Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista Colombiana de Ciencias Pecuarias
Print version ISSN 0120-0690On-line version ISSN 2256-2958
Rev Colom Cienc Pecua vol.23 no.4 Medellín Oct./Dec. 2010
Antibodies to West Nile virus in equines of Antioquia and Meta, Colombia, 2005-2008¤
Anticuerpos contra el virus del Oeste del Nilo en equinos de Antioquia y Meta, Colombia, 2005-2008
Anticorpos contra o Vírus do Nilo Ocidental em eqüinos da Antioquia e do Meta, Colômbia 2005-2008
Yenny Góez-Rivillas1, DVM; Natalia Taborda1, Microbiologist; Francisco J Díaz1*, MD, PhD; Agustín Góngora2, DVM, MSc, Dr. Sci; Juan D Rodas3, DVM, MSc, PhD; Julián Ruiz-Sáenz1 DVM, MSc; Jorge E Osorio4, DVM, MSc, PhD.
1Grupo Inmunovirología, Universidad de Antioquia, Medellín, Colombia. 2Grupo GIRGA, Universidad de Los Llanos, Villavicencio, Colombia. 3Grupo Centauro, Universidad de Antioquia, Medellín, Colombia 4University of Wisconsin, Madison, WI, USA.
(Received: 3 june, 2010; accepted: 7 september, 2010)
Summary
West Nile Virus (WNV) is an arthropod-borne agent classified in the Flavivirus genus. Infection has been demonstrated in many vertebrate species including birds, mammals and reptiles. WNV can affect the nervous system of humans, horses and birds causing mild to severe illness and sometimes death. In 1999 WNV was introduced into the Americas causing a small outbreak in New York City. In the following years, the virus spread across North America and later into Central America, the Caribbean and parts of South America. Serological evidence of WNV in Colombia was first documented in 2005 in equines from the Atlantic coast; however clinical cases in humans or animals have not been reported. We extended these studies searching for WNV antibodies in sera of equines of two other provinces in Colombia: Antioquia and El Meta. IgG and IgM antibodies were first determined and reactive sera were processed by plaque reduction neutralization test (PRNT) to confirm the specificity of results. Four horses from Antioquia but none from El Meta tested positive for WNV antibodies. These results suggest that WNV has spread across the Atlantic coast and is now invading the Andean region in Colombia.
Key Words: Colombia, Antioquia, El Meta, equines, PRNT, West Nile Virus.
Resumen
El virus del Oeste del Nilo (WNV) es un agente del género Flavivirus transmitido por artrópodos. La infección con WNV ha sido demostrada en muchas especies de aves, mamíferos y reptiles. El WNV puede afectar el sistema nervioso de humanos, caballos y aves causando enfermedad de leve a severa, ocasionando la muerte en algunos casos. En 1999, el virus fue introducido en Norteamérica causando un brote en la ciudad de New York. En los siguientes años, el virus se extendió por Norteamérica, y posteriormente fue encontrado en el Caribe, Centro y Suramérica. El primer reporte de anticuerpos para WNV en Colombia se hizo en 2005, en equinos de la costa Atlántica. En el presente estudio se extendió la búsqueda de anticuerpos a otros dos Departamentos de Colombia: Antioquia y El Meta. Primero se determinó la presencia de anticuerpos IgM e IgG, y los sueros reactivos fueron procesados para anticuerpos neutralizantes por la técnica de reduccion de placas para confirmar los resultados. Cuatro equinos de Antioquia y ninguno de El Meta fueron positivos para anticuerpos anti-WNV. Los resultados sugieren que el WNV está ampliamente distribuido en la costa Atlántica de Colombia y ha iniciado su dispersión por la zona andina.
Palabras clave: Antioquia, Colombia, El Meta, equinos, PRNT, virus del oeste del Nilo.
