Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Boletín de Investigaciones Marinas y Costeras - INVEMAR
Print version ISSN 0122-9761
Bol. Invest. Mar. Cost. vol.33 no.1 Santa Marta Jan./Dec. 2004
ANNUAL SKELETAL EXTENSION OF TWO REEF-BUILDING CORALS FROM THE COLOMBIAN CARIBBEAN SEA
Extensión esquelética anual de dos corales formadores de arrecifes del Mar Caribe colombiano.
Henry Charry, Elvira M. Alvarado and Juan A. Sánchez
ABSTRACT
The skeletal growth of the scleractinian corals Diploria labyrinthiformis (Linnaeus 1758) and Montastraea annularis (Ellis and Solander 1786) from Isla Grande (north of Rosario islands), Colombian Caribbean, was determined from annual extension increments revealed by X-radiography of 4-6 mm thick slabs obtained along the axis of maximum growth. The skeletal extension average rate for the last 22 years for D. labyrinthiformis was 3.8 mm year–1 (SE 0.10; N = 87). The correlation between growth of D. labyrinthiformis and hours of sunlight was significantly negative. The annual variances of sunlight enhanced annual statistically significant differences of growth in this species. M. annularis showed an average extension growth of 10.6 mm year –1 (SE 0.32; N = 55) during the last 14 years. The increase of M. annularis growth was coincident with the end of nearby dredging activities as well as the decrease of nutrient levels. Nevertheless, the growth rate of this species showed no statistically significant differences through the years and no correlation with variation in sunlight. A low density annual band, wider than high density band in D. labyrinthiformis and narrower in M. annularis, seems to form approximately during April and May in both species, which is coincident with the end of dry season (increase of sea surface temperature, decrease of hours of sunlight and high influence of Dique Channel waters over Rosario islands). The annual bands forming on both species at the study site might be highly related with time of reproduction of each species. Further investigation will permit a better understanding of how some local conditions or coral physiology are related with coral growth at Rosario islands.
KEY WORDS: Growth rates, Diploria labyrinthiformis, Montastraea annularis, Caribbean Sea, Colombia, Coral reefs
RESUMEN
El crecimiento del esqueleto de los corales escleractíneos Diploria labyrinthiformis (Linnaeus 1758) y Montastraea annularis (Ellis & Solander 1786) en Isla Grande (zona norte de las islas del Rosario), Caribe colombiano, se determinó a partir de los incrementos anuales en la extensión revelados por rayos-X de láminas de 4-6mm de grosor obtenidas a lo largo de los ejes de máximo crecimiento. La tasa promedio de extensión esquelética en los últimos 22 años para Diploria labyrinthiformis fue de 3.8 mm año-1 (ES 0.10; N= 87). La correlación entre crecimiento de D. labyrinthiformis y las horas solares fue significativamente negativa. Las variaciones anuales de brillo solar aumentaron diferencias anuales estadísticamente significativas en el crecimiento de esta especie. M. annularis mostró un promedio de extensión anual de 10.6 mm año–1 (ES 0.32; N= 55) durante los últimos 14 años. El incremento del crecimiento de M. annularis fue coincidente con la finalización de actividades de dragado en una localidad cercana y con la disminución de los niveles de nutrientes. No obstante, esta especie no mostró diferencias significativas a través de los años ni correlación con la variación de brillo solar. Una banda anual de baja densidad, más amplia que la de alta densidad en D. labyrinthiformis y más angosta en M. annularis, parece formarse aproximadamante durante abril y mayo en ambas especies, lo cual es coincidente con el final de la época seca (incremento en la temperatura del agua, disminución de horas de brillo solar y mayor influencia del Canal del Dique sobre las islas del Rosario). Las bandas anuales que se forman en las dos especies en el sitio de estudio podrían estar muy relacionadas con la época de reproducción de cada especie. Investigación adicional permitirá un mejor entendimiento de cómo algunas condiciones locales o la fisiología del coral están relacionadas con el crecimiento coralino en las islas del Rosario.
