capacity, being classified as slightly harmful (class 2).
Chlorpyrifos allowed the emergence of insects of the F1 generation. However, it caused immediate mortality to parasitoids when they left the host egg, making it impossible to evaluate the parasitism rate; therefore, it was classified as harmful (class 4) (Tables 2 and 4). This result is similar to that found by Cañete (2005) which noted that this insecticide at a dosage of 120 g a.i.100 L-1 affected the parasitism capacity of T. atopovirilia from treated eggs of Anticarsia gemmatalis Hübner, 1818 (Lepidoptera: Noctuidae), since it caused 100% mortality of the parasitoids soon after hatching.
Spinosad was slightly harmful (class 2) to T. atopovirilia in the pre-pupal and pupal phases, causing a reduction of 35.5 and 46.8% in the parasitism rate, respectively, and harmless (class 1) for the egg-larval period of the parasitoid, with an average of 29.6% (Table 4). However, Carmo et al. (2010), when evaluating this same compound in the pupal stage of T. pretiosum, rated it as moderately harmful (class 3).
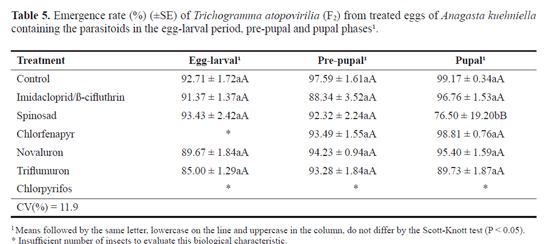
Parasitoid emergence (F2 generation). All the products sprayed on the host eggs containing the parasitoid in the egg-larval period and in the pre-pupal phase did not reduce the emergence rate of T. atopovirilia from the F2 generation and were considered harmless (class 1). However, spinosad decreased this biological characteristic in the pupal stage, with an average of 76.0% emergence (Tables 5 and 6). This result is similar to that found by Cañete (2005) who noted a reduction in the emergence of the F2 generation of T. atopovirilia caused by spinosad.
Chlorfenapyr was classified as harmless (class 1) to the parasitoid in the pre-pupal and pupal phases. However, in the egg-larval period, it was not possible to calculate the emergence rate of the insects from the F2 generation due to the high mortality of the insects of the F1 generation caused by the product (Tables 1 and 6). Except for the egg-larval period, these results are similar to those of Moura et al. (2005) which found that chlorfenapyr did not affect the emergence of T. pretiosum from the F2 generation.
Imidacloprid/ß-cyfluthrin, novaluron and triflumuron did not reduce the emergence of parasitoids of the F2 generation when treated in the egg-larval, pre-pupal and pupal stages, and were considered harmless (class 1) (Tables 5 and 6). These results are similar to those found by Carvalho et al. (2010) and Matos (2007), when they evaluated the effects of imidacloprid/ß-cyfluthrin, novaluron and triflumuron on the emergence rate of T. pretiosum and T. atopovirilia from eggs of A. kuehniella containing the parasitoids in the pupal stage. The effect of chlorpyrifos in the emergence of T. atopovirilia from the F2 generation could not be calculated, due to the fact that the product caused 100.0% mortality of the F1 generation (Table 1).
Sex ratio. The products did not affect the sex ratio of par-asitoids when treated in the egg-larval period and in the pre-pupal and pupal phases. However, when the effects of products on T. atopovirilia were evaluated in each immature stage, there were significant differences between treatments (Table 7). The sex ratio of the parasitoids treated in the egg-larval period (F1 generation) was not affected by any of the evaluated compounds (Table 7). These results were similar to those of Moura et al. (2005) when sprayed imidacloprid and chlorfenapyr/ß-cyfluthrin on T. pretiosum and did not find any effect on this biological characteristic.
Conclusions
In accordance with the toxicity classification of the IOBC, imidacloprid/ß-cyfluthrin, novaluron and triflumuron are harmless to T. atopovirilia, and spinosad and chlorfenapyr are slightly harmfull. In contrast, chlorpyrifos is highly harmful to the parasitoid T. atopovirilia, requiring further studies under greenhouse and field conditions to prove its toxicity.
Acknowledgements
The authors would like to thank the Fundacäo de Amparo à Pesquisa do Estado Minas Gerais - FAPEMIG, for providing a Master's Scholarship, and the Conselho Nacional de Desenvolvimiento Científico e Tecnológico - CNPq, for providing the necessary financial support to accomplish this work.