Resumo
O vírus do Nilo Ocidental é um agente transmitido por artrópodes e pertence ao género Flavivirus. A infecção tem sido demonstrada em várias espécies de vertebrados incluindo pássaros, mamíferos e répteis. O vírus do Nilo Ocidental pode afectar o sistema nervoso de humanos, eqüinos e pássaros, causando doença de severidade média à grave a qual pode causar a morte em alguns casos. Em 1999, o vírus do Nilo Ocidental foi introduzido no continente americano, causando um surto na cidade de Nova York. Posteriormente, o vírus se disseminou pela América do Norte e mais tarde pela América Central, Caribe e parte da América do Sul. Os primeiros relatos do vírus do Nilo Ocidental na Colômbia surgiram em 2005 afectando eqüinos na costa atlântica. O objectivo desse trabalho foi buscar anticorpos contra o vírus do Nilo Ocidental no soro de equinos de dois estados da Colômbia: Antioquia e Meta. Anticorpos da classe IgG e IgM foram primeiramente determinados e soros reactivos foram analisados pela técnica de neutralização por redução em placa (PRNT) para confirmar a especificidade dos resultados. Quatro equinos provindos da Antioquia apresentaram resultados positivos para anticorpos contra o vírus do Nilo Ocidental; entretanto não foram detectados anticorpos nos animas provindos do Meta. Estes resultados sugerem que o vírus do Nilo Ocidental tem se disseminado através da costa atlântica e está agora invadindo a região andina na Colômbia.
Palavras chave: eqüinos, PRNT, vírus do Nilo Ocidental.
¤ To cite this paper: Góez-Rivillas Y, Taborda N, Díaz FJ, Góngora A, Rodas JD, Ruiz-Sáenz J, Osorio JE. Antibodies to West Nile virus in equines of Antioquia and El Meta, Colombia. 2005-2008. Rev Colom Cienc Pecu 2010; 23:462-470.
* Corresponding author. Francisco J Díaz. Universidad de Antioquia, Sede de Investigación Universitaria (SIU), Calle 62 # 52-59, Laboratorio 532, Medellín, Colombia. E-mail: franciscodiaz314@gmail.com
Introduction
West Nile Virus (WNV) (genus Flavivirirus, family Flaviviridae) is an arthropod-borne agent antigenically classified in the Japanese encephalitis virus (JEV) antigenic complex along with St. Louis encephalitis virus (SLEV), and Murray Valley encephalitis virus (MVEV) (Beaty et al., 1995). In its natural cycle, WNV is maintained in birds and Culex spp mosquitoes. Many wild vertebrates including wolves, bears, crocodiles, alligators and bats (Tasha et al., 2007; Epp et al., 2008; Gould and Fikrig, 2004), as well as domestic animals including horses, cats and dogs can be infected (Komar et al., 2001; Read et al., 2005). The infection in humans, as well as in most non-avian vertebrates, occurs incidentally and they do not contribute to the spreading of the infection (Gould and Fikrig, 2004).
WNV can affect the nervous system of humans, horses and birds causing mild to severe illness and sometimes death. The incubation period ranges from 3 to 15 days (Nicolle et al., 2004). Horses and humans are usually asymptomatic with no detectable viremia but sometimes they develop clinical signs such as fever, depression, tremors, weakness, lack of coordination and, in more serious cases, paralysis and death (Bunning et al., 2020; Ostlund et al., 2000; Weir and Shapiro, 2004). Humans and horses usually develop low-grade viremia but virus titers may be high in nervous tissue (Venter et al., 2010).
WNV was first isolated in 1937 and has been detected throughout Africa, the Middle East, southern Europe, Russia, and India (Buckley et al., 2003). In 1999, it was introduced into the Americas (Nash et al., 2001) causing a small outbreak in New York. In the following years, the virus spread across North America and later into Central America, the Caribbean and parts of South America (Hayes and Gubler, 2006). In spite of causing disease in more than 27,000 humans, in more than 25,000 equines and hundreds of thousands of avian deaths in USA, there have been only a few reports of WNV disease in the Caribbean and Latin America (Blitvich, 2008).
Antibody response to WNV in horses has been studied in a few works (Bunning et al., 2002, Shirafuji et al., 2009). Both, neutralizing and IgM antibodies are detected by day 7 after experimental inoculation and peak by day 10 to 14. While IgM rapidly disappear thereafter, neutralizing antibodies, supposedly of the IgG class, persist for more than a year. There is a strong serologic cross-reactivity among members of the JEV antigenic complex, which is detectable in most antibody tests. Plaque reduction neutralization test (PRNT) is the most specific assay available, and it is the only one that discriminates among antibodies against these agents, at least in primary infections (Shirafuji et al., 2009). PRNT is often used as a confirmatory assay in samples that previously tested positive in widely reactive tests such as ELISA or hemaglutination inhibition (Dupuis et al., 2003; Niedrig et al, 2007; Morales et al., 2006). Cellular immune response to WNV in horses has not been extensively studied (Castillo-Olivares & Wood, 2004).