PALABRAS CLAVE: Tasas de crecimiento, Diploria labyrinthiformis, Montastraea annularis, Mar Caribe, Colombia, Arrecifes coralinos
INTRODUCTION
Rosario islands reefs are one of the most important coral ecosystems in Colombia. This area was declared as a National Natural Park in 1978 to preserve the coral community. However, the use of dynamite as a fishing-method, uncontrolled tourism, launches and boats, increase in sea surface temperature and sediment discharge due to dredging of Dique Channel resulted in the loss of coral coverage (Alvarado et al., 1986). Barón et al., (1984) reported a sediment discharge amount of 12,000 tons per day from Dique Channel since 1982, when dredging began. By 1986 a 90% mortality of Acropora palmata and A. cervicornis was evident (Ramírez 1986) and coral coverage decreased (Alvarado et al., 1986). This phenomenon was coincident with El Niño event of 1982 - 1983, when mortality of corals at all Caribbean sites increased and death of the sea urchin Diadema antillarum was reported. Recent studies suggest a low retrieval of Rosario islands coral reef which could be related with dredging detention in 1990 (Alvarado pers. observ., Schönwald unpublished). Sclerochronology provides annual growth rates of specimens which can be used to understand relationships between coral growth and environmental conditions (Dodge and Vaisnys 1980, Hudson et al., 1989, Guzmán et al., 1991). Knutson et al., (1972) demonstrated the presence of alternating high (dark images on the X-radiography positive print) and low (light images on the X-radiography positive print) density bands which are formed annually. Dodge and Vaisnys (1977), investigated the ecological effects of dredging on corals in Bermuda using growth-band analysis. Other environmental factors have been suggested to control coral growth rates: available sunlight (Buddemeier et al., 1974), water temperature (Dodge 1981, Dodge and Lang 1983, Hubbard and Scaturo 1985), oil effects (Dodge et al., 1984, Guzmán et al., 1991, Guzmán and Jarvis 1996), suspended particulate matter (Tomascik and Sander 1985) and sedimentation (Rogers 1990). However, the effect of sediments and eutrophication over coral growth remain uncertain. Previous studies have shown the negative effects of both dredging and sediments over coral skeletal extension (Dodge and Vaisnys 1977, Bak 1978) and positive effects (Hudson 1981, Carricart-Ganivet and Merino 2001), as well as contradictory effects of environmental variables over corals from the same study sites (Dodge and Thomson 1974, Logan and Tomascik 1991). Dodge and Brass (1984) found that density, calcification and extension growth rates are complementary variables and when they are combined, a better understanding between growth rates and the environment is obtained. Consequently, recent studies suggest that massive corals growth rates may be poor indicators of coral reef health where they are influenced by sedimentation (Edinger et al., 2000) and indeed they can modulate their skeletal growth independently of the amount of calcium carbonate available (Carricart-Ganivet and Merino 2001). In this article we determine the extension growth rates of Diploria labyrinthiformis (Linnaeus 1758) and Montastraea annularis (Ellis and Solander 1786) at Isla Grande (north of Rosario islands), including the possible role of time of reproduction over growth rate results. D. labyrinthiformis is a common species at Isla Grande, but has been poorly studied in Colombia and elsewhere in the Caribbean. M. annularis is a dominant species at the study area. According to the description of Knowlton et al., (1992), this study was done sensu stricto with morphotype I of the sibling species Montastraea annularis.
MATERIALS AND METHODS
Collecting site
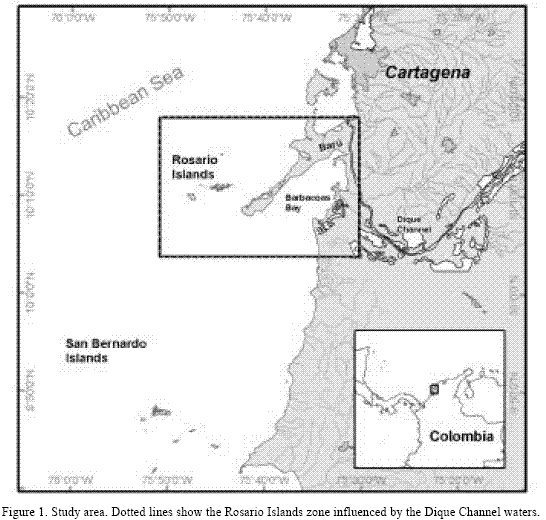
Coral specimens of D. labyrinthiformis and M. annularis were collected on May 1998, from the Isla Grande north reef at Rosario islands (Figure 1), Colombia (10º10’21” - 10º11’28”N and 75º42’36” - 73º43’52” W). Trade winds are the most important macroclimatic factor in the zone. Two principal climatic periods are known: dry season, between December and April, with high influence of Trade winds and, wet (rainy) season, between May and November. Dique Channel waters (an artificial arm of Magdalena River which crosses the country) pass through Barbacoas Bay and discharge into Rosario islands during the wet season. During the dry season the Dique Channel influences Rosario islands waters less (Leble and Cuignon 1987, Alvarado and Corchuelo 1992).
