Introduction
This article aims to describe jejunal diverticulosis, which, while rare, can compromise the life of those suffering from it; thus, its establishment as a differential diagnosis that causes gastrointestinal bleeding should be prioritized. It also clarifies diagnostic methods and shares the therapeutic options available, including surgical management.
Jejunal diverticulosis was first described in 1794 by Somerling, but it was not until 1906 that Gordinier and Sanpson informed the first case of a patient who underwent surgery for this pathology1-4. Diverticulosis occurs more frequently in the large intestine than in the small intestine, the latter with a reported incidence of 0.6%-2% in autopsy series and a reported radiological incidence of 2%-5%5,6.
In small intestine diverticulosis, the most common site of manifestation is the duodenum, followed by the jejunum and ileum6; the former occurs in equal numbers in both men and women, while jejunoileal diverticula have a greater predominance in men than in women7,8.
Pathophysiologically, diverticula are classified as true or false (pseudodiverticula) due to increased intraluminal pressure. The latter causes herniation of the mucosa and submucosa, mainly at the weakest points, which correspond to sites through which the vasa recta penetrate the muscular layer of the intestine and are covered only by the serous layer9-11.
The etiopathogenesis of small intestine diverticulosis is not clear. So, alterations in intestinal peristalsis, intestinal dyskinesia, and increased intraluminal pressure have been proposed as causes of this abnormality, which would result in the appearance of diverticula in the sites where the mesenteric vessels enter the muscular layer of the small intestine9-11.
The clinical manifestation of these diverticula is related to complications, which occur in 15%-30% of patients, of which up to 10% require surgical management1,2,12. Among these manifestations or complications are diverticulitis and perforation, with percentages between 2% and 6%, obstruction between 2% and 4%, and gastrointestinal bleeding between 3% and 8%. The latter occurs because of erosion and ulceration of the mucosa, which compromises the mesenteric vessels around the neck of the diverticulum1,2,13-16.
Generally, the diagnosis of small bowel diverticula is made incidentally on endoscopic retrograde cholangiopancreatography (ERCP), laparotomy, or enteroscopy13-17. The American Journal of Gastroenterology states that small bowel bleeding should be considered during regular upper and lower endoscopic studies13.
Diagnostic methods involved in the study of gastrointestinal bleeding include upper and lower endoscopy, entero-test, scintigraphy, digital angiography, and computed tomography (CT)(18-20).
Upper digestive endoscopy is the primary diagnostic tool in studying upper gastrointestinal bleeding. Its sensitivity ranges from 92% to 98%, with specificity between 33% and 100%18.
Colonoscopy is the first diagnostic method used in patients with lower gastrointestinal bleeding, with a sensitivity of 50% for detecting the location and cause of bleeding. However, its implementation is hampered by the need for prior bowel preparation, associated with inappropriate colon viewing if large amounts of blood or clots are present18.
Capsule endoscopy is not feasible in an emergency setting, even though it can explore the entire small intestine since viewing is affected if there is an active massive hemorrhage18.
Labeled red blood cell scintigraphy can be used to detect and locate the site of gastrointestinal bleeding. It has a sensitivity of 93% and a specificity of 95%; nonetheless, its value as a diagnostic test is limited since it has up to a 22% chance of false bleeding location19.
Mesenteric angiography offers the advantage of detecting and making a therapeutic intervention through transcatheter embolization. It has a sensitivity between 40% and 86%, and ischemia is the main risk of this procedure19.
Abdominal angiotomography is considered the first-line diagnostic method in a patient with suspected bleeding from the small intestine. It allows viewing the presence or absence of contrast material extravasation into the intestinal lumen. It has a sensitivity of 86% and a specificity of 95% for diagnosing acute gastrointestinal bleeding20.
Once jejunal diverticula are identified as the cause of bleeding, resection of the affected segment of the jejunum with primary anastomosis should be performed20.
Clinical case
A 48-year-old male patient with a history of gastritis was admitted to the emergency department, presenting with a three-day history of lower gastrointestinal bleeding, represented by hematochezia associated with a syncopal episode at the referral site. On admission, the patient was found hemodynamically stable and with severe anemia, for which an endoscopy and colonoscopy were performed with reports of “hiatal hernia” in the upper study and “presence of melena throughout the colon and distal ileum, without masses, polyps, or diverticula” in the lower study.