Literature cited
BESERRA, E. B.; PARRA, J. R. P. 2004. Biologia e parasitismo de Trichogramma atopovirilia Oatman & Platner e Trichogramma pretiosum Riley (Hymenoptera, Trichogrammatidae) em ovos de Spodoptera frugiperda (J.E., Smith) (Lepidoptera, Noctuidae). Revista Brasileira de Entomologia 48 (1): 119-129. [ Links ]
BUENO, A. F.; BUENO, R. C. O. F.; PARRA, J. R. P.; VIEIRA, S. S. 2008. Effects of pesticides used in soybean crops to the egg parasitoid Trichogramma pretiosum. Ciencia Rural 38 (6): 1495-1503. [ Links ]
CARVALHO, G. A.; PARRA, J. R .P.; BAPTISTA, G. C. 2001. Seletividade de alguns produtos fitossanitários a duas linhagens de Trichogramma pretiosum Riley, 1879 (Hymenoptera: Trichogrammatidae). Ciencia e Agrotecnologia, Lavras (25): 583-591. [ Links ]
CARVALHO, G. A.; GODOY, M. S.; PARREIRA, D. S.; REZENDE, D. T. 2010. Effect of chemical insecticides used in tomato crops on immature Trichogramma pretiosum (Hymenoptera: Trichogrammatidae). Revista Colombiana de Entomologia 36 (1): 10-15. [ Links ]
CAÑETE, C. L. 2005. Seletividade de inseticidas a espécies de Trichogramma (Hymenoptera: Trichogrammatidae). Tese (Ciencias - Zoologia), Universidade Federal do Paraná, PR. 106 p. [ Links ]
CRUZ, I.; FIGUEIREDO, M. L. C.; OLIVEIRA, A. C.; VASCONCELOS, C. A. 1999. Damage of Spodoptera frugiperda (Smith) in different maize genotype cultivated in soil under three levels of aluminium saturation. International Journal Pest Management 45 (4): 293-296. [ Links ]
CRUZ, I.; VIANA, P. A.; WAQUIL, J. M. 2002. Cultivo do milho pragas da fase vegetativa e reprodutiva. Comunicado Técnico 49, 1° edijao. Dezembro, Sete Lagoas, MG. [ Links ]
CRUZ, I.; FIGUEIREDO, M. L. C.; VIANA, P. A.; PAULA, L. A. 2006. Suscetibilidade de diferentes populaföes de Spodoptera frugiperda a inseticidas químicos. p. 234. In: Anais do XXVI Congresso Nacional de Milho e Sorgo, Belo Horizonte. [ Links ]
FERNANDES, O. D. 2003. Efeito do milho geneticamente modificado (MON810) em Spodoptera frugiperda (J.E. Smith, 1797) e no parasitoide de ovos Trichogramma spp. Thesis (Doctor in Science/Entomology) - Escola Superior de Agricultura "Luiz de Queiroz", Universidade de Säo Paulo, Piracicaba, Estado de Säo Paulo - Brazil. 182 p. [ Links ]
GASSEN, D. 1996. Manejo de pragas associadas à cultura do milho. Passo Fundo: Aldeia Norte. 127 p. [ Links ]
GUEDES, R. N. C.; LIMA, J. O. G.; ZANUNCIO, J. C. 1992. Sele-tividade dos inseticidas deltametrina, fenvalerato e fenitrotiom para Podisus connexivus (Heteroptera: Pentatomidae). Anais da Sociedade Entomológica do Brasil 21 (3): 339-346. [ Links ]
CARMO, E. L.; BUENO, A. F.; BUENO, R. C. O. F.; VIEIRA, S. S.; GOULART, M. M. P.; CARNEIRO, T. R. 2010. Seletividade de produtos fitossanitários utilizados na cultura da soja para pupas de Trichogramma pretiosum Riley, 1879 (Hymenoptera: Trichogrammatidae). Arquivo do Instituto Biológico 77 (2): 283-290. [ Links ]
GIOLO, F. P.; ANDERSON, D. G.; CRISTIANE, G. M.; WAGNER, R. H.; CRISTIANE, M; RODOLFO, V. C. 2006. Toxi-cidade de pesticidas utilizados na cultura do pessegueiro para estágios imaturos de Trichogramma pretiosum Riley (Hyme-noptera: Trichogrammatidae). BioAssay 1 (4): 1-7. [ Links ]
HASSAN, S. A.; ABDELGADER, H. 2001. A sequential testing program to assess the side effects of pesticides on Trichogram-ma cacoeciae Marchal (Hymenoptera: Trichogrammatidae). IOBC/WPRS Bulletin 24 (4): 71-81. [ Links ]