Collection and laboratory description
Fifteen colonies of each species between 20 – 50 cm in diameter were obtained at depths of 6.0 – 11.0 m by SCUBA diving, using hammer and chisel. Corals were sun exposed for one week, soaked in fresh water with 2% sodium hypochlorite for 24 – 72 h, rinsed and dried with sun exposure. Five specimens of M. annularis and four specimens of D. labyrinthiformis were cut using a 24” diamond rock saw into 4 – 6 mm thick slabs through the axis of maximum growth of each colony. The slabs were X-rayed with an Ergophos IV-SiemensTM machine with an exposure ranging from 42 – 60 kv and 12 – 30 mas over 0.12–0.30 s, and a film-target distance of ca. 30 cm. The negative prints of the X-rays were developed on photographic paper to obtain positive contact prints. One transect along the axis of maximum growth was analyzed by measuring with vernier calipers (resolution 0.1 mm) the distance between each couplet of light and dark bands which were defined as annual growth increments (Knutson et al., 1972, Buddemeier et al., 1974, Dodge and Vaisnys 1980, Logan and Tomascik 1991). In cases where the banding was not clearly defined, another transect near the axis of maximum growth was measured.
Environmental data
Hours of sunlight at Rosario islands were obtained from Instituto de Hidrología, Meteorología y Estudios Ambientales (IDEAM) archives. Sea-surface temperature was taken with the help of nighttime satellite observations by the National Oceanic and Atmospheric Administration (NOAA). Nutrient and turbidity Information was obtained from previous studies (Barón et al.,1984, Ramírez 1986, Alvarado and Corchuelo 1992, among others).
Statistical analysis
To transform the mean and variance into more homogeneous data with respect to time, standardization procedure of ring-width analysis was followed (Fritts 1976). The annual linear growth values of each coral were divided by its particular mean growth rate, providing index chronologies with an approximate value of 1.0. The index values of all corals were averaged by year to form an index master chronology (Figure 2). To smooth the year-to-year variability (high frequency variation) a three-year moving average was applied to each chronology. The index master chronology was correlated with sea-surface temperature and hours of sunlight using the Spearman test. To quantify yearly variation in growth a Repeated Measures ANOVA was used, with dredging (presence/non presence) and time (years) as factors. This kind of analysis is required because successive growth bands are measured in the same corals. The null hypothesis tested was that there are no statistically discernible differences in the mean growth rates between the years analyzed on each species, the index master chronologies of both species, and between the years 1975 – 1981 (before dredging period), 1983 – 1989 (dredging period) and 1991 - 1997 (after dredging period). With Levene and Bartlett tests (i.e. homogeneity of variance tests) and with normal probability plots (i.e. normality test) the ANOVA assumptions were tested.

RESULTS
The skeletal extension growth rate of D. labyrinthiformis was 3.8 mm year -1 (SE ± 0.10; N = 87) and 10.6 mm year -1 (SE ± 0.32; N = 56) for M. annularis at Isla Grande. The number of annual growth bands ranged from 21 to 22 years for D. labyrinthiformis and 6 to 14 years in M. annularis. There was not a clear pattern related to growth variation of D. labyrinthiformis (Figure 2A), in fact the annual growth shifted into an alternated variation along the years studied (up and down 1.0). In contrast, M. annularis (Figure 2B) showed a clear coincidence to increase growth when last dredging activities finished (1990-1991). In accordance with the time of collection and what is shown in the apex of corals on the contact prints, low density (LD band) of both species seems to be formed (or to have just formed) in April-May. The LD band formation coincides with the increase of sea-surface temperature, the decrease of hours of sunlight, and a higher influence of Dique Channel waters over Rosario Islands. High density (HD) bands were generally narrower than LD band portions in D. labyrinthiformis, while in M. annularis HD band were wider than LD band portions. Samples X-radiograph is shown in figure 3.