WNV-neutralizing antibodies have been detected in birds captured in Jamaica, Dominican Republic, Puerto Rico, Cuba and Guadeloupe (Dupuis et al., 2003; Komar et al., 2003; Dupuis et al., 2005), and in horses from México (Lorono-Pino et al., 2003), Argentina (Morales et al., 2006), Venezuela (Bosch et al., 2007) and Guatemala (Morales-Betoulle et al., 2006). First reports in Colombia appeared in 2005 and included equines from the northern coastal areas (Mattar et al., 2005; Berrocal et al., 2006). Here are we extended these studies in equines from two Colombian provinces, Antioquia and El Meta, located in the northwestern and central-eastern regions of the country, respectively (Figure 1).
Materials and methods
This is a descriptive sero-epidemiological survey. Sera were collected from healthy equines in selected towns of Antioquia and El Meta. Sampling took place in August 2005 in El Meta and between July 2006 and July 2008 in Antioquia. All sampling sites were located on privately owned ranches, where the horses were primarily used as herd cattle or for other labor. Sampled animals were selected by convenience. According to the owners, none of the horses had ever been outside of the province and none had been vaccinated against WNV. This vaccine has not been approved for use in Colombia.
Sampling sites represent different ecosystems: in Antioquia they were located in the municipalities of Turbo, Chigorodó, La Ceja and the village of Bolombolo. The first two are located in the coastal borders of the Gulf of Urabá, the southernmost corner of the Caribbean Sea, at less than 50 m over the sea level (OSL) and they are dedicated to banana plantations and livestock breeding. La Ceja is located in the highlands of the Colombian central mountain range, at 2,200 m OSL, and the land is mainly agricultural. Bolombolo is in the Cauca river valley in the Andean region, mainly a coffee-growing region about 400 m OSL. Sampling sites in El Meta are in the northwestern part of the province, in the municipalities of Acacias, Castilla, Cumaral, Guamal, Restrepo, San Martín, Villavicencio and Paratebueno. All of them are located in the Colombian eastern plains, mostly in or near the foothills of the eastern mountain range. The ecosystem is a savannah with an altitude of about 500 m OSL, and the land is mostly dedicated to agriculture, livestock breeding and oil industry. The town of Paratebueno is in the territory of the Cundinamarca province but it is close to, and not much different from, the described towns of El Meta (Figure 1). It was no possible to determine the age of 96 equines from Antioquia and 36 from El Meta; the exact origin of 67 equines from El Meta was also unknown.
Approximately 10 ml of whole blood were collected from each animal by jugular venipuncture. Blood was held at ambient temperature for at least 15 minutes to permit clotting, and then placed into coolers. At the end of the day, sera were separated by centrifugation, transferred to 2-mL cryovials and stored at -80 ºC until used in serological tests.
All the serological procedures were performed following the recommendations in the guide "Epidemic/Epizootic West Nile Virus in the United States: Guidelines for Surveillance, Prevention, and Control" by the Centers for Disease Control and Prevention, (CDC, 2003). All samples were tested for IgM and IgG antibodies against WNV using an IgM capture enzyme-linked immunosorbent assay (MAC-ELISA) and an indirect IgG ELISA, respectively. Briefly, the MAC-ELISA is based on the capture of serum IgM by an anti-horse IgM-sensitized microtitre plate, followed by the sequential addition of WNV recombinant antigen, peroxidase-labeled 6B6C-1 anti-flavivirus conjugate and ABTS substrate-chromogen mixture (Bunning et al., 2002). For detection of serum IgG the ELISA plates were coated with the WNV antigen and the presence of antibodies was revealed by the sequential addition of the animal sera, a peroxidaselabeled goat anti-horse IgG and the substratechromogen mixture (Castillo-Olivares and Wood, 2004). For both procedures a WNV recombinant antigen produced in COS-1 cells (Hennessy Research, Shawnee, KS) was used and anti-horse conjugates and substrate-chromogen mixtures were obtained from Kierkegaard & Perry Laboratories (Gaithersburg, MD). Because of the close antigenic relationship among flavivirus and the wide reactivity of the ELISAs, sera reactive in these tests were considered as seropositive for flavivirus IgM or IgG.