IBGE, 2012. Levantamiento sistemático da produjao agrícola: Pesquisa mensal de previsào e acompanhamento das safras agrícolas no ano civil. Fundajao Instituto Brasileiro de Geografia e Estatística, Rio de Janeiro. 25 (4): 1-88. [ Links ]
LOURENQÄO, A. L. F.; SANTOS, H. R. 2005. Danos de Spodoptera frugiperda (J. E. Smith, 1797) (Lepidoptera: Noctuidae) na cultura do milho (Zea mays L.). Revista de Agricultura 80 (3): 340-355. [ Links ]
MANZONI, C. G.; GRÜTZMACHER, A. D.; GIOLO, F. P.; HÄRTER, W. R.; CASTILHOS R. V.; PASCHOAL, M. D. F.2007. Seletividade de Agroquímicos utilizados na produjao integrada de maja aos parasitóides Trichogramma pretiosum Riley e Trichogramma atopovirilia Oatman & Platner (Hymenoptera: Trichogrammatidae). BioAssay 2 (1): 11. Available at: http://www.bioassay.org.br/articles/2,1/BA2.1pdf. Review date: 22 July 2012. [ Links ]
MATOS, M. M. 2007. Seletividade a Trichogramma atopovirilia Oataman & Platner, 1983 de agroquímicos utilizados na citricultura paulista para o controle do bicho-furao-dos-citros, Gymnandrosoma aurantianum Lima, 1927. 54p. Thesis (Master of Science/Entomology). Universidade de Sao Paulo, Escola Superior de Agricultura "Luiz de Queiroz", Piracicaba, Estado de Sao Paulo, Brazil. [ Links ]
MOURA, A. P.; CARVALHO, G. A.; RIGITANO, R. L. O. 2005. Toxicidade de inseticidas utilizados na cultura do tomateiro a Trichogramma pretiosum. Pesquisa Agropecuária Brasileira 40 (3): 203-210. [ Links ]
NÜRNBERG, S. D.; GRÜTZMACHER, A. D.; KOVALESK, A.; CAMARGO, E. S.; PASINI, R. A. 2009. Toxicidade de agrotóx-icos utilizados na produjao integrada de maja a Trichogramma pretiosum Riley, 1879 (Hymenoptera: Trichogrammatidae) em condiföes de laboratorio. Revista Brasileira de Agrociecia 15 (1-4): 49-56. [ Links ]
SCOTT, A. J.; KNOTT, M. A. 1974. A cluster analysis method for grouping means in the analysis of variance. Biometrics 30 (3): 507-512. [ Links ]
STERK, G.; HASSAN, S. A.; BAILLOD, M.; BAKKER, F.; BIGLER, F.; BLÜMEL, S.; BOGENSCHÜTZ, H.; BOLLER, E.; BROMAND, B.; BRUN, J.; CALIS, J. N. M.; COREMANS-PELSENEER, J.; DUSO, C.; GARRIDO, A.; GROVE, A.; HEIMBACH, U.; HOKKANEN, H.; JACAS, J.; LEWIS, G.; MORETH, L.; POLGAR, L.; ROVERSTI, L.; SAMS0E-PE-TERSEN, L.; SAUPHANOR, B.; SCHAUB, L.; STÄUBLI, A.; TUSET, J. J.; VAINIO, A.; VAN DE VEIRE, M.; VIGGIANI, G.; VIÑUELA, E.; VOGT, H. 1999. Results of the seventh joint pesticide testing programme carried out by the IOBC/WPRS working group 'Pesticides and Beneficial Organisms'. BioControl 44 (1): 99-117. [ Links ]
VAN DE VEIRE, M.; STERK, G.; VAN DER STAAIJ, M.; RAMAKERS, P. M. J.; TIRRY, L. 2002. Sequential testing scheme for the assessment of the side-effects of plant protection products on the predatory bug Orius laevigatus. BioControl 47 (1): 101-113. [ Links ]
VOGT, H. 2001. Effects of Quassia products on Chrysoperla carnea (Stephens) (Neuroptera, Chrysopidae). IOBC/WPRS Bulletin, Dossenheim 24 (4): 47-52. [ Links ]
Suggested citation:
MAIA, JADER BRAGA; GERALDO ANDRADE CARVALHO; RODRIGO LOPES DE OLIVEIRA; OLINTO LASMAR and MARIA ISABELLA SANTOS LEITE. 2013. Effects of insecticides used in corn on immature stages of Trichogramma atopovirilia (Hymenoptera: Trichogrammatidae). Revista Colombiana de Entomología 39 (2): 205-210.