The Repeated Measures ANOVA of table 1 indicates significant differences in annual growth along 1975 - 1997 period in D. labyrinthiformis (F = 2.39; P < 0.005) and no significant differences in the annual growth of the 1984 - 1997 period in M. annularis. Tukey HSD test shows that recent years (1995, 1996) have significant differences in relation to the years 1977, 1983 and 1985 in D. labyrinthiformis. We found significant statistical differences between growth variation of both species (Two-way Repeated Measures ANOVA: F = 4.97; P < 0.000) during 1984 - 1997 period (Figure 4). Repeated-measures ANOVA of D. labyrinthiformis growth rates were not significant for before (1975 - 1981), during (1983 - 1989) and after (1991 - 1997) the last dredging (F = 3.1798; P > 0.05: Table 1). Using years as a factor, significant differences for the annual growth rate during 1991 - 1997 period were found in this species, therefore another repeated-measures ANOVA was done and it shows no significant differences before or during the last dredging (excluding 1991 - 1997 period). Growth rates in M. annularis during and after dredging were not statistically significant (F = 2.705; P > 0.05: Table 1) in spite of the increase of growth after last dredging activities.


Results of Spearman correlation test for annual growth rates of D. labyrinthiformis (± standard error) and M. annularis (± standard error) with sea-surface temperature (SSTs in °C) and annual sunlight (hours ± standard error) are included in Table 2. Master index of both species are different in each test because the growth period analyzed with sea surface temperature was 1984-1996 while with annual sunlight was 1984-1997. Sea surface temperature at Isla Grande varied very little during 1984-1996 (average range = 27.9 - 29.0 °C). Indeed, sea surface temperature was not significantly negatively correlated with growth of D. labyrinthiformis (Table 2). Contrastingly, growth of D. labyrinthiformis was significantly negatively correlated with hours of sunlight (r = - 0.60; P < 0.039). Annual growth of M. annularis was positively but not significantly correlated with both sea surface temperature and hours of sunlight (Table 2).

DISCUSSION
In the present study, growth rates of D. labyrinthiformis and M. annularis are slightly higher than those reported in previous studies at high and low latitudes of the Caribbean (Table 3). Latitudinal growth rate differences are controlled by reduction in winter water temperatures and light levels with increasing latitude. Consequently, coral growth rates are inversely related to latitude in the Atlantic (Logan and Tomascik 1991, Logan et al., 1994).

However, growth rates are also controlled by local environmental conditions. D. labyrinthiformis growth rates at Isla Grande seems to be influenced by changes in hours of sunlight, while a clear increment in skeletal extension of M. annularis was seen when dredging activities finished (1990 - 1991). Nevertheless, it was not statistically significant. It is possible that tolerance to light and sediment level are the principal causes of the inverse results showed between correlation of growth in both species with hours of sunlight. In addition, this could be producing the significant differences in skeletal extension of D. labyrinthiformis and M. annularis in the last 14 years. Symbiont species of hosts have diverse mechanisms of photo-acclimation (Chang et al., 1983). Iglesias-Prieto and Trench (1994) showed that different species of symbiotic dinoflagellates have several photosynthetic responses under identical farming conditions. Leletkin and Zvalinsky (1981) provided evidence that at a low light intensity the photosynthesis rate of zooxanthellae is greater compared with those adapted to a normal light intensity. Increase of nutrients may also have an effect on zooxanthellae. For example, Marubini and Davies (1996) report an increase in areal density of dinoflagellates and a decrease in extension growth in Porites porites and M. annularis due to nitrate enrichment.
Sediment levels per se affect coral species in a different way. It is well known that species differ in their capability to remove sediments which are in contact with their colonies (e.g. Rogers 1990). D. labyrinthiformis and D. strigosa have shown better capability to remove sediments than M. annularis and M. cavernosa (Hubbard and Pocock 1972, Rogers 1990 and references therein). Dredging activities at the Dique Channel started again in 1982 and its influence at the Rosario islands was evident (Barón et al., 1984). Alvarado and Corchuelo (1992), observed the influence of the Dique Channel waters in the southern Rosario islands during wet season (May - November), while during dry season they found a slight influence in the north. It is also important to note that amounts of nutrients and turbidity increased between 1982 - 1990 at Isla Grande (Alvarado et al., 1986, Alvarado and Corchuelo 1992). In previous studies (e.g. Dodge et al., 1974, Dodge and Vaisnys 1977, Bak 1978, Dodge 1981, Cortés and Risk 1985, Tomascik 1990), dredging activities near coral reefs were found to produce a reduction in coral growth and increase of coral mortality. Dredging activities near coral reefs increase turbidity, reducing the available light for photosynthesis and increasing the sediment load on corals, which have sub-lethal and lethal effects (Rogers 1990). Notwithstanding, Tomascik and Sander (1985) explained that coral extension may increase at sites with moderate increments of suspended particulate matter in the water column, because they are a significant source of additional food for the polyps. Dodge and Brass (1984) and Heiss (1996) determined that higher levels of phosphate reduce calcification and growth, while other nutrients promote coral extension. Hence, recent studies have discussed that growth rates are poor indicators of reef health and suggest that high extension and low density skeletal growth could be a result of eutrophic conditions (Edinger et al., 2000).