During the first three days of hospitalization, he persisted with hematochezia, hypotension, and severe anemia that did not correct despite being polytransfused. So, a mesenteric arteriography was requested, in which no lesions were found to explain the bleeding. The patient became hemodynamically unstable, for which he was transferred to the intensive care unit (ICU), requiring transfusion, vasopressor support, and invasive mechanical ventilation. In addition, he had a picture of hematemesis requiring upper digestive tract endoscopy that reported “angiodysplasia of the duodenal bulb, which received sclerotherapy.” Due to the persistence of hemodynamic instability, lower gastrointestinal bleeding, and severe anemia, an abdominal CT angiography was performed, which revealed a “dilation in the ileal small intestinal segment, which could correspond to a Meckel’s diverticulum, with extravasation of the contrast medium through this segment” (Figure 1). Then, a selective embolization was performed using arteriography (Figures 2 and 3).
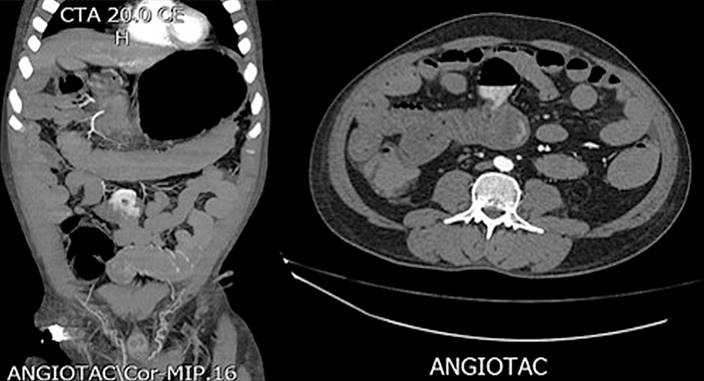
Figure 1 Computed axial tomography showing a thin-walled cystic structure with an air-fluid level corresponding to a jejunal diverticulum. Source: Radiology Service, Hospital Comuneros. Selected by the authors.
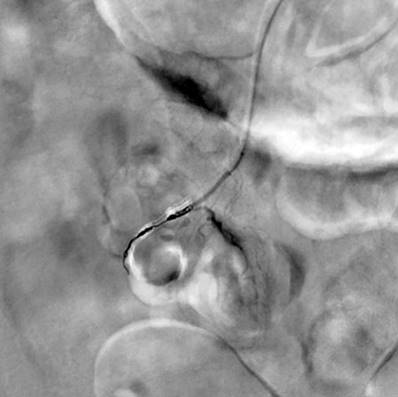
Figure 2 Angiographic embolization of the jejunal diverticulum highlighting a cystic figure compatible with a jejunal diverticulum. Source: Hemodynamics Service, Hospital Comuneros. Selected by the authors.

Figure 3 Angiographic embolization of the diverticulum. Source: Hemodynamics Service, Hospital Comuneros. Selected by the authors.
The patient showed clinical improvement and hemodynamic stability, for which the vasopressor support was removed. Thus, we decided to schedule an intestinal resection. A median laparotomy was performed in which a single jejunal diverticulum was located 40 cm from the angle of Treitz (Figure 4), with residual blood content in the small intestine and colon. Then resection of the diverticulum and jejunojejunal anastomosis were performed. Finally, he was discharged from the hospital on the tenth day of hospitalization.
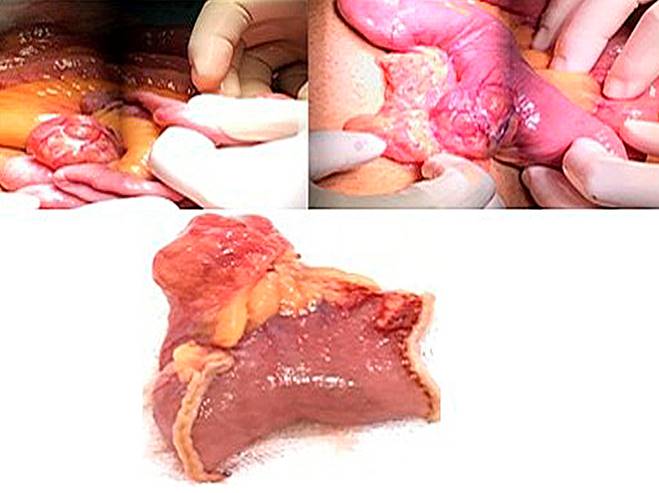
Figure 4 Macroscopic anatomical piece. Presence of an open diverticulum that allows its content to be observed. Source: Surgery Service, Hospital Comuneros. Selected by the authors.