All serum samples that tested as reactive for IgM or IgG were titrated for WNV-specific neutralizing antibodies by PRNT at the National Wildlife Health Center (NWLHC), Madison, WI, USA, using the New York 1999 strain, as previously described (Komar et al., 2001). End point 90% neutralization titer (PRNT90) was determined using two-fold serial dilutions beginning at 1:20. A PRNT90 titer of 1:40 or greater was considered as specific for WNV. Sera reactive for IgM or IgG but with PRNT titers to WNV lower than 1:40 were presumptively considered seropositive to a different flavivirus.
Results were analyzed using descriptive statistics. Fisher's exact test and chi-square test for linear trend were used to determine statistically significant differences and trends, respectively. Level of significance accepted was p<0.05. This work was approved by the ethics committee for animal experiments of the Universidad de Antioquia, as stated in the minutes No. 33 of August 10, 2006.
Results
A total of 419 samples were obtained from healthy equines from the two provinces (59.2% from Antioquia and 40.8% from El Meta). The age of the animals ranged from 3 months to 25 years, with a mean of 14 years. Most (94.7%) sera were non-reactive in both, IgM and IgG ELISA tests, and were not tested by PRNT. Five samples were reactive for IgM and 17 for IgG. No sample was positive by both IgM and IgG tests. None of the five IgM reactive sera were positive by PRNT but four (23.5%) of those IgG reactive were PRNT positive (Table 1).
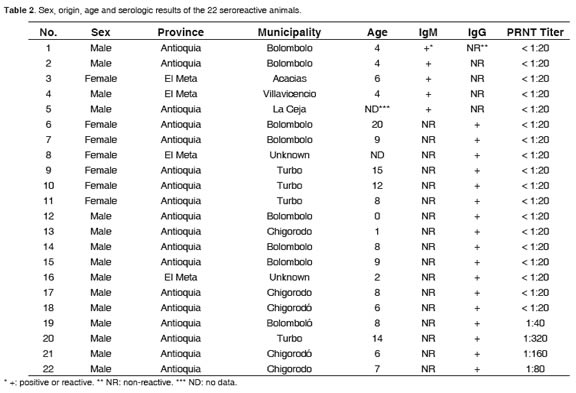
More detailed information about the animals that were reactive by any test is presented in table 2. PRNT90 titers of seropositive horses ranged from 80 to 320. All four animals that were confirmed as WNV seropositive by PRNT were from the province of Antioquia, three of the coastal municipalities of Turbo and Chigorodó, and one from the Andean town of Bolombolo. All of them were males but no statistically significant difference by sex was observed in any of the serological tests performed. The four PRNT positive animals were between 6 and 14 years old. There was a significant trend in the reactivity for IgG by age (p=0.04 chisquare test for linear trend, Figure 2). No such a trend was observed for the other two tests but the number of reactive/ positive animals was too low to be analyzed.
Discussion
In 1999 WNV was introduced as a threat to both, public and animal health, in The Americas. Its spread along the Western Hemisphere was an anticipated outcome because many species of birds migrate every year from north to south. However, in South America WNV has only been isolated in Central Argentina in 2006; it was obtained from the brain of 3 horses that died with encephalitis in February 2006 (Morales et al., 2006). Additional evidence of introduction of WNV into South America came from serological studies. In Colombia, WNV neutralizing antibodies have been reported only in horses of the northern Atlantic region (Mattar et al., 2005; Berrocal et al., 2006).
This work examined the presence of antibodies to WNV in the Urabá zone, also in the Atlantic region, and for the first time, in two other Colombian regions: the Andean zone and the eastern plains. The finding of neutralizing antibodies in sera of three horses from the municipalities of Turbo and Chigorodó, located near the Caribbean Sea, was not unexpected since Mattar et al. (2005) and Berrocal et al. (2006), using a similar methodology, found WNV seropositive horses in the neighboring province of Córdoba, and other provinces of the Atlantic region. Bosch et al. (2007) also demonstrated WNV circulation in the Atlantic coast of Venezuela. These and other studies show that WNV has already reached a significant spread in the northern coastal regions of South America and likely in the entire Caribbean basin (Dupuis et al., 2003; Blitvich et al., 2003; Quirin et al., 2004).