We found that low density (LD) band formation of both species studied is formed when Dique Channel waters have high incidence over Rosario Islands, as has been suggested by some authors (Edinger et al., 2000, Carricart-Ganivet and Merino 2001). Generally, the annual band formation has been related with environmental changes, such as available light and water temperature (Highsmith 1979), salinity (related with precipitation changes), and time of reproduction (Guzmán and Cortés 1989). In this study, LD band deposition of both D. labyrinthiformis and M. annularis seems to happen during April-May, during the transition from dry to rainy season (highest water temperature and lowest time of sunlight). Dodge and Thomson (1974) found that formation of LD band of D. labyrinthiformis in Bermuda takes place in summer (at high water temperature), while Logan and Tomascik (1991) found that LD band formation in the same species at the same locality occurs during winter period (lowest sea water temperature). Previous studies of M. annularis in the Caribbean have related HD band formation with summer and autumn when sea water temperature increases and hours of sunlight decrease (Macintyre and Smith 1974, Highsmith 1979, Hudson 1981). At Rosario islands, sea surface temperature variation is insignificant between dry and rainy period (27.6 ± 0.54 °C and 28.6 ± 0.58 °C, respectively), this indicates that sea surface temperature does not have a significant effect on seasonal growth rates of both studied species at Isla Grande. Guzmán and Cortés (1989) also suggested that sea water temperature does not influence coral growth rate in the eastern Pacific at Costa Rica (low latitude) due to the small variation between dry and rainy period.
Wellington and Glynn (1983) suggested that the lower growth rate in Panama corals could be related to the energy spent in reproduction. Guzmán and Cortés (1989) found that in Porites lobata LD band was formed during the dry season (higher hours of sunlight) and when corals were not reproducing. At Isla Grande, gametogenic cycle of D. labyrinthiformis begins between August-October and finishes during May-June, coinciding in general with dry period and higher hours of sunlight. According to this, main expenditure of energy (maturation of gonads) occurs during the dry season, while during May-June this expenditure declines due to gamete liberation (Alvarado et al., in press.). In contrast, gametogenic cycle of M. annularis at Isla Grande begins on April and finishes with gamete liberation during September - October (Sánchez et al., 1999), coinciding with rainy season and less hours of sunlight. In summary, the difference observed between annual bands deposition of both species at Isla Grande might be highly related with time of reproduction of each species inasmuch as LD band formation in M. annularis coincides with the rainy season and the beginning of the gametogenic cycle using a high expenditure of energy, while in D. labyrinthiformis coincides with the dry season and the end of the cycle. Coral reproductive cycle is influenced by the life strategy of each organism. In corals, and generally in any organism, reduction of growth means a reduction in the future reproduction and the survival (Hall and Hughes 1996). Corals do not stop growing in order to reproduce, they decrease their growth rate during reproduction periods. It appears to be clear in both studied species in which skeletal extension is wider before their reproductive cycle begins.
This is the first sclerochronological study in the Colombian Caribbean, which allowed retrospective skeletal extension growth analysis. It is difficult for us not to refer to the diminishing of coral population at Isla Grande during at least the two previous decades. At the first sight, some authors related this significant occurrence with dredging activities, but indeed there were other confounding environmental changes (i.e. El Niño event, death of the sea urchin Diadema antillarum, among others). We attempted to find any skeletal coral growth rate change during the last two decades over the study area, even though the direct incidence of the dredging activities was not the main purpose of this research. However, we can not confirm any assumption due to the lack of information such as environmental data, sub-annual bands formation or complementary growth rates variables. Nowadays, it is well known that density, calcification and extension growth rates are complementary variables and when they are studied in concert a better understanding between growth rates and the environment is obtained (Dodge and Brass 1984). Nevertheless, we report here LD band formation during high incidence of Dique Channel waters over Rosario islands and an apparent relation of density bands formation with time of reproduction of each species. Additional investigation of these species is necessary to understand how coral physiology or other local conditions are related with coral growth at Rosario Islands.