The pathology report described a 4 cm segment of the small intestine with a single pocket-shaped structure on the wall, corresponding to a jejunal diverticulum, with a recent thrombosis of the venous vessels and no ulceration or perforation.
Discussion
Small intestine diverticulosis is a rare entity with even less common complications. The first reports of this pathology were made in 1794, and those of the first complications in 18535; the prevalence of this pathology and its complications are low. The etiology is still unclear, although the currently most accepted aetiological theory indicates that the formation of diverticula can be caused by dysfunction or dyskinesia of the jejunal smooth muscle, which triggers an alteration in its contraction. It causes the development of weak areas, leading to herniation of the mucosa, submucosa, and serosa, but not the muscular layer (false diverticulum).
The case presented is very relevant given that both the clinical manifestation and the presence of complications of this pathology were found in a patient in the fifth decade of life. Notably, the clinical manifestation is more common around the seventh decade of life, as well as gastrointestinal bleeding, which occurs between 3% and 8% of the population with jejunal diverticula7,8. This complication should be considered for causal differential diagnoses of gastrointestinal bleeding since it does not have pathognomonic symptomatology that allows differentiation from other causes, hence the compromised hemodynamic status of patients, as in the previously described case, which required management in the ICU due to hemodynamic instability and severe anemia secondary to profuse and constant bleeding from the diverticulum.
Other complications in jejunal diverticula are diverticulitis and perforation, in 2%- 6% of the population with jejunal diverticulosis, and obstruction, in 2%-4%8,9. Chronic complications include chronic postprandial abdominal pain, emesis, diarrhea or constipation, weight loss, megaloblastic anemia, and steatorrhea; these symptoms reflect chronic complications such as malabsorption, obstruction, and bacterial overgrowth. As we have documented in all the studies reviewed in the literature, its non-specific chronic symptoms have led to this pathology being confused with others such as irritable bowel syndrome, acid-peptic disease, or cholelithiasis, resulting in erroneous diagnoses and, therefore, wrong treatments14-16. Being a rare pathology, its diagnosis is generally made incidentally.
According to the American Journal of Gastroenterology, small bowel bleeding should be considered when standard upper and lower endoscopic studies are available. During the review of the base articles, we found a clinical triad identified in 1971 by Nobles that alerted to the presence of jejunal diverticula, which consisted of diffuse abdominal pain, anemia, and dilated thin intestinal loops. Concerning our patient, Nobles’ triad was added to typical endoscopic methods, which would make us suspect of thin loop diverticula11,12.
The high and low endoscopic methods do not enable this pathology’s diagnosis as they do not display the jejunum, so the contrasted techniques are the first choice. At the same time, abdominal CT angiography is the first-line diagnostic method in patients in whom bleeding from the small intestine is suspected since this method reveals the existence or absence of extravasation of the contrast material in the intestinal lumen, as in the previous case. Mesenteric angiography is another critical diagnostic and therapeutic method, although this procedure’s most relevant complication must be considered: ischemia15-17.
The treatment will be chosen depending on the patient’s complications. Among the options are the conservative method and the surgical method. The former is associated with complications such as diverticulitis or abscesses, and the latter, in the case of gastrointestinal bleeding, is of choice, which allows resection of the affected segment of the jejunum with primary anastomosis.
Conclusions
Jejunal diverticulosis is a pathology that goes unnoticed thanks to its low incidence and little knowledge, which is why it is not diagnosed and managed promptly. Managing diverticular disease of both the small intestine and the colon should focus on the symptoms or complications of patients.
We currently have several technological methods to make a more accurate diagnosis of this pathology, as is abdominal CT angiography, which shows whether or not there is extravasation of the contrast material in the intestinal lumen.











 text in
text in 