The finding of WNV-neutralizing antibodies in a horse of Bolombolo, Antioquia suggests that the virus has begun to disseminate across the Andean region of Colombia. Bolombolo is located in the mid Cauca river valley, a likely route of bird migration. However, the PRNT90 titer of this horse was 1:40, the lowest titer accepted as specific for WNV. Five other horses from Bolombolo that tested reactive in the IgG test were negative by PRNT90. In view of the these results, and considering that neutralizing antibodies can also be produced in response to repeated exposure to antigenically related agents, we cannot rule out that this titer could have been elicited by repeated exposure to different flaviviruses circulating in that zone.
On the other hand, only two (1.2%) equines from the province of El Meta were reactive for IgG and two more for IgM anti-WNV, none of which was positive by PRNT. This could indicate that WNV had not arrived to that region at the moment of sampling (year 2005) but a more recent invasion cannot be ruled out. Continuous surveillance of WNV is warranted in El Meta, where the horse is a very important element in the economy and culture of the region.
A point that deserves consideration is the significant number of samples that tested reactive in the IgM or IgG tests, 5 and 13, respectively, but that were not confirmed as WNV-positive by PRNT.
They represent cross-reacting antibodies elicited by other circulating flavivirus or, alternatively, false positive results produced by the lack of specificity of the tests. In the case of IgG antibodies, we favor the first explanation since the frequency of this antibodies increased significantly with age, as expected in a population that has been repeatedly exposed to flavivirus infections (Figure 2). Previous infections with viruses like SLEV, Ilheus, Bussuquara, Rocio, Cacipacore, or Iguape, which has been previously detected circulating in Colombia or neighboring countries (Groot, 1964, Gubler, 2007) could have elicited these antibodies.
The occurrence of positive IgM results not confirmed by PRNT is more difficult to explain. It might represent WNV-specific antibodies developed in the course of a very recent infection. However, since neutralizing antibodies seems to appear by the same time that IgM (Shirafuji, 2009), and since early IgG overlaps with late IgM, it is strange that none of the IgM reactive sera were positive by any of the other two tests. None of this IgM-reactive equines exhibited illness at the moment of sampling or in the previous days but asymptomatic infection in horses seems to be the rule rather than the exception (Venter, 2010).
In the light of the results of this, and other serological studies that reported WNV antibodies, it is natural to ask why WNV has apparently not caused severe neurological and fatal disease in horses or in humans in the Caribbean, Central and South America, with the exception of the three horses that died in Argentina in 2006 (Morales et al., 2006). There are several arguable hypotheses: one is that WNV invasion of these territories is still in an early phase and the number of infected individuals is too low for an apparent impact. Alternatively, severe human and animal cases may be occurring, but going unnoticed by the limited awareness, poor surveillance or lack of laboratory resources. Some authors have argued that immunity to other flaviviruses prevalent in tropical regions might be protecting native populations from severe forms of WNV infections (Deardorff et al., 2006).
Another hypothesis is that the WNV strain that is invading Central and South America is not as virulent as the strain introduced in 1999 in New York City. A 2003 WNV isolate from the Mexican state of Tabasco exhibited a mutation that suppresses a glycosylation site in the envelope protein, and this mutation confers an attenuated phenotype in a mouse model (Beasley et al., 2004). Finally, WNV could have been present in tropical Americas for a long time. Over the years, it could have selected for vertebrate populations resistant to pathogenic WNV infection. All of these hypotheses lack experimental or observational support at this time and should be tested in studies that include virus isolation or direct detection assays. We are now working in that direction.
In summary, we have provided evidence of the circulation of WNV, or other closely related viruses, in equines of two regions of the province of Antioquia, Colombia: the Urabá zone and the mid Cauca river valley. The clinical and epidemiological implications of this finding are still obscure.
Acnowledgements
We would like to thank Diana Echeverry for her work in the ELISA tests, and Sair Arboleda for her help with Figure 1. Also, thanks to National Wild Life Health Center, Madison, Wisconsin, USA, for performing the PRNT. This work was supported by the Comité para el Desarrollo de la Investigación, CODI, Universidad de Antioquia, project # SIU8700-1420.