ACKNOWLEDGMENTS
This research was part of the project “Reproduction, growth and transplantation survivorship of the coral species Montastraea annularis, Diploria labyrinthiformis and Porites astreoides in the National Natural Park Corales del Rosario and San Bernardo, Colombian Caribbean”, grant to EM Alvarado, JA Sánchez and R García (Jorge Tadeo Lozano University and COLCIENCIAS 1202-09-221-96). We thank R García for the contribution with the project. R García, OL Arenas, MF Gil, LH Chasqui, A Alfonso and M Morelos for the help in the field work. HM Guzmán and RD Llinás for useful advice and material support. RE Dodge (Nova Southeastern Oceanographic Center) for providing important articles. I Barraquer for X-raying. M Vargas and INGEOMINAS for cutting the corals. LM Moncada, OL Rodriguez and all laboratory personnel at Universidad de Bogotá Jorge Tadeo Lozano for their support in preliminary chemical tests. S Dell and A Vaughn for radiograph scanning. D Trujillo and A Galán for providing statistical programs. J Dillard and R Slusar for grammar review. Comments from JP Carricart-Ganivet and three anonymous reviewers notably improved the manuscript. Contribution No. 61 Museo del Mar, Centro de Investigaciones Científicas, Universidad de Bogotá Jorge Tadeo Lozano.
REFERENCES
Alvarado, E.M., F. Duque, L. Flórez-Gonzalez and R. Ramírez. 1986. Evaluación cualitativa de los arrecifes coralinos de las islas del Rosario (Cartagena-Colombia). Bol. Ecotrópica: Ecosistemas Tropicales 15: 1-30. [ Links ]
Alvarado, E.M. and M.C. Corchuelo. 1992. Los nutrientes, la temperatura y la salinidad provenientes del Canal del Dique como factores de deterioro en el Parque Nacional Natural Corales del Rosario (Cartagena, Colombia). VIII Seminario Nacional de Ciencias y Tecnologías del Mar, Santa Marta, Colombia. 1: 277-287. [ Links ]
Alvarado, E.M., R. Garcia-U and A. Acosta. In press. Sexual reproduction of the reef-building coral Diploria labyrinthiformis (Linnaeus, 1758), (Scleractinia: Faviidae), Colombian Caribbean. Rev. Biol. Trop. [ Links ]
Bak, R.P.M. 1978. Lethal and sublethal effects of the dredging on reef corals. Mar. Poll. Bull. 9 (1): 14-16. [ Links ]
Barón, P.A., T. Fernández and A. Pion. 1984. Evaluación del impacto producido por el Canal del Dique como principal fuente de contaminación de la Bahía de Barbacoas y las islas del Rosario. Informe de avance INDERENA-CIP, Bogotá, Colombia. 17pp. [ Links ]
Buddemeier, R.W., J.E. Maragos and D.W. Knutson. 1974. Radiographic studies of reef coral exoskeletons: rates and patterns of coral growth. J. Exp. Mar. Ecol. 14: 179-200. [ Links ]
Carricart-Ganivet, J.P., G. Horta-Puga, M.A. Ruíz-Zárate and E. Ruíz-Zárate. 1994. Tasas retrospectivas de crecimiento del coral hermatípico Montastrea annularis (Scleractinia: Faviidae) en arrecifes al sur del Golfo de México. Rev. Biol. Trop. 42 (3): 515-521. [ Links ]
Carricart-Ganivet, J.P. and M. Merino. 2001. Growth responses of the reef-building coral Montastraea annularis along a gradient of continental influence in the southern Gulf of Mexico. Bull. Mar. Sci. 68 (1): 133-146. [ Links ]
Chang, S.S., B.B. Prézelin and R.K. Trench. 1983. Mechanisms of photoadaptation in three strains of the symbiotic dinoflagellate Symbiodinium microadriaticum. Mar. Biol. 76: 219-229. [ Links ]
Cortés, J. and M.J. Risk. 1985. A reef under siltation stress: Cahuita, Costa Rica. Bull. Mar. Sci. 36 (2): 339-356. [ Links ]
Dodge, R.E. and J. Thomson. 1974. The natural radiochemical and growth records in contemporary hermatypic corals from the Atlantic and Caribbean. Earth Plan. Sci. Lett. 23: 313-322. [ Links ]
Dodge, R.E., R.C. Aller and J. Thomson. 1974. Coral growth related to resuspension of bottom sediments. Nature. 247: 574-577. [ Links ]
Dodge, R.E. and J. R. Vaisnys. 1977. Coral populations and growth patterns: Responses to sedimentation and turbidity associated with dredging. J. Mar. Res. 35: 715-730. [ Links ]
______.1980. Skeletal growth chronologies of recent and fossil corals. p 493-517. In Rhoads D.C. & R.A. Lutz (Eds.). Skeletal growth of aquatic organisms. Plenum Press, New York. 750pp. [ Links ]
Dodge, R.E. 1981. Growth characteristics of reef-building corals within and external to a naval ordinance range: Vieques, Puerto Rico. Proc. 4th. Int. Coral Reef Symp. 2: 241-248. [ Links ]
Dodge, R.E. and J.C. Lang. 1983. Environmental correlates of hermatypic coral (Montastrea annularis) growth on the East Flower Gardens Bank, northwest Gulf of México. Limnol. Oceanogr. 28 (2): 228-240. [ Links ]
Dodge, R.E. and G.W. Brass. 1984. Skeletal extension, density and calcification of the reef coral, Montastrea annularis: St. Croix, U. S. Virgin Islands. Bull. Mar. Sci. 34 (2): 288-307. [ Links ]
Dodge, R.E., S.E. Wyers, H.R. Frith, A.H. Knap, S.R. Smith and T.D. Sleeter. 1984. The effects of oil dispersants on the skeletal growth of the hermatypic coral Diploria strigosa. Coral Reefs. 3: 191-198. [ Links ]
Edinger, E.N., G.V. Limmon, J. Jompa, W. Widjatmoko, J.M. Heikoop and M.J. Risk. 2000. Normal coral growth rates on dying reefs: Are coral growth rates good indicators of reef health?. Mar. Poll. Bull. 40 (5): 404-425. [ Links ]
Fritts, H.C. 1976. Tree rings and climate. Academic Press, London. 567pp. [ Links ]
Ghiold, J. and P. Enos. 1982. Carbonate production of the coral Diploria labyrinthiformis in South Florida patch reefs. Mar. Geol. 45: 281-296. [ Links ]
Guzmán, H.M. and J. Cortés. 1989. Growth rates of eight species of scleractinian corals in the Eastern Pacific (Costa Rica). Bull. Mar. Sci. 44 (3): 1186-1194. [ Links ]
Guzmán, H.M, J.B.C. Jackson and E. Weil. 1991. Short–term ecological consequences of a major oil spill on Panamanian subtidal reef corals. Coral Reefs. 10: 1–12. [ Links ]
Guzmán, H.M. and K.E. Jarvis. 1996. Vanadium century record from Caribbean reef corals: a tracer of oil pollution in Panama. Ambio. 25 (8): 523-526. [ Links ]
Hall, V.R. and T.P. Hughes. 1996. Reproductive strategies of modular organisms: Comparative studies of reef-building corals. Ecology. 77: 950-963. [ Links ]
Heiss, G.A. 1996. Annual band width variation in Porites sp. from Aqaba, Gulf of Aqaba, Red Sea. Bull. Mar. Sci. 59 (2): 393-403. [ Links ]
Highsmith, R.C. 1979. Coral growth rates and environmental control of density banding. J. Exp. Mar. Biol. Ecol. 37: 105-125. [ Links ]
Hubbard, J.A.E.B. and Y.B. Pocock. 1972. Sediment rejection by recent scleractinian corals: a key to palaeo-environmental reconstruction. Geol. Rdsch. 61: 598-626. [ Links ]
Hubbard, D.K. and D. Scaturo. 1985. Growth rates of seven species of scleractinian corals from Cane Bay and Salt River, St. Croix, USVI. Bull. Mar. Sci. 36 (2): 325-338. [ Links ]
Hudson, J.H. 1981. Growth rates in Montastrea annularis: a record of environmental change in Key Largo Reef Marine Sanctuary, Florida. Bull. Mar. Sci. 31: 444-459 [ Links ]