References
1. Barrett AD, Beaty BJ, Tesh RB. West Nile Virus isolation in human and mosquitoes, Mexico. Emerg Infect Dis 2005; 11:1449-1552. [ Links ]
2. Beasley DW, Davis CT, Estrada-Franco J, Navarro-López R, Campomanes-Cortes A, Tesh RB, Weaver SC, Barrett AD. Genome sequence and attenuating mutations in West Nile virus isolate from Mexico. Emerg Infect Dis 2004; 10:2221-2224. [ Links ]
3. Beaty BJ, Calisher CH, Shope RE. Arboviruses. In: Lennette EH, Lennette DA, Lennette ET (ed.). Diagnostic procedures for viral, rickettsial and chlamydial infections, 7th ed., Washington, Public Health Association, 1995. p. 189-212 [ Links ]
4. Bernabeu-Wittel M, Ruiz-Pérez M, del Toro MD, Aznar J, Muniain A, de Ory F, Domingo C, Pachón. West Nile virus past infections in the general population of Southern Spain. J Enferm Infecc Microbiol Clin 2007; 25:561-565 [ Links ]
5. Berrocal L, Peña J, González M, Mattar S. West Nile virus; ecology and epidemiology of an emerging pathogen in Colombia. Rev Salud Publica (Bogota) 2006; 8:218-228. [ Links ]
6. Blitvich BJ, Fernández-Salas I, Contreras-Cordero JF, Marlenee NL, González-Rojas JI, Komar N, Gubler DJ, Calisher CH, Beaty BJ. Serologic evidence of West Nile virus infection in horses, Coahuila State, Mexico. Emerg Infect Dis 2003; 9:853-6. [ Links ]
7. Blitvich BJ. Transmission dynamics and changing epidemiology of West Nile virus. Anim Health Res Rev 2008; 9:71-86. [ Links ]
8. Bosch I, Herrera F, Navarro JC, Lentino M, Dupuis A, Maffei J, Jones M, Fernández E, Pérez N, Pérez-Emán J, Guimarães AE, Barrera R, Valero N, Ruiz J, Velásquez G, Martinez J, Comach G, Komar N, Spielman A, Kramer L. West Nile virus, Venezuela. Emerg Infect Dis 2007; 13:651-653. [ Links ]
9. Botha EM, Markotter W, Wolfaardt M, Paweska JT, Swanepoel R, Palacios G, Nel LH, Venter M. Genetic determinants of virulence in pathogenic lineage 2 West Nile virus strains. Emerg Infect Dis 2008; 14:222-230. [ Links ]
10. Buckley A, Dawson A, Moss SR, Hinsley SA, Bellamy PE, Gould EA. Serological evidence of West Nile virus, Usutu virus and Sindbis virus infection of birds in the UK. J Gen Virol 2003; 84:2807-2817. [ Links ]
11. Bunning ML, Bowen RA, Cropp CB, Sullivan KG, Davis BS, Komar N, Godsey MS, Baker D, Hettler DL, Holmes DA, Biggerstaff BJ, Mitchell CJ. Experimental infection of horses with West Nile virus. Emerg Infect Dis 2002; 8:380-386. [ Links ]
12. Castillo-Olivares J, Wood J. West Nile virus infection of horses. Vet Res 2004; 35:467-483. [ Links ]
13. CDC. Epidemic/Epizootic West Nile Virus in the United States: Guidelines for Surveillance, Prevention, and Control. U.S. 2003. [Accessed on April 20, 2009]. URL: http://www.cdc.gov/ ncidod/dvbid/westnile/resources/wnv-guidelines-aug-2003.pdf [ Links ]
14. Deardorff E, Estrada-Franco J, Brault AC, Navarro-López R, Campomanes-Cortes A, Paz-Ramírez P, Solis-Hernandez M, Ramey WN, Davis CT, Beasley DW, Tesh RB, Barrett AD, Weaver SC. Introductions of West Nile virus strains to Mexico. Emerg Infect Dis 2006; 12:314-318. [ Links ]
15. Dupuis AP, Marra PP, Kramer LD. Serologic evidence of West Nile virus transmission, Jamaica, West Indies. Emerg Infect Dis 2003; 9:860-863. [ Links ]
16. Dupuis AP, Marra PP, Reitsma R, Jones MJ, Louie KL, Kramer LD. Serologic evidence for West Nile virus transmission in Puerto Rico and Cuba. Am J Trop Med Hyg 2005; 73:474-476. [ Links ]
17. Elizondo-Quiroga D, Davis CT, Fernández-Salas I, Escobar-Lopez R, Velasco Olmos D, Soto Gastalum LC, Aviles Acosta M, Elizondo-Quiroga A, Gonzalez-Rojas JI, Contreras Cordero JF, Guzman H, Travassos da Rosa A, Blitvich BJ, Epp T, Waldner C, Leighton FA, Berke O, Townsend HG. Seroprevalence and risk factors for infection with West Nile virus in Saskatchewan horses, 2003. Can J Vet Res 2007; 71:256-263. [ Links ]
18. Epp T, Waldner C, Corrigan R, Curry P. Public health use of surveillance for West Nile virus in horses: Saskatchewan, 20032005. Transbound Emerg Dis 2008; 55:411-6. [ Links ]
19. Farajollahi A, Panella NA, Carr P, Crans W, Burguess K, Komar N. Serologic evidence of West Nile virus infection in black bears (Ursus americanus) from New Jersey. J Wildl Dis 2003; 39:894-6. [ Links ]
20. Gómez A, Kilpatrick AM, Kramer LD, Dupuis AP 2nd, Maffei JG, Goetz SJ, Marra PP, Daszak P, Aguirre AA. Land use and West Nile virus seroprevalence in wild mammals. Emerg Infect Dis 2008; 14:962-5. [ Links ]
21. Gould LH, Fikrig E. West Nile virus: a growing concern? J Clin Invest 2004 Apr; 113:1102-1107. [ Links ]
22. Groot H. Estudios sobre virus transmitidos por artópodos en Colombia. Revista de la academia colombiana de Ciencia Exactas, Físicas y Naturales 1964; 12:2-23. [ Links ]
23. Gubler DJ. The continuing spread of West Nile virus in the western hemisphere. J Clin Infect Dis 2007; 45:1039-1046. [ Links ]
24. Gyure KA. West Nile virus infections. J Neuropathol Exp Neurol 2009; 68:1053-1060. [ Links ]
25. Hayes EB, Gubler DJ. West Nile Virus: Epidemiology and Clinical Features of an Emerging Epidemic in the United States. Annual Review of Medicine 2006; 57:181-194. [ Links ]
26. Kleiboeker SB, Loiacono CM, Rottinghaus A, Pue HL, Johnson GC. Diagnosis of West Nile virus infection in horses. J Vet Diagn Invest 2004; 16:2-10. [ Links ]
27. Komar N. West Nile viral encephalitis. Rev Sci Tech 2000; 19:166-176. [ Links ]
28. Komar N, Panella NA, Boyce E. Exposure of domestic mammals to West Nile virus during an outbreak of human encephalitis, New York City, 1999. Emerg Infect Dis 2001; 7:736-738. [ Links ]
29. Komar N, Panella NA, Burns JE, Dusza SW, Mascarenhas TM, Talbot TO. Serologic evidence for West Nile virus infection in birds in the New York City vicinity during an outbreak in 1999. Emerg Infect Dis 2001; 7:621-625. [ Links ]
30. Komar O, Robbins MB, Klenk K, Blitvich BJ, Marlenee NL, Burkhalter KL, Gubler DJ, Gonzálvez G, Peña CJ, Peterson AT, Komar N. West Nile virus transmission in resident birds, Dominican Republic. Emerg Infect Dis 2003; 9:1299-1302. [ Links ]
31. Lanciotti RS, Roehrig JT, Deubel V, Smith J, Parker M, Steele K, Crise B, Volpe KE, Crabtree MB, Scherret JH, Hall RA, MacKenzie JS, Cropp CB, Panigrahy B, Ostlund E, Schmitt B, Malkinson M, Banet C, Weissman J, Komar N, Savage HM, Stone W, McNamara T, Gubler DJ. Origin of the West Nile virus responsible for an outbreak of encephalitis in the northeastern United States. Science 1999; 286:2333-2337. [ Links ]
32. Lefrançois T, Blitvich BJ, Pradel J, Molia S, Vachiéry N, Martinez D. West Nile virus in Guadeloupe: introduction, spread, and decrease in circulation level: 2002-2005. Ann N Y Acad Sci 2006; 1081:206-215. [ Links ]