Hudson, J.H., G.V.N Powell, M.B. Robblee and T.J Smith. 1989. A 107 – year–old coral from Florida Bay: barometer of natural and man–induced catastrophes?. Bull. Mar. Sci. 44 (1): 283-291. [ Links ]
Iglesias-Prieto R. and R.K. Trench. 1994. Acclimation and adaptation to irradiance in symbiotic dinoflagellates. I. Responses of the photosynthetic unit to changes in photon flux density. Mar. Ecol. Prog. Ser. 113: 163-175. [ Links ]
Knowlton, N., E. Weil, L.A. Weight and H.M. Guzmán. 1992. Sibling species in Montastraea annularis, coral bleaching, and the coral climate record. Science 255: 330-333. [ Links ]
Knutson, D.W., R.W. Buddemeier and S.V. Smith. 1972. Coral chronometers seasonal growth bands in reef corals. Science. 177: 270-272. [ Links ]
Leble, S. and R. Cuignon. 1987. El Archipiélago de las Islas del Rosario, estudio morfológico, hidrodinámico y sedimentológico. Bol. Cient. CIOH. 7: 37-52. [ Links ]
Leletkin, V.A. and V.I. Zvalinsky. 1981. Photosynthesis of coral zooxanthellae from different depths. Proc. 4th Int. Coral Reef Symp. 2: 33-37. [ Links ]
Logan, A. and T. Tomascik. 1991. Extension growth rates in two coral species from high-latitude reefs of Bermuda. Coral Reefs. 10: 155-160. [ Links ]
Logan, A., L. Yang and T. Tomascik. 1994. Linear skeletal extension rates in two species of Diploria from high-latitude reefs in Bermuda. Coral Reefs. 13: 225-230. [ Links ]
Macintyre, I.G. and S.V. Smith. 1974. X–radiograph studies of skeletal development in coral colonies. Proc. 2nd Int. Coral Reef Symp. 2: 277-287. [ Links ]
Marubini, F. and P-S. Davies. 1996. Nitrate increases zooxanthellae population density and reduces skeletogenesis in corals. Mar. Biol. 127: 319-328. [ Links ]
Ramirez, A. 1986. Ecología descriptiva de las llanuras madreporarias del Parque Nacional Submarino de los Corales del Rosario (Mar Caribe), Colombia. Bol. Ecotrópica: Ecosistemas Tropicales 14: 34-63. [ Links ]
Rogers, C.S. 1990. Responses of coral reefs and organisms to sedimentation. Mar. Ecol. Prog. Ser. 62: 185-202. [ Links ]
Sánchez, J.A., E.M. Alvarado, M.F. Gil, H. Charry, O.L. Arenas, L.H. Chasqui and R.P. García. 1999. Synchronous mass spawning of Montastraea annularis (Ellis & Solander) and Montastraea faveolata (Ellis & Solander) (Faviidae: Scleractinia) at Rosario Islands, Caribbean coast of Colombia. Bull. Mar. Sci. 65 (3): 873-879. [ Links ]
Tomascik, T. and F. Sander. 1985. Effects of eutrophication on reef–building corals. I. Growth rate of the reef–building coral Montastrea annularis. Mar. Biol. 87 (2): 143–155. [ Links ]
Tomascik, T. 1990. Growth rates of two morphotypes of Montastrea annularis along an eutrophication gradient, Barbados, W. I. Mar. Poll. Bull. 21 (8): 376-381. [ Links ]
Wellington, G.M. and P.W. Glynn. 1983. Environmental influences on skeletal banding in eastern Pacific (Panama) corals. Coral Reefs. 1: 215-222. [ Links ]
DATE RECEIVED: 13/05/03 DATE ACCEPTED: 24/09/04
ADDRESSES OF THE AUTHORS:
503 West End Avenue, Elizabeth, NJ 07202 USA E-mail: hcharry@yahoo.com (HC).
Centro de Investigaciones Científicas (Museo del Mar), Universidad de Bogotá Jorge Tadeo Lozano, calle 22 No. 3-30 piso 7, Bogotá D.C., Colombia. E-mail: elvira.alvarado@utadeo.edu.co (EMA).
Departamento de Ciencias Biológicas, Universidad de los Andes, carrera 1E No 18 A –10 (J304A) PO Box 4976, Santafé de Bogotá D.C., Colombia. E-mail: juansanc@uniandes.edu.co (JAS).