33. Lorono-Pino MA, Blitvich BJ, Farfan-Ale JA, Puerto FI, Blanco JM, Marlenee NL, Rosado-Paredes EP, Garcia-Rejon JE, Gubler DJ, Calisher CH, Beaty BJ. Serologic evidence of West Nile virus infection in horses, Yucatan State, Mexico. Emerg Infect Dis 2003; 9:857-859. [ Links ]
34. Mattar S, Edwards E, Laguado J, González M, Álvarez J, Komar N. West Nile virus antibodies in Colombian horses. Emerg Infect Dis 2005; 11:1497-8. [ Links ]
35. Morales-Betoulle ME, Morales H, Blitvich BJ, Powers AM, Davis EA, Klein R, Cordón-Rosales C. West Nile virus in horses, Guatemala. Emerg Infect Dis 2006; 12:1038-1039. [ Links ]
36. Morales MA, Barrandeguy M, Fabbri C, Garcia JB, Vissani A, Trono K, Gutiérrez G, Pigretti S, Menchaca H, Garrido N, Taylor N, Fernández F, Levis S, Enría D. West Nile virus isolation from equines in Argentina. Emerg Infect Dis 2006; 12:1559-1561. [ Links ]
37. Nash D, Mostashari F, Fine A, Miller J, O'Leary D, Murray K, Huang A, Rosenberg A, Greenberg A, Sherman M, Wong S, Layton M. The outbreak of West Nile virus infection in the New York City area in 1999. N Engl J Med 2001; 344:1807-1814. [ Links ]
38. Nicolle L, Gutkin A, Smart G, Dawood M, Drebot M, Van Caeseele P, Giulivi A, Minuk G. Serological studies of West Nile virus in a liver transplant population. Can J Infect Dis Med Microbiol 2004; 15:271-274. [ Links ]
39. Niedrig M, Donoso Mantke O, Altmann D, Zeller H. First international diagnostic accuracy study for the serological detection of West Nile virus infection. BMC Infect Dis 2007; 7:72. [ Links ]
40. Nielsen CF, Reisen WK, Armijos MV, Maclachlan NJ, Scott TW. High subclinical West Nile virus incidence among nonvaccinated horses in northern California associated with low vector abundance and infection. Am J Trop Med Hyg 2008; 78:45-52. [ Links ]
41. Ostlund EN, Andresen JE, Andresen M. West Nile encephalitis. Vet Clin North Am Equine Pract 2000; 16:427-441. [ Links ]
42. Parsons R, Lelic A, Hayes L, Carter A, Marshall L, Evelegh C, Drebot M, Andonova M, McMurtrey C, Hildebrand W, Loeb MB, Bramson JL. The memory T cell response to West Nile virus in symptomatic humans following natural infection is not influenced by age and is dominated by a restricted set of CD8+ T cell epitopes. J Immunol 2008; 181:1563-1572. [ Links ]
43. Quirin R, Salas M, Zientara S, Zeller H, Labie J, Murri S, Lefrançois T, Petitclerc M, Martinez D. West Nile virus, Guadeloupe. Emerg Infect Dis 2004; 10:706-708. [ Links ]
44. Read RW, Rodríguez DB, Summers BA. West Nile virus encephalitis in a dog. Vet Pathol 2005; 42:219-222. [ Links ]
45. Shirafuji H, Kanehira K, Kamio T, Kubo M, Shibahara T, Konishi M, Murakami K, Nakamura Y, Yamanaka T, Kondo T, Matsumura T, Muranaka M And Katayama Y. Antibody responses induced by experimental West Nile virus infection with or without previous immunization with inactivated Japanese encephalitis vaccine in horses. J Vet Med Sci 2009; 71:969-974. [ Links ]
46. Tasha E, Cheryl W, Keith W, Hugh T. Factors associated with West Nile virus disease fatalities in horses. Can Vet J 2007; 48:1137-1145. [ Links ]
47. Van der Meulen KM, Pensaert MB, Nauwynck HJ. West Nile virus in the vertebrate world. Arch Virol 2005; 150:637-657. [ Links ]
48. Venter M, Steyl J, Human S, Weyer J, Zaayman D, Blumberg L, Leman PA, Paweska J, Swanepoel R. Transmission of West Nile Virus during Horse Autopsy. Emerg Infect Dis 2010; 16:573-575. [ Links ]
49. Weir E, Shapiro H. CMAJ. West Nile virus: round five. CMAJ 2004; 170:1669-1670. [ Links ]